Introduction to Spinal Cord Injury
Original Editor - Ewa Jaraczewska based on course by Melanie Harding
Top Contributors - Ewa Jaraczewska, Lucinda hampton, Jess Bell, Kim Jackson and Tarina van der Stockt
Introduction[edit | edit source]
Spinal cord injury (SCI) results in the loss of a muscle's ability to generate power, sensation, and the ability to control the bowel and bladder. Rehabilitation efforts concentrate on preventing secondary complications associated with mobility loss. State-of-the-art technology provides mechanical assistance for patients to relearn walking or moving their arms.
History of Spinal Cord Injury Rehabilitation[edit | edit source]
Spinal cord injury treatment was first developed by Theodor Kocher (Switzerland) and William Wagner (Germany), but references to spinal cord injuries go back to classical times.[1]
- Edwin Smith's surgical papyrus in Egypt from around 3000 BC describes paraplegia following injury to the spine.
- Hippocrates (circa 460–370 BC) describes traction in the treatment of spinal cord injury.
- Paul of Aegina (AD 625–690) uses a windlass for spine dislocation and suggests laminectomy.
- Avicenna (circa 980-1037) defines paralysis following a spine fracture as a fatal injury.
- Roland of Parma (circa 1230) from Salerno used manual extension in the treatment of spinal cord injury. He was a pioneer of early intervention.
- Ambroise Paré (1564–1598) proposed laminectomy for the treatment of spinal cord injury.
- Astley Cooper (1768–1841) and Charles Bell (1774–1842) wanted to introduce spinal cord injury treatment in London teaching hospitals. Bell focused on accurate diagnostic procedures. According to him, the surgery was dangerous. He was teaching that renal failure was the cause of death following a spinal cord injury.
- Henry Cline (1750–1827) first performed laminectomy for the treatment of spinal cord injury.
- Wilhelm Wagner (1848–1900) in Germany developed the first treatment protocol for spinal cord injuries. Together with Paul Stolper (1865–1906), he co-authored a book on spinal cord injury which included the anatomy, pathology, mechanism of injury and practical treatment.
- Theodor Kocher (1841–1917) from Switzerland is known for his research related to the anatomy and physiology of spinal cord injury.
- First World War: helped to develop the concept of the modern management of spinal injuries due to the high number of war casualties with spinal cord injury.
- Between the first and the second World War, the custodial care for patients with a spinal cord injury continued in the UK.
- Charles Frazier (1870–1936) in the US provided a statistical analysis of the outcomes of spinal cord injury surgery, prognosis, life expectancy, and discharge home and work. A page and a half in his book included information on the physical management of spinal cord injury.
- George Riddoch (1888–1947) is known for his work on rehabilitation and the pathophysiology of spinal cord injuries.
- Donald Munro (1889–1973) is the father of the treatment of paraplegia.
- Ludwig Guttmann (1899–1980) is considered a founder of the modern treatment of spinal injuries. He formed dedicated spinal cord injury units managing the spine, bladder, bowel, skin, and providing education, and rehabilitation. Conservative management progressed from traction, bed rest, and bracing to surgical management and bracing, shortening patients' time on bedrest.[2]
Aetiology[edit | edit source]
The traumatic causes of spinal cord injury include:
- Falls: from a height or simple falls
- Motor vehicle accidents (MVAs)/motor vehicle crashes
- Sports-related accidents
- Violence
- Other remaining causes of injury[3][4]
MVAs and falls are the most common causes of injury accounting for nearly equal percentages. In developed countries, the main cause of SCI used to be MVAs, but recently has become falls. In low-income countries, falls are the most common cause of SCI.[3]
Non-traumatic causes of spinal cord injury include the following:
- Degeneration
- Inflammatory or auto-immune
- Neoplasms
- Vascular
- Infection
- Tuberculosis (Sub-Saharan Africa). [2]
Patient demographics: (you can find World by Income and Region information here)
- Developed countries:
- Male to female ratio: from 1.10:1 to 6.69:1
- Mean age: from 14.6 to 67.6 years
- Low-income countries:
