Subcortical Vascular Dementia: Case Study
Top Contributors - Bomi Jang, Jonathan Tam, Kiley Praught, Emily Mulligan, Sofia Lamarche, Uchechukwu Chukwuemeka, Lucinda hampton, Harrison Mah and Koon Kei Gary Lai
This fictional case study is produced by first-year physiotherapy students at Queens’ University for education purposes.
Abstract[edit | edit source]
The purpose of this case study page is to describe a fictional case of an older adult with a new diagnosis of subcortical vascular dementia with symptoms affecting her daily function and quality of life. The authors have described a possible presentation for this patient and subsequent treatment plan. Interventions suggested for this patient include progressive balance, functional, and gait training to work towards goals that the patient has identified. After progressing through the training program, the patient will be transitioned to community exercise and physical activity (such as tai chi) to maintain and mitigate the effects of the disease. Technology will also be used in her recovery in the form of Serious Games. Outcomes include patient reports as well as formal measurements such as MMTs, ROM, Berg Balance Scale, gait speed, MiniBESTest, TUG with and without dual task, ABC scale, MoCA, Mini-Cog, and Romberg (performed by PT and other HCPs). Outside of physiotherapy, referrals that would be beneficial were highlighted including to a neurologist, occupational therapist, speech-language pathologist, social worker/ therapist, as well as community care and the Alzheimer’s Society. Although this patient is experiencing a progressive disease, it is important to optimize her function to allow her to continue in activity and participation in her life, as outlined in this case.
Abbreviations[edit | edit source]
- ABC Scale = Activities-specific Balance Confidence Scale
- ADL = activities of daily living
- AROM = active range of motion
- BADL = basic activities of daily living
- BBS = Berg Balance Scale
- BMI = body mass index
- CBMS = Community Balance and Mobility Scale
- C/S = cervical spine
- FAB = Frontal Assessment Battery Test
- GP = general practitioner
- HCP = healthcare provider
- HEP = home exercise program
- HR = heart rate
- IADL = instrumental activities of daily living
- ICF = International Classification of Functioning, Disability and Health
- L/E = lower extremity
- LHIN = Local Health Integration Networks
- MCID = minimal clinically important difference
- MMT = manual muscle test
- MoCA = Montreal Cognitive Assessment
- MVC = motor vehicle collision
- OT = occupational therapist
- PCP = Primary Care Practitioner
- PPA = Primary Progressive Aphasia
- PSW = personal support worker
- PT = physiotherapist/physical therapy
- ROM = range of motion
- RR = respiratory rate
- SLP = speech language pathologist
- SLT = speech and language therapy
- TIA = transient ischemic attack
- TV = television
- TUG = Timed Up and Go
- U/E = upper extremity
- WNL = within normal limits
- y/o = years old
Introduction[edit | edit source]
Dementia is a broad term characterizing a spectrum of symptoms that impacts one’s brain function[1]. The condition presents through a progressive and incurable neurodegenerative disease that can impact an individual’s physical, mental, and emotional health[1]. Worldwide, an estimated 55 million people are living with dementia with this population more heavily favouring low and middle income countries[1]. The projected increase in this number demands acknowledgement and accessibility of treatment across interdisciplinary health care professions. A challenging aspect of health care intervention on dementia is the relative inability to reverse disease progression and restore normal function[1]. Due to this limitation, intervention should be focused on slowing progressive losses and optimizing function in each stage of the disease to maintain the best quality of life.
The role of physiotherapy related to dementia treatment can encompass several interventions targeting things such as; pain relief, physical deconditioning, and balance impairments[2]. Across current literature, it is suggested that some of the most effective physiotherapy interventions for individuals in early stage dementia include exercises to maintain mobility, developing fall prevention programs, and learning adaptations to maximize independence of the patient in their daily activities[2]. Furthermore, related literature suggests that incorporating physical activity, of even mild to moderate intensity a few times a week, into treatment interventions can help combat dementia’s cognitive deficits[3].
Vascular Dementia is a common type of dementia described by a blockage of blood supply to the brain resulting in the brain cells to be deprived of oxygen and nutrients, leading to death[1]. Vascular Dementia is further divided into two subtypes: Subcortical Dementia and Cerebral Amyloid Angiopathy[1]. Subcortical Vascular Dementia (also known as Binswanger’s Disease), as seen in the following case study, is caused by a disease of the small vessels in the brain resulting in reduced blood flow[1].
The current case study illustrates a patient recently diagnosed with stage 3 subcortical vascular dementia, who presents at an outpatient neurological rehabilitation centre following a fall. The patient suffers several cognitive deficits related to concentration, communication, and organization. The patient also demonstrates physical deficits manifested in psychomotor slowness, poor balance, weakness, and overall slow motor function, impacting their ability to complete daily activities. The purpose of the following case study serves as a documentation of the effects of neurological physical therapy treatment interventions during rehabilitation on the patient’s experience and outcomes and helps the learner walk through a possible case scenario of Subcortical Vascular Dementia.
Client Characteristics[edit | edit source]
- Pseudonym: B.B.
- Age: 60 y/o
- Gender: Female
- Ethnicity: Caucasian
- Marital status: Married (husband)
- Occupation: Retired secretary for the government
BB’s physician called to inquire if a physiotherapist at the outpatient neurological rehabilitation clinic could accept B.B.’s case. B.B. presented to her primary care practitioner (PCP) after a minor fall 2 weeks ago. After concerns noted from PCP, the PCP and neurologist have done a workup and have diagnosed the patient with stage 3 (mild decline) vascular dementia, specifically a subcortical presentation, also known as Binswanger’s Disease. Focus of physiotherapy will be to improve her baseline function as much as possible and then maintain function throughout the course of her progressive disease[4].
Primary condition: Vascular dementia subtype subcortical dementia, Stage 3 (mild decline)
Nature of the condition: Mild decline, Stage 3
- Physical manifestations including psychomotor slowness and weakness, as well as decreased balance, and coordination
- Cognitive manifestations: confusion, difficulty paying attention or concentrating, trouble organizing thoughts, trouble making plans and communicating them, slowed thinking, memory loss and speech difficulties
Primary reason the patient was referred: Recent diagnosis with vascular dementia and concerns with subsequent poor functional status from PCP and neurologist.
