Iron
Original Editor - Lucinda hampton
Top Contributors - Lucinda hampton, Kim Jackson and Rucha Gadgil
Introduction[edit | edit source]
Iron is an essential element for almost all living organisms as it participates in a wide variety of metabolic processes, including oxygen transport, deoxyribonucleic acid (DNA) synthesis, and electron transport. However, as iron can form free radicals, its concentration in body tissues must be tightly regulated because in excessive amounts, it can lead to tissue damage.
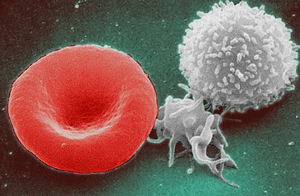
Iron is responsible for the production of hemoglobin, a protein that allows red blood cells (image at R red blood cell) to carry oxygen to every part of your body. It's also a component of myoglobin, which is similar to hemoglobin but found in muscle cells. Low iron levels body reduce red blood cell production, which impacts health.
Disorders of iron metabolism are among the most common diseases of humans and encompass a broad spectrum of diseases with diverse clinical manifestations including:
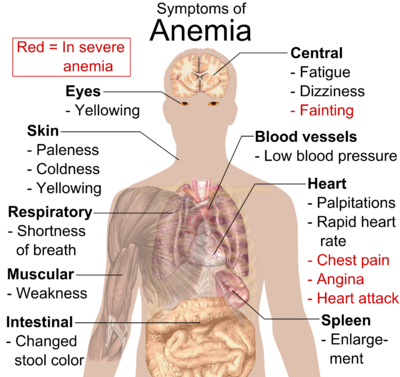
- Anaemia (including Anemia of chronic disease)
- Iron overload disorders eg Hemochromatosis
- Possibly to Neurodegenerative Diseases[1][2].
Iron Function[edit | edit source]
Iron is an essential element for blood production.
- In the living body, iron is not freely available. The bulk of the metal is locked up in ferritin, hemosiderin, myoglobin, and in the hemoglobin in red cells. The iron-binding proteins, transferrin and lactoferrin, which possess only a minute fraction of the total body iron, are normally only partly saturated with Fe and have an exceptionally high association constant of about 10 36 for the metal.[3]
- About 70 percent of your body's iron is found in the red blood cells in hemoglobin and in muscle cells called myoglobin. Hemoglobin is essential for transferring oxygen in the blood from the lungs to the tissues. Myoglobin, in muscle cells, accepts, stores, transports and releases oxygen.
- About 6 percent of body iron is a component of certain proteins, essential for respiration and energy metabolism, and as a component of enzymes involved in the synthesis of collagen and some neurotransmitters. Iron also is needed for proper immune function.
- About 25 percent of the iron in the body is stored as ferritin, found in cells and circulates in the blood. The average adult male has about 1,000 mg of stored iron (enough for about three years), whereas women on average have only about 300 mg (enough for about six months). When iron intake is chronically low, stores can become depleted, decreasing hemoglobin levels.
When iron stores are exhausted, the condition is called iron depletion. Further decreases may be called iron-deficient erythropoiesis and still further decreases produce iron deficiency anemia.[4]
Iron is the nutrient that helps blood move oxygen from the lungs to the rest of the body and it helps muscles store & use oxygen.
Low iron levels can impact :
- Energy levels
- Concentration
- Memory
- Performance at work / school
- Cognitive development
- Learning & Earning potential[5]
Iron Deficiency[edit | edit source]
World Health Organization (WHO) estimates that two billion people are anemic worldwide and attribute approximately 50% of all anemia to iron deficiency. It occurs at all stages of the life cycle but is more prevalent in pregnant women and young children. Anemia is the result of a wide variety of causes that can be isolated, but more often coexist
Inorganic iron is needed for hemoglobin synthesis and oxygen transport - Low iron intake and/or bioavailability are responsible for
- Most anemia in industrialized countries;
- In developing countries they account for only about half of the anemia; infectious and inflammatory diseases (especially malaria), blood loss from parasitic infections, and other nutrient deficiencies (vitamin A, riboflavin, folic acid, and vitamin B12) account for the other 50% of anemias[2].
Dietary Iron[edit | edit source]
Dietary iron occurs in two forms: heme and nonheme.
- The primary sources of heme iron are hemoglobin and myoglobin from consumption of meat, poultry, and fish. Heme iron is highly bioavailable (15%-35%) and dietary factors have little effect on its absorption, whereas nonheme iron absorption is much lower (2%-20%) and strongly influenced by the presence of other food components.
- Nonheme iron is obtained from cereals, pulses, legumes, fruits, and vegetables. The quantity of nonheme iron in the diet is manyfold greater than that of heme-iron in most meals so despite its lower bioavailability, nonheme iron generally contributes more to iron nutrition than heme-iron.