- Male to female ratio: from 1.00:1 to 7.59:1
- Mean age: from 29.5 to 46.0 years[3]
Level and severity of the injury
- Cervical spinal cord injury is common in both developed countries and non-developed countries
- There is a lower percentage of complete injury vs incomplete injury
- Tetraplegia is more common than paraplegia in both developed countries and non-developed countries
- Motor-complete injuries (ASIA A or B) were more common for patients with traumatic SCI, while there were more motor-incomplete injuries (ASIA C or D) for patients with non-traumatic SCI[3]
Epidemiology[edit | edit source]
Developed countries:
- Incidence: from 13.1 to 163.4 per million people
- Prevalence: from 490 to 526 per million population
- Mortality: from 3.1% to 22.2%[3]
Low-income countries:
- Incidence: from 13.0 to 220.0 per million people
- Prevalence: about 440.026 per million people
- Mortality: 1.4% to 20.0%[3]
Examples of life expectancy for a 20-year-old:
- healthy individual: 79.5 years
- individual with incomplete spinal cord injury: 72.9 years
- individual with paraplegia: 65.5 years
- individual with low tetraplegia: 60.7 years
- individual with high tetraplegia (C5 and above): 56.9 years.[2]
You can read more information on the epidemiology of spinal cord injury worldwide here.
Complications of Spinal Cord Injuries[edit | edit source]
- Respiratory complications: are the main comorbidities among individuals with cervical and high thoracic injury. Complications include pneumonia, atelectasis and other respiratory issues. With a higher level of spinal cord injury, there is an increased risk of respiratory complications.[5][6]
- Decubitus (pressure) ulcers: is defined as a localised injury to the skin and/or the underlying tissue. It usually develops over a bony prominence, as a result of pressure and/or shear forces.[7]
- Joint Contractures: lead to pain, deformity, loss of function, decreased levels of independence and lower quality of life. Patients with tetraplegia are especially exposed to shoulder and elbow joint contracture.[7]
- Myositis ossificans: is characterised by the formation of a new extraosseous (ectopic) bone in the soft tissue surrounding peripheral joints. The pathophysiology of this condition is not well defined.
- Autonomic dysreflexia: an episode of hypertension in response to unmodulated sympathetic reflexes after spinal cord injury.[8]
- Syringomyelia: a rare neurogenic disease which can damage the spinal cord due to the formation of a fluid-filled area in the form of a cyst, usually found in the high cervical spine. Predominantly presents with sensory symptoms such as pain and temperature insensitivity.[9]
- Urinary tract infections due to repeated or prolonged use of catheters, both indwelling and intermittent catheterisation. There is a high risk of introducing different types of bacteria to the urinary system which leads to urinary tract infections.[10]
- Pyelonephritis: kidney inflammation caused by a bacterial infection. It occurs when a urinary tract infection spreads from the bladder to the kidneys.[11]
- Kidney and bladder stones: there is an increased risk of bladder stone formation and morbidity from the surgeries to remove them in persons with spinal cord injury. This often occurs in the first year after SCI, but the natural history of bladder stones among this patient population is poorly defined.[12]
- Kidney failure
- Bladder carcinomas: the development of a neurogenic bladder after a spinal cord injury can lead to bladder cancer.[13]
- Urinary Incontinence or constipation: bowel dysfunction is one of the most prevalent secondary complications post-spinal cord injury and improving bowel dysfunction is considered one of the highest priorities among persons with SCI.[7]
- Nociceptive and neuropathic pain: may occur in up to 60% of patients with a spinal cord injury. Among these individuals, 58% reported nociceptive pain (musculoskeletal pain). Neuropathic pain at or below the damage level was observed in 5% to 53% of patients.[14]
- Spasticity: this is a frequent complication after spinal cord injury and it usually occurs a few weeks after the acute onset of SCI and may develop over months or years.[15]
- Postural hypotension: a sudden drop in blood pressure during position changes, eg., from lying down to a sitting position.