Patient herself has noticed having difficulty generating speech at times, finds it easier for herself to give simpler answer as it is less frustrating and effortful, patient herself has noticed progressive balance issues/feeling unsteady, reduced activity due to fear of falling, has noticed reduced ability to be independent in BADLs and IADLs, also concerns from husband[4].
Relevant comorbidities: Hypertension (well-managed with medications by PCP)[4], high BMI (due to sedentary lifestyle), no previous major traumas/MVC/falls (minor fall 2 weeks ago; no major injuries as per PCP), past abdominal surgery (C-section) 35 years ago
Examination Findings[edit | edit source]
(Note: assessed over multiple sessions)
Subjective[edit | edit source]
History of Present Illness[4]
- Patient has had a recent new diagnosis with vascular dementia from her physician and neurologist specialist.
- The patient had a minor fall at home, which led to her going to the doctor’s office. Although the fall was minor, her physician then had some concerns over balance, gait, etc.
- The patient and her physician has noticed progressive balance issues/feeling unsteady, reduced activity due to fear of falling.
- Patient has noticed having difficulty generating speech at times, finds it easier to give simpler answer as it is less frustrating and effortful.
Diagnostic Tests
Results of tests from neurologist
- Neurologist performed the Frontal Assessment Battery (FAB) test to differentiate between Alzeihmer’s disease and Vascular dementia[5].
- CT scan of the brain shows the brain lesions typical of the disease including widespread, microscopic areas of damage to the brain resulting from the thickening and narrowing (atherosclerosis) of arteries that supply blood to the subcortical areas of the brain[6].
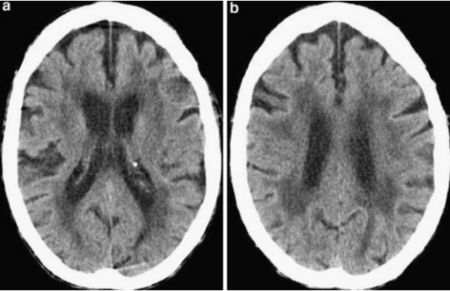
- Past Medical History:
- Abdominal surgery (C-section) 35 years ago,
- Previous transient ischemic attack
- No other major trauma, MVCs, falls, or surgeries
- Minor fall 2 weeks prior
- Controlled hypertension
- Medication
- Prinivil 10 mg once a day for high blood pressure[8]
- Daily women’s multivitamin (One a Day Women’s 50+)
- Health Habits
- Sedentary lifestyle; activity level has decreased over past 5 years
- Non-smoker; has never smoked in the past
- Occasionally consumes alcohol in social settings; takes on average 2 drinks/ week
- No other substance use
- Social History
- Main support system is husband (65 y/o, in good health) (married for 40 years)
- Has some friends close by when in need of support (more so emotional support)
- One adult son who lives two hours away
- Two young grandchildren (3 and 5 years old), enjoys spending time with them and babysitting, but has had decreased ability to do so recently
- Enjoys reading, knitting, and watching TV in her down time
- Current functional status[4]
- Independent in BADLs
- Currently experiencing difficulty with IADLs, notably including anything that requires more demands on coordination and balance (cooking, cleaning, etc.)
- More comfortable ambulating around the house
- Finds community ambulation difficult (eg. grocery shopping, running errands, etc.) due to being unsteady
- Finds transfers in and out of her bed difficult because of soft mattress, relies on husband for assistance
- Transfers in and out of chairs inconsistent
- Experiences some shortness of breath and feels tired/ weak after 2 minutes of walking
- Patient has noticed having difficulty coming up with words at times; finds it easier for herself to give simpler answer as it is less frustrating and effortful
- Lives in Bungalow with husband, two stairs to get into the house with a side railing on both sides
- Functional history
- Previously independent in all BADLs and IADLs
- No difficulty to ambulate in community
- No use of gait aid
- An hour long walk every weekend with her husband
- Family history
- Father died of ischemic stroke at age of 70[4]
- Mother died of natural causes at age of 90
- No siblings
- Precautions:
- Confusion at times
- Falls risk - guard closely
Objective[edit | edit source]
- Observation[4]:
- Forward head posture, postural increased kyphosis in thoracic spine
- Patient appears alert and oriented, occasional moments of confusion (husband compensating)
- Observed patient walking into the room on first visit, seemed to be a little unsteady, no use of gait aid
- Slow exaggerated movements when watching patient come into exam room
- Vital signs:
- BP: 115/80 mmHg (controlled with medication as per above), HR: 87 bpm, RR: 17 bpm
- Range of Motion (ROM)
- Impaired ROM
- slightly limited cervical extension ¾ ROM
- limited trunk extension ½ ROM
- All other ROM are within normal limits
- Impaired ROM
- Strength (MMT)
- Reduced strength bilaterally, notably in L/E
| Muscle Group | Right | Left |
| Shoulder Flexion | 4- | 4- |
| Shoulder Extension | 4 | 4 |
| Elbow Flexion | 4- | 4 |
| Elbow Extension | 4 | 4+ |
| Hip Flexion | 3+ | 3+ |
| Hip Extension | 3+ | 3 |
| Knee Flexion | 3 | 3 |
| Knee Extension | 3+ | 3 |
| Ankle Dorsiflexion | 3 | 3 |
| Ankle Plantarflexion | 3+ | 3+ |
- Reflexes:
- Hyperactive muscle stretch reflexes throughout
- Exaggerated deep tendon reflexes (clonus)
- Hoffmann's sign bilaterally
- Extensor plantar response on Babinski bilaterally
- Skin sensation: intact U/E and L/E sensation to light touch, pinprick, vibration, and temperature
- Myotomes/dermatomes: normal in U/E and L/E
- Romberg test:
- Eyes open: 23 seconds (able to stand feet together with significant sway)
- Eyes closed: 5 seconds (needed to took a step to recovery her balance)[9]
- Coordination
- Finger to nose test: mild dysmetria observed bilaterally, clumsy with few impaired pursuits, increased length of time taken to accomplish task, unable to perform with a moving target[10]
- Mild dysmetria observed when performing heel-to-shin test bilaterally
- Combined Cortical Sensations
- Stereognosis, barognosis, and graphesthesia were unremarkable.
- Double simultaneous stimulation: There was no extinction to double simultaneous stimulation.
