Lewy Body Disease
Original Editor - Henry Tan, Kevin Lam, Alex Mong, Andrew To as part of the Queen's University Neuromotor Function Project
Top Contributors - Henry Tan, Andrew To, Alex Mong, Lucinda hampton, Kim Jackson, Kevin Lam, Vidya Acharya, Claire Knott, 127.0.0.1, Evan Thomas and Lauren Lopez
Introduction[edit | edit source]
Lewy body disease or Lewy body dementia (LBD), is a neurodegenerative disease (specifically a synucleinopathy) related to Parkinson disease.[1]
There are 2 types of LBD generally described in the literature:[2]
- Dementia with Lewy Bodies: dementia occurring first or within one year of movement disorder.
- Parkinson Disease Dementia: dementia occurring in a patient who receives a diagnosed of Parkinson's disease and then develops dementia symptoms after one year or more of the diagnosis.
People with either type generally develop similar symptoms as the disease progresses. Distinct Parkinsonian symptoms such as slowness of movement, rigidity, REM sleep behaviour disorder and visual hallucinations can help to distinguish DLB from Alzheimer’s disease.[3]
Epidemiology[edit | edit source]
- Occurs in older patients (onset typically in 50-70 years of age), and is sporadic. However a family history of LBD and Parkinson disease will increase a person's risk.[4]
- LBD is one of the most common causes of dementia (accounting for up to 20% to 30% of cases[4]) along with Alzheimers disease (AD)[3].
Etiology[edit | edit source]
The etiology of LBD is unknown. Genetics, environmental factors, and changes linked to aging, may have a role and research is still ongoing.[5]
LBD has been found to be strongly linked to a protein called alpha-synuclein. The abnormal accumulation of this protein in certain regions of the brain causes dramatic cognitive and motor deficits affecting behaviour, mood, movement, and thinking.
Clinical Presentation[edit | edit source]
The clinical features of LBD are the consequence of the blockage of information transfer from the striatum to the cortex, more notably the frontal lobe.
Core features:[6]
- Fluctuating cognitive impairment especially in executive function, attention and alertness
- Visuospatial impairment, including visual hallucinations (detailed and vivid)
- Concurrent parkinsonian symptoms may be present but are less common, more frequently occurring years after the onset of dementia eg early extrapyramidal features (dystonia, akathisia, muscle rigidity, bradykinesia, tremor, tardive dyskinesia
Watch this 3 minute video showing a personal story "It happened little by little. First he would forget things, then he'd lose track of what he was doing. LBD took over the life of the man you're about to meet."
Pathophysiology[edit | edit source]
The characteristic feature of dementia with Lewy bodies is the accumulation of Lewy bodies throughout the brain. These intracellular inclusions result from the aggregation of misfolded α-synuclein. Neurofibrillary tangles are also present, however they lack an amyloid core, as seen in AD[1].
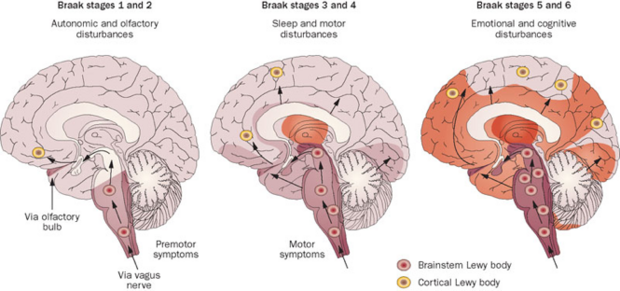
Source (http://labiotech.eu/major-cns-disease-milestones-in-biotech-2015/)
Diagnostic Procedures[edit | edit source]
It is important to realise that there is a significant overlap between many neurodegenerative diseases, and that a clear-cut distinction between entities is not always possible. No precise test can accurately diagnose LBD. A thorough assessment is useful to reach an alternative working diagnosis (or rules out similar conditions):
- Detailed history and examination
- Assessment of cognitive function
- Blood tests (e.g., vitamin B12 levels, chemistry panel, thyroid profile, syphilis, HIV) to rule out other causes of dementia
- Imaging studies (e.g., CT scan, MRI scan, SPECT scan, PET scan)
- Cerebrospinal fluid examinations have no significant role when conducted in these patients
- Sleep evaluation for REM sleep behavior disorder[5]
Management[edit | edit source]
There is no effective treatment for LBD and the condition is progressive. Pharmacological Management includes cholinesterase inhibitors. They treat the cognitive symptoms of LBD and are central to treatment. eg rivastigmine, galantamine, and donepezil. Acetylcholine is an important neurotransmitter for memory. People with eg LBD have reduced levels present in their brains. Cholinesterase inhibitors inhibit this enzyme , improving brain network communication.[4].