- Impaired temperature control: individuals with a high level of spinal cord injury demonstrate an increase in body temperature together with increased heat retention.[16]
- Depression: is relatively common after SCI, occurring in over 70% of individuals with SCI. It affects mood, ambitions, views, problem solving abilities and energy level.[17]
- Osteoporosis: rapid bone loss and elevated risk of fracture have been related to SCI. A significant increase in bone resorption and deficiency in bone anabolic activity leads to rapid bone loss following SCI, especially in the distal femur and proximal tibia. [18]
- Sexual dysfunction: the need for sexual expression and intimacy declines. This is related to the biological consequences of SCI and emotional disorders or a lack of interpersonal contact.[14]
Classification of Spinal Cord Injuries[edit | edit source]
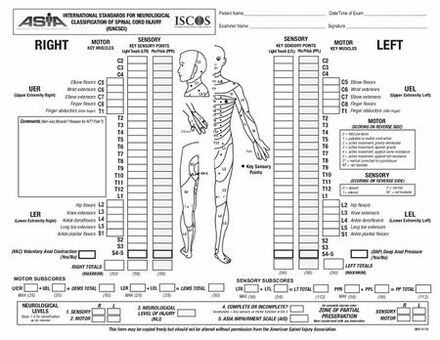
The American Spinal Injury Association (ASIA) and the International Spinal Injury Society (ISCoS) introduced a standardised classification for individuals with a spinal cord injury called the International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI).[19] This classification allows clinicians to determine, based on the neurological examination, the level and the severity of the spinal cord injury and it can be used in all phases of post-injury recovery. It includes:[19]
- Sensory testing in twenty-eight dermatomes
- Muscle functions of ten key muscles of the upper and lower extremities on both sides of the body.
Purpose of classification standardisation for spinal cord injuries:
- Standardisation for clinical documentation
- Standardisation for communication of SCI-related neurological impairments
- Defining inclusion and exclusion criteria for research purpose
- Defining outcome assessment in research studies
The final revision of ISNCSCI was released in 2019 and is included in the International Standards Training e-Program (InSTeP).[20][21]
The following are impairment scales:
- ASIA A: Complete injury where no sensory or motor function is preserved in the sacral segments S4–S5.[20]
- ASIA B: Sensory incomplete characterised by preserved sensory function, but not motor function at the most caudal sacral segments S4–S5 AND no motor function present more than three levels below the motor level on either side of the body.[20]
- ASIA C: Motor incomplete with a motor function preserved at the most caudal sacral segments on voluntary anal contraction (VAC) OR the patient meets the criteria for sensory incomplete status (sensory function preserved at the most caudal sacral segments (S4–S5), with sparing of motor function more than three levels below the motor level on either side of the body.[20]
- ASIA D: Motor incomplete as above with at least half of key muscle functions below the single neurological level of injury having a muscle grade ≥ 3.[20]
- ASIA E:Normal sensation and motor function in all segments and the patient had prior deficits, then the ASIA grade is E. Someone without an SCI does not receive an ASIA grade.[20][22]
A patient with an ASIA A at first examination within 72 hours since the injury has a 3% chance of walking. A patient with an ASIA B has a 31% chance of becoming an ASIA D at a one year follow up. Patients with an ASIA C impairment have a 67% chance of becoming an ASIA D impairment and an individual with ASIA D spinal cord injury has an 85% chance of recovery and walking in one year. [2]
Read here for a detailed discussion of the ISNCSCI assessment. You can download the ASIA form from ASIA website.
Spinal Cord Injury Syndromes[edit | edit source]
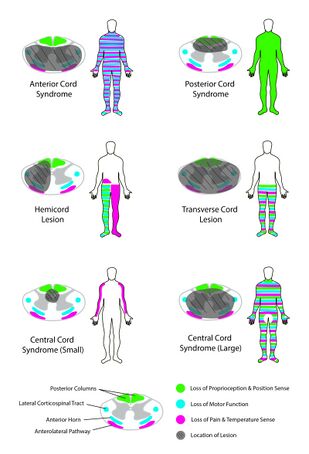
Syndromes not included in the International Standards examination which are described as incomplete spinal injury syndromes are: Anterior Cord, Posterior Cord, Brown-Séquard, Central Cord, Conus Medullaris, and Cauda Equina.