- Kinesthesia/ proprioception
- Limb matching test: Mild loss of accuracy in L/E bilaterally
- 6 minutes walk test: 136 m
- 1st break for 56 seconds at 80m mark
- 2nd break for 123 seconds at 120m mark
- 10m walk test
- Gait speed over the test was measured to be 0.73m/s
- 3D motion analysis of gait showed: [4][11][12][13]
- Gait was noticeably slow
- Wide base
- Reduced step length
- Normal arm swing
- Decrease in stride length
- Increase in support phase, reduced swing time
- An increase in stride-to-stride variability
- Inconsistency between sides
- Poor weight transfer
- Features over course of gait cycle
- Initial stance: Minor decrease in ankle dorsiflexion and knee flexion
- Mid-stance: Minor decrease in knee extension, hip extension, and ankle dorsiflexion
- Pre-swing: Minor decrease in knee flexion and ankle plantarflexion
- Early and mid-swing: minor decrease in knee flexion
- Late swing: minor decrease in knee extension and ankle dorsiflexion
- Findings are consistent with MMT of lower extremities
- Inability to perform tandem walk
- Berg balance Scale (BBS): 36/56
- MiniBESTest
- Timed Up and Go (TUG): 17.5 seconds
- Total score: 20/28[14].
- Activities-Specific Balance Confidence Scales (ABC): 43%
- Cognitive Tests
- Timed Up and Go (TUG) Dual-Task testing:
- Cognitive challenge: counting backwards in sets of 7 from 100: 28.1 seconds[15].
- Physical challenge: carrying a cup with half full water: 32.5 seconds
- Mini-Cog Test (Short memory test with a simple clock drawing test): 1
- On-site occupational therapist performed a MoCA: 25/30 (rapid screening instrument for mild cognitive dysfunction)[6].
- Timed Up and Go (TUG) Dual-Task testing:
Clinical Impression[edit | edit source]
B.B. is a previously independently functioning 60 year old female presenting to outpatient neurological rehabilitation following a minor fall 2 weeks prior. B.B. presents with several marked cognitive and physical impairments that hinder her usual activity level. At time of referral to outpatient rehabilitation, B.B. presents with various physical deficits such as diffuse bilateral lower extremity weakness, impaired coordination and imbalance. Noted cognitive deficits include; attention impairment, noted inability to organize thoughts and communication difficulties. However, psychomotor slowness appears to be the most pronounced impairment relating to both physical and cognitive functioning. The interaction of these deficits are reflected in B.B.’s slow and unsteady motor functions, and seemingly unorganized and limited communication abilities. Previous transient ischemic attack, subjective and objective assessment findings and hallmark characteristic of psychomotor slowness correlate with the diagnosis of stage three subcortical vascular dementia otherwise known as Binswanger Disease. The diagnosis of this condition has impacted B.B.’s independence. B.B. has difficulty completing her IADLs due to imbalance and perceived fear of falling and she is frustrated due to inability to easily organize and communicate thoughts. B.B. is hoping to gain strategies to combat these deficits in order to be able to feel comfortable babysitting her grandchildren again, as she has felt isolated from them in the process of her diagnosis. Previous independent function, limited medical history, age, available support from her husband and stage of diagnosis are factors indicating that B.B. is a suitable candidate for neurological rehabilitation in an outpatient setting. Interventions focused on improving balance, fall prevention, and strengthening could support achieving optimal function for the current stage of diagnosis of this progressive neurodegenerative disease.
Problem list:
- Progressive balance deficits leading to occasional falls decreasing confidence and ability to participate in regular activities.
- Decreased strength in the right and left L/Es with weakness contributing to unsteady gait and standing balance.
- Incoordination and unsteadiness in standing and gait.
- Marked psychomotor slowness in both mental and physical activities.
- Decreased ability to communicate due to Primary Progressive Aphasia.
- At risk of developing further complications and intensifying the impact of the progressive disease through non-optimal lifestyle habits such as drinking, smoking, limited activity and previously diagnosed high blood pressure.
- Memory impairments reported through subjective history as forgetfulness and objective assessment as a positive result in a mini-cog test.
- Social system concerns and subjective feelings of unpredictable fluctuations in mood, behaviour and personality.
- Decreased ability to perform BADLs and especially IADLs independently.
- Family and support system has a lack of knowledge on how to care for people living with dementia.
Intervention[edit | edit source]
Patient-centered treatment goals
These goals were developed in consultation with the circle of care, including the patient and caregiver as well as the ICF model.
Short Term[edit | edit source]
- Body structure and function: Increase LE strength, evidenced by MMTs to 4/5 bilaterally in 4 weeks by daily strengthening exercises (HEP).
- Body structure and function: Improve balance evidenced by a berg balance score to 50/56 in 4 weeks by taking part in a falls prevention/ balance program.
- Activity: Patient will be able to walk 200metres with a gait aid within 6 minutes in 2 weeks
Long Term Goals[edit | edit source]
- Participation: Increase community participation by signing up for weekly tai chi class in 8 weeks.
- Participation: To be able to independently perform groceries shopping in 8 weeks
- Activity: Patient will be able to walk 400metres (in a closed environment) without a gait aid within 6 minutes in 6 weeks
- Body structure and function: Patient will maintain ROM within normal limits in 12 weeks
B.B's. Treatment Plan[edit | edit source]
Physical therapy (PT) is indicated in this case. PT has shown to improve or slow loss in mobility, strength, balance, and endurance[16] [17][18]. These improvements correlated to improved functional independence in mobility and ADLs in individuals with dementia[16] [17][18]. There are no guidelines to best practice and many studies do not state the therapeutic interventions used; therefore, this warrants the need for further research in this area and for this population. However, consistent with general best practice guidelines, we will focus on making therapy engaging, functional, and task-oriented.
Before starting therapy, we will ensure we are oriented to the challenges of progressive aphasia B.B. might be experiencing and brainstorm/consult with her on strategies we could use to better communicate with each other such as:
- Bringing her husband or another support person
- Booking for longer appointments so that conversations are not rushed
- Having a whiteboard available for her in clinic as an option
- Using a communication board with symbols as needed
- Speak slowly in simple, adult sentences and listen carefully.