The other pharmacological agents are used to treat behavioural symptoms.
- Home care nurses play a crucial role in regularly assessing the patient and providing support services.
- Education of the caregiver is essential, the loved ones needing to be aware of the behavior changes, hallucinations, and fluctuations in cognition. Caregivers have to monitor the patient closely as they have a love level of functioning, with most unable to perform ADLs and are prone to falls and aspiration pneumonia.
- The pharmacist needs to educate caregivers that no medical therapy will cure the cognitive changes and the drugs simply manage behaviour and motor deficits ( and many have adverse effects).
- A mental health nurse is often needed as depression is common. Close communication between members of the interprofessional team is vital to improve outcomes.[5]
Prognosis[edit | edit source]
LBD has a poor prognosis, with an average life expectancy from initial diagnosis to death 5-8 years. Death usually occurs from one of the many complications eg falls, immobility, cardiac complications, medication side effects, pneumonia, swallowing problems, suicide.[4]
Physiotherapy Management[edit | edit source]
Physiotherapy for Lewy Body Disease is similar to that of Parkinson’s Disease. It can help manage parkinsonism that is prevalent in LBD by providing intervention such as strengthening and flexibility exercises and gait training. Aerobic exercise should be included to optimise cardiovascular fitness.[7] Physiotherapy is especially helpful in improving balance and postural stability to minimize the risk of falls. With the addition of exercise, non-motor symptoms such as cognition, sleep and fatigue will improve. As the disease progresses and the dementia increases, exercise can be hard to do. Therefore, it is important to incorporate exercise in the early and middle stages of Lewy Body Disease. A study[8] suggests that a high-intensity functional exercise program has positive outcomes on balance in these patients.
Tips to help make exercise easier to maintain:[7]
- Provide visual cues by demonstrating exercises
- Play upbeat music or music the person enjoys
- Arrange exercise classes or include the support/care-person
- Do exercises in sitting
- Make exercise fun and enjoyable
References[edit | edit source]
- ↑ 1.0 1.1 Radiopedia Dementia with Lewy bodies Available:https://radiopaedia.org/articles/dementia-with-lewy-bodies (accessed 13.9.2022)
- ↑ McKeith IG, Galasko D, Kosaka K, Perry EK, Dickson DW, Hansen LA, Salmon DP, Lowe J, Mirra SS, Byrne EJ, Lennox G. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB) Report of the consortium on DLB international workshop. Neurology. 1996 Nov 1;47(5):1113-24. PMID:8909416
- ↑ 3.0 3.1 National Institute on Aging. Lewy Body Dementia: Information for Patients, Families, and Professionals. (Accessed 4 May 2017). https://www.nia.nih.gov/alzheimers/publication/lewy-body-dementia/basics-lewy-body-dementia
- ↑ 4.0 4.1 4.2 4.3 Haider A, Spurling BC, Sánchez-Manso JC. Lewy body dementia. InStatPearls [Internet] 2022 Oct 23. StatPearls Publishing.Available:https://www.ncbi.nlm.nih.gov/books/NBK482441/ (accessed 6.10.2023)
- ↑ 5.0 5.1 5.2 Haider A, Spurling BC, Sánchez-Manso JC. Lewy body dementia. InStatPearls [Internet] 2021 Jul 12. StatPearls Publishing.Available:https://www.ncbi.nlm.nih.gov/books/NBK482441/ (accessed 13.9.2022)
- ↑ Gnanalingham KK, Byrne EJ, Thornton A, Sambrook MA, Bannister P. Motor and cognitive function in Lewy body dementia: comparison with Alzheimer's and Parkinson'ss. Journal of Neurology, Neurosurgery & Psychiatry. 1997 Mar 1;62(3):243-52. http://jnnp.bmj.com/content/jnnp/62/3/243.full.pdf
- ↑ 7.0 7.1 Lewy Body Dementia Association. What is LBD? Available from: https://www.lbda.org/category/3437/what-is-lbd.htm [Accessed May 5, 2017]
- ↑ Sondell A, Littbrand H, Holmberg H, Lindelöf N, Rosendahl E. Is the Effect of a High-Intensity Functional Exercise Program on Functional Balance Influenced by Applicability and Motivation among Older People with Dementia in Nursing Homes?. The journal of nutrition, health & aging. 2019 Dec 1;23(10):1011-20.