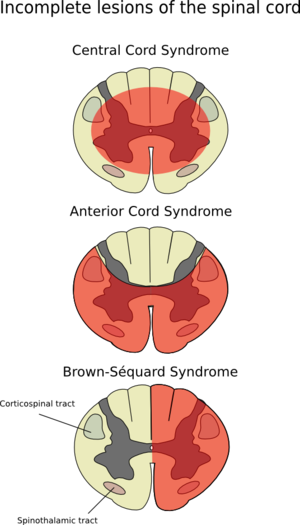
Anterior Cord: This syndrome is characterised by damage to the anterior two-thirds of the spinal cord, due to decreased or an absence of blood supply, typically as a consequence of flexion injuries.[20] The corticospinal and spinothalamic tracts are compromised leading to a loss of motor function, pain sensation, and temperature sensation at and below the injury level.[20] The light touch and joint position sense are both preserved. The individual with a diagnosis of anterior cord syndrome has a 10 - 20% chance of motor recovery.[2]
Posterior Cord: An extension injury to the spine or post-surgical haematomas can lead to the development of this syndrome. The clinical symptoms include sensory loss, mainly proprioception, and balance impairment due to a lack of proprioception. Functional outcomes include ambulation with assistive devices.[2]
Brown-Séquard: Presents clinically with ipsilateral loss of proprioception and vibration and motor control at and below the level of lesion, sensory loss at the level of the lesion, and contralateral loss of pain and temperature sensation. This is due to injury to one side of the spinal cord. Brown Séquard syndrome is rarely present alone and often includes some characteristics of the central cord syndrome, sometimes referred to as Brown Séquard-plus syndrome.[20]
Central Cord: This syndrome is a consequence of a hyperextension injury (eg. from a fall) when a person has an underlying history of cervical spondylosis.[20] Spine fracture or dislocation does not need to occur at the same time. The clinical presentation includes an incomplete injury with greater weakness in the upper limbs compared to the lower limbs. Prognosis is good, but it may depend on the person's age with better recovery in younger patients.[2]
Conus Medullaris: The leading cause of this syndrome is an injury to the sacral cord, the conus medullaris, and lumbar roots.[2] Clinically there is loss of bladder and bowel and lower limb function, but the symptoms may vary depending on the level of the lesion. In some cases, it can be wrongly diagnosed as a cauda equina injury.[20]
Cauda Equina: Due to injury of the lower motor neurone, this syndrome is characterised by flaccid paralysis of the muscles of the lower limbs, and areflexic bowel and bladder. There may be a partial or complete loss of sensation and muscles atrophy.[20]
Prognosis and Outcomes[edit | edit source]
When discussing the prognosis and outcomes of a spinal cord injury, one must recognise the limitations of the neurological examination as well as the diversity of spinal cord injuries. The examination results may vary depending on the patient's willingness and ability to participate, the physiotherapist's experience and training in test administration and results from the interpretation and complexity of the sacral examination.[23] The review of the literature indicates that examinations performed within the 24 hours to one week post-injury period are the most reliable and predictive of neurological outcomes.[23] Other factors affecting the prognosis of a spinal cord injury include: injury level, severity, aetiology, the timing of intervention, disease-modifying complications and patient-specific genetic factors.[24]
Other factors impacting neurological and functional recovery post spinal cord injury include:
- Age:
- Younger age at the time of injury is a good positive predictor.[23]
- Patients over 50 years of age have a decreased likelihood of recovering walking ability one-year post-injury, relative to patients under age 50.[23]
- People with injury at age ≥65 had less motor score change and functional recovery relative to younger individuals at discharge from rehabilitation.[23]