In the initial stages, we will do community-based physical therapy two times per week focusing on improving functional abilities and balance. The goal of this initial stage is to improve B.B's functional abilities so she can safely participate in exercise/physical activity outside of the clinic. Intervention plan in this stage will include primarily balance and lower extremity strength training. In addition, while lower extremity strength and balance are being worked on, we will give B.B. a single point cane. Based on B.B’s Berg Balance Score and recent fall, it is advised that B.B. should use an assistive device to safely ambulate the community.
A functional training program was created in consultation with B.B. based on two meaningful tasks she identified she would like to improve upon. These tasks include: “Playing with grandchildren” and community ambulation activities such as “grocery shopping”.
- Balance training: 30 seconds x 2 repetitions, twice per day, daily (two times at clinic, the rest is performed as part of the home exercise program).
- Progression from normal stance width → narrow base of support → tandem stance → single leg stance → standing with trunk rotation
- Balance training will be done on hard surfaces (tile or wood flooring) and transition to soft surfaces (carpet or even standing on a pillow).
- Perturbation based training. Including tossing/catching a ball and the therapist delivered external perturbations.
- This will only be done in a clinic for safety reasons.
- If B.B. is struggling, we may add mirror therapy to help her reflect on her positioning
- Functional training: 10 reps, 2 sets, 2 minutes rest between sets, 2-3 days per week.
- ¼ squat progressing to ½ squat (be done at a counter or table for balance support if needed)
- Sit to stand
- Knee to counters
- Step ups (beginning with the support of a railing and progressing to without)
- Lifting objects from the ground (progressing up to 10lbs)
- Gait training
- Interim education and training with a single point cane with a 2 point step through gait pattern until balance is improved such that the patient is no longer a falls risk.
- Gait training focused on functional community ambulation. We will focus on increasing gait speed, managing varying terrain, curbs and obstacles and walking in a distracting environment.
- Treadmill training
- Walking on the treadmill with the use of handrail as needed. Twice per week during rehabilitation sessions. Targeting intensity at 3-5/10 rate of perceived exertion (RPE) and terminate at 6 minutes or patient requests to stop. Log sheet attached for tracking progression over the four weeks.
Ultimately, the focus of this training will be to progress B.B. to a stage where she can successfully practice and take part in the meaningful activities she identified. Throughout B.B’s functional training, specific strategies will be used to enhance her learning and promote automaticity of movement. We will promote problem solving based learning by providing minimum guidance, knowledge of result feedback and promoting self-assessment. Other strategies we will use include:
- Tactile cueing
- Maintaining an external focus of attention
- Performing random practice with a high number of repetitions
- External focus of attention
- Having B.B. do self-reflection/self-assessment about her performance
- Minimizing PT guidance
Once balance deficits are improved upon, we will drop down to one physical therapy session biweekly to focus on maintaining function and monitoring disease progression. At this point B.B. can transition to exercise in the community. We will provide B.B. with extensive education about the benefits of regular physical activity on delaying disease progression and optimizing function. We would prescribe B.B. to engage in physical activity consistent with Canada’s Exercise Guidelines for Older Adults (>65). While the Canadian Alzheimer's Society simply recommends the guidelines for adults, we would use the ones for older adults since they include the addition of weekly balance training[19]. These guidelines include:
- 150min moderate to vigorous physical activity, 2 days muscle/bone strengthening exercise per week plus weekly balance training[20].
Once B.B. has progressed past basic strength and balance training, we will also implement the use of serious games technology. Serious games refers to any game that’s primary purpose is something other than entertainment. There are many different serious games which have shown to improve physical fitness, functional mobility, strength, and balance in older adults[21] [22]. Additionally, they have also been shown to increase exercise compliance in older adults[22]. Current research has reflected the efficacy in treating balance deficits in dementia through use of serious games may have a more beneficial effect than medical treatment[3]. Additionally, since participation in serious games promotes mental and physical exercise, literature has reflected the benefits in patients living with dementia to be greater than traditional exercise alone[3]. In light of technology advances, there has become more options for serious games delivered through video games or virtual reality format that may be more engaging for patients[3]. There are various options available but finding one (or more) that B.B. enjoys is key. Some options include Wii Fit and Microsoft Kinect. Serious games using either a camera motion monitor (i.e. Microsoft Kinect) or a force sensor (i.e. Wii Fit board) also allow for biofeedback via visual, tactile (vibrations), and audio means. An additional benefit of serious games is that they allow us to monitor progress over time and identify motor deficit progression.
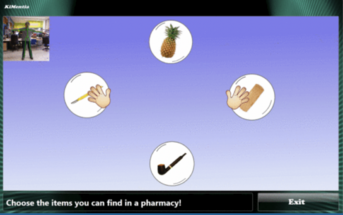
The study conducted by Urturi Breton et al.[23], showcases the use of a specific serious games intervention for patients living with dementia that presented promising results relating to patient motivation and cognitive stimulation. The intervention was called KiMentia and it promoted both mental and physical stimulation through presenting challenges to be completed on a screen while a kinetic sensor senses body movements that are required to complete the challenges[23]. Various tasks are available to suit different levels of cognitive functioning in different patients. An example task was categorizing images of objects into locations where they are likely to be found while the patient has to move the answers on the screen to the correct locations through body movements[23]. This type of intervention could be beneficial to B.B’s treatment plan as it will challenge her balance by the internal perturbations of body movements, and cognitively by the planning and organizing the objects into their correct categories.
In terms of continuing balance training in the community, we will advise B.B. to sign up for weekly tai chi class as she has expressed interest in beginning this physical activity. Tai chi has been shown to improve balance, reduce falls, and improve quality of life as well as short term cognitive function in dementia patients[24][25].
We have chosen to transition B.B. to community-based exercise to promote independence and make B.B. a more active member of their community. We have also chosen to keep regular appointments to monitor B.B. as their disease progresses and adjust our treatment plan as needed. Ongoing assessment is crucial to B.B’s. success given the progressive nature of the disease. Additional resources that we will consider as disease progress include referral to a social worker for community aids for both B.B. and their family, and speech language pathology for assistance with their aphasia (see below).
Patient education is also very important so that they understand their conditions and learn strategies that can help them live in the community. Furthermore, reminding B.B. to follow up with her GP and neurologist regarding medications for blood pressure control to prevent a subsequent stroke and possible medications for managing cognitive decline may be pertinent. Lastly, education should be extended to B.B.’s family and support system in order for them to understand disease etiology, disease progression, and the best ways they can support B.B..
