Telomere
Original Editor - Lucinda hampton
Top Contributors - Lucinda hampton, Vidya Acharya and Tolulope Adeniji
Introduction[edit | edit source]
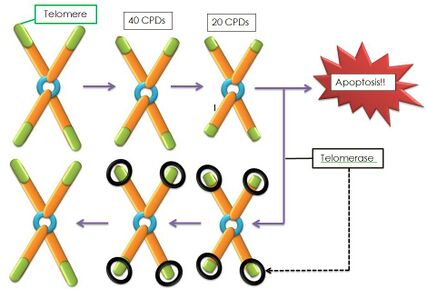
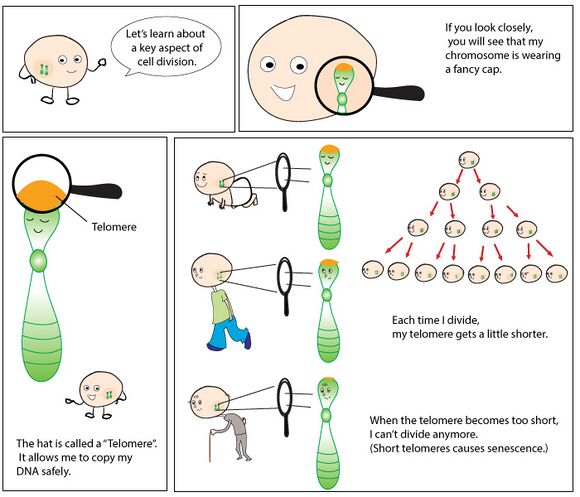
Telomeres are repetitive deoxyribonucleic acid (DNA) sequences which cap the ends of chromosomes, protecting them from degradation (note: unlike other parts of DNA telomeres do not contain information needed to make proteins). With every cell division, the telomere shortens and when a critical telomere length is attained, cellular replication can no longer take place, and it becomes “senescent”. To counter this shortening, telomerase plays a critical role. Telomerase is an enzyme that repairs the telomeres of the chromosomes so that they do not become progressively shorter during successive rounds of chromosome replication[1]. Telomeres also allow cells to distinguish between natural chromosomal termini and double-strand breaks in the DNA.[2] The telomeres themselves in turn are protected by protein complexes called “shelterins”. These shelterins role is to regulate telomerase activity on the telomere and prevent telomerase overactivity to avoid excessive telomere elongation[1].
Does this process mean anything regarding our mortality? Does the fate of individual cells mean that much with telomere shortening counting down the remaining time our bodies have to live? These questions remain to be answered.[3]
Lifestyle and Telomeres[edit | edit source]
Most studies indicate an important role of lifestyle habits on the degree of biological aging, with diet, stress and exercise all playing a part.
- A healthy diet rich in of dietary fiber and unsaturated lipids exerts a protective role on telomere health, whereas high consumption of sugar and saturated lipids accelerates telomere attrition e.g. Mediterranean diet versus a modern western diet. Those effects are likely to be related to oxidative stress and inflammation, as antioxidant and anti-inflammatory properties of nutrients are associated with longer telomeres.
- Physical activity may protect telomeres however no consensus has been reached on the optimal exercise dose.[4] The beneficial effects of physical activity on telomeres could be driven by an increase in telomerase activity following an acute bout of exercise, potentially alleviating oxidative stressors and positively effecting skeletal muscle[5]. Lifestyle (regular) physical activity is believed to positively affect the telomeres’ length prolonging the human lifespan. A systematic review suggests that exercises positively influence the telomere length compared to inactivity and aerobic exercise of moderate intensity over six months is the most effective in slowing the rate of telomere shortening[6].
- Shorter telomere length is associated with older age and increased disease morbidity and mortality. Emerging studies suggest that stress accelerates the erosion of telomeres from very early in life and possibly even influences the initial (newborn) setting of telomere length[7].
- Smoking and excess weight speed up telomere shortening as well as the aging process.[8]
Chronic Disease[edit | edit source]
Shorter telomeres are not only associated with age and disease. Shorter telomere length and low telomerase activity are linked to several chronic preventable diseases. These include hypertension, cardiovascular disease, insulin resistance, type 2 diabetes, depression, osteoporosis, and obesity[9].
Telomere shortening and the absence of telomerase in normal tissues is a tumour-suppression mechanism. By contrast, tumours aberrantly upregulate telomerase, which elongates short telomeres and allows continuous growth. Telomere maintenance is critical for stem cell function and normal tissue homeostasis but is also hijacked in cancers as a means to cellular immortality.[10]
Physiotherapy[edit | edit source]
The importance of exercise as a medicine is receiving more and more attention. This has many implication in the health world and in particular the physiotherapy world. Education regarding lifestyle and health is a critical part of lifelong health. For more see Physical Activity and Physical Activity as an Anti-Aging Medicine.
References[edit | edit source]
- ↑ 1.0 1.1 Haupt S, Niedrist T, Sourij H, Schwarzinger S, Moser O. The Impact of Exercise on Telomere Length, DNA Methylation and Metabolic Footprints. Cells 2022, 11, 153. Available:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8750279/ (accessed 23.7.2022)
- ↑ Frontiers Research topics Available:https://www.frontiersin.org/research-topics/781/telomeres-structure-functions-and-therapeutic-opportunities (accessed 24.7.2022)
- ↑ The conversation Tick, tock… how stress speeds up your chromosomes’ ageing clock Available:https://theconversation.com/tick-tock-how-stress-speeds-up-your-chromosomes-ageing-clock-127728 (accessed 23.8.2022)
- ↑ Sánchez-González JL, Sánchez-Rodríguez JL, Juárez-Vela R, Ruiz de Viñaspre-Hernandez R, González-Sarmiento R, Martin-Vallejo FJ. Analysis of Telomere Length and Its Implication in Neurocognitive Functions in Elderly Women. Journal of Clinical Medicine. 2022 Mar 21;11(6):1728.
- ↑ Balan E, Decottignies A, Deldicque L. Physical activity and nutrition: two promising strategies for telomere maintenance?. Nutrients. 2018 Dec 7;10(12):1942. Available:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6316700/ (accessed 24.7.22)
- ↑ Song S, Lee E, Kim H. Does Exercise Affect Telomere Length? A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Medicina. 2022 Feb 5;58(2):242.
- ↑ Shalev I, Entringer S, Wadhwa PD, Wolkowitz OM, Puterman E, Lin J, Epel ES. Stress and telomere biology: a lifespan perspective. Psychoneuroendocrinology. 2013 Sep 1;38(9):1835-42.Available:https://pubmed.ncbi.nlm.nih.gov/23639252/ (accessed 23.7.2022)
- ↑ Long long life Telomere Shortening: Causes, and Consequences on Aging Available: http://www.longlonglife.org/en/transhumanism-longevity/aging/telomere-shortening-causes-consequences-on-aging/(accessed 24.7.2022)
- ↑ Very well health The Telomere Theory of Aging Available:https://www.verywellhealth.com/telomere-shortening-the-secret-to-aging-2224346 (accessed 24.7.2022)
- ↑ Hiyama E, Hiyama K. Telomere and telomerase in stem cells. British journal of cancer. 2007 Apr;96(7):1020-4. Available:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2360127/ (accessed 24.7.2022)