- In central cord injuries, patients over age 50 show less recovery compared to younger patients. 41% of older patients versus 97% of younger individuals have a chance of ambulation, 29% versus 83% will have bladder recovery, 24% versus 63% have a chance of bowel function returning, and 24% versus 77% can regain independence in dressing.[2]
- Type of injury:
- Penetrating (violent) trauma to the spinal cord will more likely lead to complete spinal cord injury. There will less likely be AIS improvement seen at one-year follow-up.[25]
- A completed spinal cord injury due to a blunt trauma to the spinal cord demonstrates a higher rate of conversion to an incomplete injury.[26]
- Gender:
- Female patients show better neurological recovery in ASIA motor scores from an initial examination to one year. This applies to individuals with neurologically complete and incomplete injuries.[27]
- The majority of published data do not support gender having an effect on neurological recovery after spinal cord injury.[23]
Resources[edit | edit source]
- Rehabilitation Evidence: https://scireproject.com/evidence/rehabilitation-evidence/
- American Spinal Injury Association. International Standards Training e-Program 2019: https://asia-spinalinjury.org/learning/
- Facing Disability. Spinal Cord Injury Resources:https://facingdisability.com/resources
References[edit | edit source]
- ↑ Silver JR. History of the treatment of spinal injuries. Postgraduate medical journal. 2005 Feb 1;81(952):108-14.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 Harding M. Introduction to Spinal Cord Injuries. Physiopedia Course 2022.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Kang Y, Ding H, Zhou HX, Wei ZJ, Liu L, Pan DY, Feng SQ. Epidemiology of worldwide spinal cord injury: a literature review. Journal of Neurorestoratology. 2018;6:1-9
- ↑ Kumar R, Lim J, Mekary RA, Rattani A, Dewan MC, Sharif SY, Osorio-Fonseca E, Park KB. Traumatic Spinal Injury: Global Epidemiology and Worldwide Volume. World Neurosurg. 2018 May;113:e345-e363.
- ↑ Raab A. Respiratory complications in spinal cord injury and the potential for reduction. Doctoral dissertation, sn: sl.2021
- ↑ Madasa V, Boggenpoel B, Phillips J, Joseph C. Mortality and secondary complications four years after traumatic spinal cord injury in Cape Town, South Africa. Spinal Cord Ser Cases, 2020;6(84).
- ↑ 7.0 7.1 7.2 Perrouin-Verbe B, Lefevre C, Kieny P, Gross R, Reiss B, Le Fort M. Spinal cord injury: A multisystem physiological impairment/dysfunction. Revue Neurologique. 2021 May 1;177(5):594-605.
- ↑ Eldahan KC, Rabchevsky AG. Autonomic dysreflexia after spinal cord injury: systemic pathophysiology and methods of management. Autonomic Neuroscience. 2018 Jan 1;209:59-70.
- ↑ Shenoy VS, Sampath R. Syringomyelia. In: StatPearls. StatPearls Publishing, Treasure Island (FL); 2021.
- ↑ Moshi HI, Sundelin GG, Sahlen KG, Sörlin AV. A one-year prospective study on the occurrence of traumatic spinal cord injury and clinical complications during hospitalisation in North-East Tanzania. African health sciences. 2021 Aug 2;21(2):788-94.
- ↑ Nikolaidis P, Dogra VS, Goldfarb S, Gore JL, Harvin HJ, Heilbrun ME, Heller MT, Khatri G, Purysko AS, Savage SJ, Smith AD. ACR appropriateness criteria® acute pyelonephritis. Journal of the American College of Radiology. 2018 Nov 1;15(11):S232-9.
- ↑ Kasabwala K, Borofsky M, Stoffel JT, Welk B, Myers JB, Lenherr SM, Elliott SP. MP54-16 ASSOCIATION OF URINARY STONES WITH PATIENT-REPORTED COMPLICATIONS IN SPINAL CORD INJURY. The Journal of Urology. 2021 Sep;206(Supplement 3):e955-6.
- ↑ Kreydin E, Welk B, Chung D, Clemens Q, Yang C, Danforth T, Gousse A, Kielb S, Kraus S, Mangera A, Reid S. Surveillance and management of urologic complications after spinal cord injury. World journal of urology. 2018 Oct;36(10):1545-53.
- ↑ 14.0 14.1 Tarnacka B, Korczyński B, Frasuńska J. Long-term complications following spinal cord injury and ageing. Advances in Psychiatry and Neurology/Postępy Psychiatrii i Neurologii, 2021;29(4):234-45.