Outcomes[edit | edit source]
After four weeks, objective measures were performed again to determine any improvements that were made after the various exercises and treatments were performed.
The Berg Balance Scale was re-administered giving a score of 40/56 (MCID = 4, >41 shows patient is independent[26] [27]. Her gait speed increased to 0.87 m/s (MCID=0.13 m/s, community ambulator 0.8-1.2[28][29]). Her miniBESTest score increased to 24/28 (MCID = 3.4-4, normative value for this age = 24.7[30][31] ) and the TUG test decreased to 13.4 secs (>13.5s predicts future falls, MCID = 3.4[32][33]). The dual task TUG with a cognitive challenge decreased to 27 seconds (>15 s predicts falls while doing cognitive task) while the dual task physical challenge decreased to 29.6 seconds[33]. Her 6-minute walk test increased to 300 m (MCID = 86. Healthy range = 380-782 m[34][35]). Her ABC scale score increased to 60% (less than 58.13 predictor for falls) which indicates she is in the moderate level of confidence for patients[36]. For the other cognitive tests, her mini-cog test score remained at a 1 while the MoCA score re-administered by the OT decreased to a 23/30 (>26 is normal cognition[37]). Her dysmetria has decreased about the same amount on each side as her coordination increased during the finger to nose and heel-to-shin test. Her limb-matching test has improved as well with improved ability to match the same limb position in her lower extremity to the test position. All her previous ROM measures that were normal were maintained but her cervical extension improved giving her nearly full ROM and her trunk extension increased to ¾ ROM. Romberg test has improved to 29.5 seconds with eyes open and 11 seconds with eyes closed after re-administering after four weeks (20 secs average for her age[38]).
The changes in her MMTs and other outcome measures after four weeks are listed in the charts below:
| Muscle group | Right | Left |
| Shoulder Flexion | 4+ | 4+ |
| Shoulder Extension | 4 | 4 |
| Elbow Flexion | 4+ | 4+ |
| Elbow Extension | 4 | 4+ |
| Hip Flexion | 4 | 4 |
| Hip Extension | 4 | 3+ |
| Knee Flexion | 4- | 4- |
| Knee Extension | 4 | 4- |
| Ankle Dorsiflexion | 4 | 4 |
| Ankle Plantarflexion | 4+ | 4+ |
| Measures | Initial Assessment | Re-assessment After 4-Weeks |
|---|---|---|
| AROM | Full body WNL except:
C/S extension: 3/4 of full ROM Trunk extension: 1/2 of full ROM |
Full body WNL:
C/S extension: WNL Trunk extension: 3/4 of full ROM |
| Berg Balance Scale | 36/56 | 40/56 |
| Gait Speed (based on 10-Metre Walk Test) | 0.73 m/s | 0.87 m/s |
| MiniBESTest | 20/28 | 24/28 |
| Timed Up and Go (TUG) | 17.5 seconds | 13.4 seconds |
| TUG with Dual Task | Cognitive Task: 28.1 seconds
Physical Task: 32.5 seconds |
Cognitive Task: 27.0 seconds
Physical Task: 29.6 seconds |
| 6-Minute Walk Test | 136 metres | 300 metres |
| Activities-specific Balance Confidence Scale (ABC Scale) | 43% | 60% |
| MoCA | 25/30 | 23/30 |
| Mini-Cog | 1 | 1 |
| Romberg Test (eyes open) | 23 seconds | 29.5 seconds |
| Romberg Test (eyes closed) | 5 seconds | 11 seconds |
The past four weeks of treatment have shown improvements in her strength, ROM, and her overall gait speed and endurance, which all contributed to her increased confidence and independence. She may still require some assistance in her BADLs and IADLs after discharge that her husband or son can help with. Continued collaboration and training can be maintained with the inter-professional team to ensure that strength, ROM, gait and coordination is maintained. The activity with the occupational therapist can be continued to ensure maintenance in BADLs and IADLs, as well as cognition and coordination. An option of home support with a personal support worker is again presented to B.B. when she requires it as her cognition starts to decline further due to her dementia. B.B. will still need continued treatments for her cognition as her dementia continues to progress. The speech-language pathologist she was previously referred to will continue to address her primary progressive aphasia as the severity of it increases over time. Her overall cognition will also be continually monitored by the neurologist she was previously referred to as her dementia progresses.
Discharge Planning[edit | edit source]
Due to the progressive nature of subcortical vascular dementia as well as the fact that many PT interventions are employed for maintenance, the inter-professional has decided that care will be provided indefinitely. Initially, outpatient PT appointments occur twice a week. As the patient becomes more familiar with her HEP and re-gains some independence, it was decided that the patient will schedule appointments for 3-month follow-ups only or as needed.
The inter-professional team has decided that formal discharge will only occur when the PCP or neurologist deems B.B.’s vascular dementia to be severe and/or when family members decide that they can no longer care for B.B. at home and transfer her to a long-term care facility. These two items would indicate that B.B. is no longer a suitable candidate for community outpatient PT. However, the outpatient team will refer on to the inter-professional team at the long-term facility to continue care so that B.B. can maintain as much function as possible and live with the best quality of life.
Interprofessional Interventions[edit | edit source]
Neurologist[edit | edit source]
A neurologist would play a crucial role in the long-term care of B.B. as she copes with diagnosis and progresses through the stages of vascular dementia. This field of medicine is the relationship between the brain and the behavioural or cognitive presentation of an individual, with an emphasis on neurological disorders[39]. Since the cognitive losses caused by dementia are such a hallmark feature of the disease, proper assessment and treatment interventions are critical to optimizing function[1]. A neurologist could conduct assessments to determine level of cognitive functioning while tracking the impact of disease progression on this level over time[39]. In addition to assessment, a neurologist may offer medical treatments to reduce symptoms as interventions that lie beyond the scope of physiotherapy[39]. Finally, a neurologist may also assess the efficacy of rehabilitation and medical strategies on function[39]. As reflected in this case study, the inclusion of neurology into B.B's health care offered critical importance to her correct diagnosis of Vascular Dementia through use of Frontal Assessment Battery (FAB) Test, and the ordering and interpretation of imaging[40]. A neuropsychologist could offer these assessments as well as continuous treatment interventions that target B.B's cognitive deficits relating to memory, concentration, planning and communication.