- ↑ Sun WM, Ma CL, Xu J, He JP. Reduction in post-spinal cord injury spasticity by a combination of peripheral nerve grafting and acidic fibroblast growth factor infusion in monkeys. Journal of International Medical Research. 2021 Jun;49(6):[[1]].
- ↑ Price MJ, Trbovich M. Thermoregulation following spinal cord injury. Handbook of Clinical Neurology 2018 Jan 1 (Vol. 157, pp. 799-820). Elsevier.
- ↑ Bell TM, Vetor AN, Zarzaur BL. Prevalence and treatment of depression and posttraumatic stress disorder among trauma patients with non-neurological injuries. J Trauma Acute Care Surg 2018; 85: [[2]].
- ↑ Liu D, Wang B, Qiu M, Huang Y. MiR‐19b‐3p accelerates bone loss after spinal cord injury by suppressing osteogenesis via regulating PTEN/Akt/mTOR signalling. Journal of Cellular and Molecular Medicine. 2021 Jan;25(2):[[3]].
- ↑ 19.0 19.1 Rupp R, Schuld C, Biering-Sørensen F, Walden K, Rodriguez G, Kirshblum S. A taxonomy for consistent handling of conditions not related to the spinal cord injury (SCI) in the International Standards for Neurological Classification of SCI (ISNCSCI). Spinal cord. 2022 Jan;60(1):18-29.
- ↑ 20.00 20.01 20.02 20.03 20.04 20.05 20.06 20.07 20.08 20.09 20.10 20.11 Rupp R, Biering-Sørensen F, Burns SP, Graves DE, Guest J, Jones L, Read MS, Rodriguez GM, Schuld C, Tansey-Md KE, Walden K, Kirshblum S. International Standards for Neurological Classification of Spinal Cord Injury: Revised 2019. Top Spinal Cord Inj Rehabil. 2021 Spring;27(2):1-22.
- ↑ International Standards for Neurological Classification of SCI (ISNCSCI) Worksheet. Available at:https://asia-spinalinjury.org/international-standards-neurological-classification-sci-isncsci-worksheet/ (last accessed 27.03.2022).
- ↑ Kirshblum S, Schmidt Read M, Rupp R. Classification challenges of the 2019 revised International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI). Spinal Cord. 2022 Jan;60(1):11-17.
- ↑ 23.0 23.1 23.2 23.3 23.4 23.5 Kirshblum S, Snider B, Eren F, Guest J. Characterizing natural recovery after traumatic spinal cord injury. Journal of neurotrauma. 2021 May 1;38(9):1267-84.
- ↑ Jaja BNR, Jiang F, Badhiwala JH, Schär R, Kurpad S, Grossman RG, Harrop JS, Guest JD, Toups EG, Shaffrey CI, Aarabi B, Boakye M, Fehlings MG, Wilson JR. Association of Pneumonia, Wound Infection, and Sepsis with Clinical Outcomes after Acute Traumatic Spinal Cord Injury. J Neurotrauma. 2019 Nov 1;36(21):[[4]].
- ↑ Khorasanizadeh M, Yousefifard M, Eskian M, Lu Y, Chalangari M, Harrop JS, Jazayeri SB, Seyedpour S, Khodaei B, Hosseini M, Rahimi-Movaghar V. Neurological recovery following traumatic spinal cord injury: a systematic review and meta-analysis. J Neurosurg Spine. 2019 Feb 15:1-17.
- ↑ Roach MJ, Chen Y, Kelly ML. Comparing Blunt and Penetrating Trauma in Spinal Cord Injury: Analysis of Long-Term Functional and Neurological Outcomes. Top Spinal Cord Inj Rehabil. 2018 Spring;24(2):121-132.
- ↑ Sipski ML, Jackson AB, Gómez-Marín O, Estores I, Stein A. Effects of gender on neurologic and functional recovery after spinal cord injury. Arch Phys Med Rehabil. 2004 Nov;85(11):1826-36