Occupational Therapist[edit | edit source]
Including an occupational therapist in B.B's treatment is important for therapeutic treatments to maintain skill or develop new strategies to complete activities of daily living. An occupational therapist would work in collaboration with B.B to determine areas in which her disease presentation is affecting her daily life and develop learning strategies to overcome or adapt to these effects while promoting independence[41]. Therapeutic interventions over the course of dementia progression can include; adapting home environment, graded assistance training in BADL/IADL performance and recommendation for assistive devices usage such as mobility aids[41]. Specific to B.B's case, at her current stage she is experiencing difficulties in performing her IADLs, especially in tasks requiring more balance. Therefore an Occupational Therapist can identify these activities of difficulties and build treatment interventions to improve independence her performance of such tasks[41].
Speech Language Pathologist[edit | edit source]
A referral to a speech language pathologist (SLP) for BB will be crucial. PPA is a very disabling disorder for which there is, at present, no available treatment. BBs presentation is consistent with early symptoms of PPA, having the most difficulty with word-finding. Speech and language therapy (SLT) has been extensively used in patients with aphasia of different etiologies and has been shown to be effective. Rather than reversing the disease, SLP for PPA aims to work within the patient’s limitations, maximize their communicative abilities, and mitigate decline. Speech language pathologists use therapeutic interventions with PPA patients including picture naming, description of picture actions, complex auditory-verbal comprehension, reading and writing, facilitation of expression of feelings and opinions, and enhancement of conversational skills. SLP will consider what is most important and currently frustrates the client[42].
Social Work/ Counselling/ Therapist[edit | edit source]
A referral to social work and/ or a community counselor/ therapist might be appropriate for BB and her husband to explore to help them navigate this new diagnosis. A therapist might be able to assist them in new emotions, stressors, and relationship challenges with the life change of a dementia diagnosis and as the disease progresses.
Community Support Services[edit | edit source]
Referrals to Home and Community Care Support Services (LHIN) could be considered currently in consultation with B.B. and her husband about their home situation and support system, especially moving forward with her disease. They offer a wide variety of services and support including: nursing/personal support worker (PSW), community physiotherapy and occupational therapy, social work, nutrition and food counselling (dietitians), palliative care, medical supplies and equipment, access to long-term care homes & supported living, access to adult day programs, and referrals to other community supports. This will be especially important as BB’s disease progresses and her husband requires more assistance as a caregiver [43].
BB’s local “The Alzheimer Society” could provide her and her husband with more tailored support for dementia. They advertise that they can help and support dementia patients with programs, services, education and resources tailored to you. Their services include peer support groups, learning series/ education, day programs and respite care, art and music therapy, brain health programs, and other local events[19].
Discussion[edit | edit source]
B.B. is a 60-year-old female who was initially diagnosed by a physician with Stage 3 Vascular Dementia Subcortical Subtype (also known as Binswanger’s Disease) and referred to an outpatient neurological rehabilitation centre. She was first admitted to a hospital due to a minor fall 2-weeks ago. Furthermore, it is believed by the PCP that B.B. had suffered a TIA within the past few months but went undiagnosed and untreated. Previously, B.B. was completely independent in all BADLs and IADLs despite living with a sedentary lifestyle and controlled hypertension.
As of lately, B.B. has noticed a decrease in her abilities to perform ADLs independently, decreased confidence in balance and strength, and several symptoms of cognitive decline including, but not limited to: memory loss, psychomotor slowness and weakness, and difficulty organizing her thoughts. Although vascular dementia is incurable, it would be beneficial for the inter-professional team to manage and prevent complications.
Regarding problems amenable to physical therapy care, it is remarkably important to address B.B.’s lack of confidence in balance by improving balance, strengthening weak muscles that may contribute to impaired balance, and maintaining functional range of motion. Some problems that physiotherapists may not be able to treat but ought to keep in mind include B.B.’s memory impairments, unpredictable changes in mood, behavior, and personality, and primary progressive aphasia which may create communication barriers with the patient.
Specific to B.B’s treatment plan, the physiotherapist will provide her with community-based PT treatment two times per week, primarily focused on improving balance and functional abilities. Following Canada’s Exercise Guidelines for Older Adults (>65 y/o), there will be 150 minutes of moderate to vigorous aerobic exercise and two days of strengthening exercises each week in addition to balance training. Strategies employed to improve compliance include the use of serious games and activities that B.B. enjoys such as Tai-Chi. Gait training with the use of a prescribed single point cane will also be used to decrease the risk of falls and improve functional community ambulation. Some practice strategies that would be beneficial to the interventions is providing minimum guidance, allowing self-evaluation and error detection, and feedback in the form of “knowledge of results”.
A reassessment was performed by the physiotherapist after four weeks of rehabilitation intervention. Overall, there is an increase in muscle strength in both the lower and upper extremities as measured in the manual muscle tests, mainly attributed to the strength training happening twice weekly. There is also a clinically significant improvement in B.B.’s balance as shown in the Berg Balance Scale, the MiniBESTest, and the Romberg test. With gait training and treadmill training, B.B. is able to increase her gait speed and walking endurance, which were measured by the 10-metre walk test, 6-minute walk test, and the TUG test. Based on the result of B.B’s 6-minute walk test (300m), it suggests that B.B. is safe to live in the community with minimal assistance; however, this may change over time as her dementia progresses. With the prescription of a cane and balance training, B.B. has significantly increased her confidence in her balance during daily activities, as measured by the ABC Scale. There was a slight yet insignificant decrease in time to complete the TUG dual task which is likely due to the mixed result from the neurologist’s intervention and normal dementia progression. Finally, there is no change in her Mini-Cog test and a worsening of her MoCA score, suggesting that B.B.’s cognitive ability is gradually deteriorating despite concurrent medical management by the neurologist and rehabilitation by the occupational therapist.
In conclusion, this case study demonstrates the challenges, impairments, and management interventions that can be used for patients with early stage Subcortical Vascular Dementia. The study also provides evidence of interventions and associated outcome measures that may be beneficial for this population. It is always important to understand problems within the scope of PT practice and when to refer out to other healthcare providers. Lastly, while this case study provides one way of providing treatment to this population, inter=professional teams ought to consider the context of each individual patient and their related goals.
Self Study Questionaires[edit | edit source]
1. Which of the following a common symptom in patients with Subcortical Vascular Dementia?
A) Psychomotor slowness
B) Memory impairments
C) Mood changes
D) Balance deficits
E) All of the above
2. Based on B.B.’s case, she received a score of 36/56 on the Berg Balance Scale, putting her at risk for falls. In order to improve her balance, what is the best evidence-based intervention that may benefit B.B.’s return to the community?
A) Mental imagery of balancing on one-leg
B) Tai-Chi
C) Tying her lower extremities together to challenge balance with narrower BOS
D) Sitting on a bed with no upper extremity support while the therapist adds external perturbations (e.g. shoves, lean and release, etc.)
3. Which of the following may NOT be an appropriate outcome measure for B.B.’s case in the initial assessment?
A) Community Balance and Mobility Scale (CBMS)
B) Timed Up and Go (TUG) Test
C) Activities-specific Balance Confidence Scale (ABC)
D) 10-metre walk test
Answers: 1) E 2) B 3) A
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Arvanitakis Z, Shah RC, Bennett DA. Diagnosis and management in Dementia. JAMA. 2020; 322(16): 1589-1599. http://doi.org/10.1001/jama.2019.4782
- ↑ 2.0 2.1 Hall AJ, Burrows L, Lang IA, Endacott R, Goodwin VA. Are physiotherapists employing person-centred care for people with dementia? an exploratory qualitative study examining the experiences of people with dementia and their carers. BMC Geriatrics. 2018; 18(1). https://doi.org/10.1186/s12877-018-0756-9
- ↑ 3.0 3.1 3.2 3.3 Ning H, Li R, Ye X, Zhang Y, Liu L. A review on serious games for dementia care in ageing societies. IEEE Journal of Translational Engineering in Health and Medicine. 2020; 8: 1–11. https://doi.org/10.1109/jtehm.2020.2998055
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 Román GC, Erkinjuntti T, Wallin A, Pantoni L, Chui HC. Subcortical ischaemic vascular dementia. The Lancet Neurology. 2002; 1(7): 426-436.
- ↑ Oguro H, Yamaguchi S, Abe S, Ishida Y, Bokura H, Kobayashi S. Differentiating Alzheimer’s disease from subcortical vascular dementia with the FAB test. Journal of neurology. 2006; 253(11): 1490-1494.
- ↑ 6.0 6.1 Văcăraș V, Cordoș AM, Rahovan I, Frunze S, Mureșanu DF. Binswanger's disease: Case presentation and differential diagnosis. Clinical Case Reports. 2020; 8(12): 3449–3456. https://doi.org/10.1002/ccr3.3459
- ↑ Guermazi A, Miaux Y, Rovira-Cañellas A, Suhy J, Pauls J, Lopez R, et al. Neuroradiological findings in vascular dementia. Neuroradiology. 2007; 49(1): 1-22.
- ↑ Prinivil, Zestril (lisinopril) dosing, indications, interactions, adverse effects, and more. (2021, November 11). Available from: https://reference.medscape.com/drug/prinivil-zestril-lisinopril-342321 (accessed May 22 2022
- ↑ Gras LZ, Kanaan SF, McDowd JM, Colgrove YM, Burns J, Pohl PS. Balance and gait of adults with very mild alzheimer disease. Journal of Geriatric Physical Therapy. 2015; 38(1): 1–7. https://doi.org/10.1519/jpt.0000000000000020
- ↑ Bergeron D, Vermette A, De La Sablonnière J, Cayer A-M, Laforce R, Bouchard RW. Finger-to-nose test findings in alzheimer’s disease. Journal of Alzheimer's Disease. 2016; 55(4): 1335–1337. https://doi.org/10.3233/jad-160941
- ↑ Kovacs CR. Age-related changes in gait and obstacle avoidance capabilities in older adults: a review. Journal of applied gerontology. 2005; 24(1): 21-34.
- ↑ Beauchet O, Allali G, Berrut G, Hommet C, Dubost V, Assal F. Gait analysis in demented subjects: Interests and perspectives. Neuropsychiatric disease and treatment. 2008; 4(1): 155.
- ↑ Kyrdalen IL, Thingstad P, Sandvik L, Ormstad H. Associations between gait speed and well‐known fall risk factors among community‐dwelling older adults. Physiotherapy research international. 2019; 24(1): e1743.
- ↑ Magnani PE, Genovez MB, Porto JM, Zanellato NF, Alvarenga IC, Freire RC, et al. Use of the bestest and the mini-bestest for fall risk prediction in community-dwelling older adults between 60 and 102 years of age. Journal of Geriatric Physical Therapy. 2019; 43(4): 179–184. https://doi.org/10.1519/jpt.0000000000000236
- ↑ Cedervall Y, Stenberg AM, Åhman HB, Giedraitis V, Tinmark F, Berglund L, et al. Timed up-and-go dual-task testing in the assessment of cognitive function: A mixed methods observational study for development of the UDDGAIT protocol. International Journal of Environmental Research and Public Health. 2020; 17(5): 1715. https://doi.org/10.3390/ijerph17051715
- ↑ 16.0 16.1 National Collaborating Centre for Mental Health. (2007). Therapeutic interventions for people with dementia: Cognitive symptoms and maintenance of functioning. Available from: (Accessed May 13 2019)
- ↑ 17.0 17.1 Rolland Y. Exercise and Dementia. Pathy's Principles and Practice of Geriatric Medicine. 2012; 1:911-921.
- ↑ 18.0 18.1 Forbes D, Thiessen EJ, Blake CM, Forbes SS, Forbes S. Exercise programs for people with dementia. Sao Paulo Medical Journal. 2014; 132: 195-196.
- ↑ 19.0 19.1 Alzheimer society of Kingston, Frontenac, Lennox & Addington counties. (2022). Avaiable from: https://alzheimer.ca/kfla/en (accessed May 12 2022)
- ↑ Canada.ca. Available from: https://www.canada.ca/en/public-health/services/publications/healthy-living/physical-activity-tips-older-adults-65-years-older.html (Accessed May 2 2022)
- ↑ Soares AV, Júnior NB, Hounsell MS, Marcelino E, Rossito GM, Júnior YS. A serious game developed for physical rehabilitation of frail elderly. European Research in Telemedicine/La Recherche Européenne en Télémédecine. 2016; 5(2): 45-53.
- ↑ 22.0 22.1 Rossito GM, Berlim TL, da Silva Hounsell M, Vinicius A, em Neuroreabilitação–NUPEN, N. D. P. (2014). SIRTET-K3D: a serious game for balance improvement on elderly people.
- ↑ 23.0 23.1 23.2 23.3 Breton UZS, Zapirain BG, Zorrilla AM. Kimentia: Kinect based tool to help cognitive stimulation for individuals with dementia. 2012 IEEE 14th International Conference on e-Health Networking, Applications and Services (Healthcom). 2012 https://doi.org/10.1109/healthcom.2012.6379430
- ↑ Lim KHL, Pysklywec A, Plante M, Demers L.The effectiveness of Tai Chi for short-term cognitive function improvement in the early stages of dementia in the elderly: a systematic literature review. Clinical Interventions in Aging. 2019; 14: 827.
- ↑ Nyman SR, Ingram W, Sanders J, Thomas PW, Thomas S, Vassallo M, et al. Randomised controlled trial of the effect of Tai Chi on postural balance of people with dementia. Clinical Interventions in Aging. 2019; 14:2017.
- ↑ Berg KO, Wood-Dauphinee SL, Williams JI, Maki B. Measuring balance in the elderly: validation of an instrument. Canadian journal of public health = Revue canadienne de sante publique. 1992; 83 (Suppl 2): S7–S11.
- ↑ Donoghue D, Stokes EK. How Much Change Is True Change? the Minimum Detectable Change of the Berg Balance Scale in Elderly People. Journal of Rehabilitation Medicine.2009; 41(5): 343–346., https://doi.org/10.2340/16501977-0337.
- ↑ Longhurst, Jason, et al. Physical Therapy for Gait, Balance, and Cognition in Individuals with Cognitive Impairment: A Retrospective Analysis. Rehabilitation Research and Practice. 2020; 2020: 1–12., https://doi.org/10.1155/2020/8861004.
- ↑ Lusardi Michelle M, et al. Functional Performance in Community Living Older Adults. Journal of Geriatric Physical Therapy. 2003; 26(3): 14–22., https://doi.org/10.1519/00139143-200312000-00003.
- ↑ Godi, Marco, et al. Responsiveness and Minimal Clinically Important Difference of the Mini-Bestest in Patients with Parkinson’s Disease. Gait & Posture. 2020; 80:14–19., https://doi.org/10.1016/j.gaitpost.2020.05.004.
- ↑ Potter Kirsten, Brandfass K. The Mini-Balance Evaluation Systems Test (Mini-Bestest). Journal of Physiotherapy. 2015;61(4): 2015, p. 225., https://doi.org/10.1016/j.jphys.2015.04.002.
- ↑ Barry, Emma, et al. Is the Timed up and Go Test a Useful Predictor of Risk of Falls in Community Dwelling Older Adults: A Systematic Review and Meta- Analysis. BMC Geriatrics. 2014; 14(1): https://doi.org/10.1186/1471-2318-14-14.
- ↑ 33.0 33.1 Shumway-Cook, Anne, et al. Predicting the Probability for Falls in Community-Dwelling Older Adults Using The Timed up & Go Test. Physical Therapy. 2000; 80(9): 896–903. https://doi.org/10.1093/ptj/80.9.896.
- ↑ Casanova C. et al. The 6-Min Walk Distance in Healthy Subjects: Reference Standards from Seven Countries. European Respiratory Journal 2010; 37(1): 150–156. https://doi.org/10.1183/09031936.00194909.
- ↑ Wise Robert A, Cynthia D Brown. Minimal Clinically Important Differences in the Six-Minute Walk Test and the Incremental Shuttle Walking Test. COPD: Journal of Chronic Obstructive Pulmonary Disease. 2005;2(1):125–129., https://doi.org/10.1081/copd-200050527.
- ↑ Raad Jason, et al. A Brief Review of the Activities-Specific Balance Confidence Scale in Older Adults. Archives of Physical Medicine and Rehabilitation. 2013; 94(7):1426–1427. https://doi.org/10.1016/j.apmr.2013.05.002.
- ↑ Davis Daniel HJ, et al. The Montreal Cognitive Assessment for the Diagnosis of Alzheimer’s Disease and Other Dementia Disorders. Cochrane Database of Systematic Reviews. 2013: https://doi.org/10.1002/14651858.cd010775.
- ↑ Agrawal Yuri, et al. The Modified Romberg Balance Test. Otology & Neurotology. 2011; 32(8):1309–1311., https://doi.org/10.1097/mao.0b013e31822e5bee.
- ↑ 39.0 39.1 39.2 39.3 Elsey C, Drew P, Jones D, Blackburn D, Wakefield S, Harkness K, et al. Towards diagnostic conversational profiles of patients presenting with dementia or functional memory disorders to memory clinics. Patient Education and Counseling. 2015; 98(9): 1071–1077. https://doi.org/10.1016/j.pec.2015.05.021
- ↑ Miki E, Kataoka T, Okamura H. Clinical usefulness of the Frontal Assessment Battery at bedside (FAB) for elderly cancer patients. Supportive Care in Cancer. 2012; 21(3):857–862. https://doi.org/10.1007/s00520-012-1595-4
- ↑ 41.0 41.1 41.2 Therapeutic interventions for people with dementia – cognitive symptoms ... (n.d.). Available from: https://www.ncbi.nlm.nih.gov/books/NBK55462/ ( Accessed May 12 2022)
- ↑ Farrajota L, Maruta, C, Maroco J, Martins IP, Guerreiro M, de Mendonça A. Speech therapy in primary progressive aphasia: A pilot study. Dementia and Geriatric Cognitive Disorders Extra. 2012; 2(1), 321–331. https://doi.org/10.1159/000341602
- ↑ Warrick N, Prorok JC, Seitz D. Care of community-dwelling older adults with dementia and their caregivers. Canadian Medical Association Journal. 2018; 190(26). https://doi.org/10.1503/cmaj.170920