Bioavailability
- Major inhibitors of iron absorption are phytic acid (a natural antioxidant that is mainly found in grains, nuts, and seeds)[6], polyphenols, calcium, and peptides from partially digested proteins.
- Enhancers are muscle tissue and ascorbic acid[2] ie vitamin C. To enhance iron uptake the following are options: lemon juice drizzled over food; salads containing high vitamin C sources such as red capsicum or tomato; kiwi fruit, strawberries, papaya or a glass of freshly squeezed orange juice; mixing baby spinach in salads - green leafy vegetables contain iron and vitamin C.
Note the absorption of non-haem iron varies considerably, and has been shown to be higher in people who have greater iron requirements. This suggests the body adapts to low iron by increasing its absorption[7].
Competition studies suggest that several other heavy metals may share the iron intestinal absorption pathway. eg lead, manganese, cobalt, and zinc. As iron deficiency often coexists with lead intoxication, this interaction can produce particularly serious medical complications in children[2].
Some iron-rich foods include:
- Liver and other Organ Meats
- Shellfish- such as oysters, clams, mussels
- Steak and other red meats
- Chicken, Turkey, Duck and other poultry
- Tofu
- Dark leafy vegetables (spinach, kale)
- Legumes- such as chickpeas, lentils, soybeans, kidney beans
- Nuts & seeds, especially pumpkin seeds
- Quinoa[5]
Iron Requirements[edit | edit source]
Dietary iron requirements vary by age and gender. The recommended daily intake (average daily intake that is sufficient to meet the needs of the majority of people) for males varies from eight to 11 milligrams a day for ages one to 18 years, and eight milligrams for all other ages.
Women have higher requirements. For ages 14-50 years, recommended daily intakes range from 15 milligrams (14-18 years) to 18 milligrams a day. Needs are higher during pregnancy, jumping to 27 milligrams per day. However during lactation they are slightly less, at nine to ten milligrams a day.
Iron requirements for vegetarians have been estimated as 1.8 times more than non-vegetarians (based on limited research)[7].
Iron and Bacteria[edit | edit source]
In response to a systemic bacterial infection, the immune system initiates a process known as iron withholding (resulting in hypoferremia). If bacteria are to survive, then they must obtain iron from their environment.
- During bacterial infection, pathogen and host compete for iron (Fe). The inflammatory response associated with infection shifts Fe from the circulation into storage, resulting in hypoferremia and iron-deficient erythropoiesis, and ultimately contributing to the anemia of inflammation.
- Bacteria employ both membrane-bound transferrin receptors and high-affinity iron-binding proteins called siderophores to acquire Fe.
- Humans utilize the iron-binding proteins lactoferrin, transferrin, and ferritin to move Fe away from sites of infection and into storage. Synthesis and action of these proteins are regulated by inflammatory cytokines.
- Humans respond to infection with inflammatory cytokine-induced hypoferremia.[8]
Iron deficiency is very common in humans and animals. Low concentrations of iron in serum (hypoferremia), caused by the hormone hepcidin, inhibit the body’s response to vaccines and infections; conversely, increasing iron can boost immunity[9].
- Hepcidin is a circulating peptide hormone secreted by the liver that plays a central role in the regulation of iron homeostasis. It is the master regulator of systemic iron homeostasis, coordinating the use and storage of iron with iron acquisition. This hormone is primarily produced by hepatocytes and is a negative regulator of iron entry into plasma.[2]
Iron in Neurodegeneration[edit | edit source]
Iron levels in the brain and body increase sharply up to 30 years of age due to a metabolic need during the growth process and remain stable during adulthood. In the aging brain however, region-specific increase of total iron is observed, probably triggered by inflammation, increased blood-brain barrier (BBB) permeability, redistribution of iron within the brain, and changes in iron homeostasis and shows highest iron levels in the basal ganglia.
Iron dyshomeostasis can cause neuronal damage to iron-sensitive brain regions. Neurodegeneration with brain iron accumulation reflects a group of disorders caused by iron overload in the basal ganglia. High iron levels and iron related pathogenic triggers have also been implicated in sporadic neurodegenerative diseases including Alzheimer’s disease (AD), Parkinson’s disease (PD), and multiple system atrophy (MSA). Iron-induced dyshomeostasis within vulnerable brain regions is still insufficiently understood[2]
References[edit | edit source]
- ↑ Frontiers in neuroscience. 2019 Mar 1;13:180.Available from: https://www.frontiersin.org/articles/10.3389/fnins.2019.00180/full(Accessed 13.3.2021)
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Abbaspour N, Hurrell R, Kelishadi R. Review on iron and its importance for human health. Journal of research in medical sciences: the official journal of Isfahan University of Medical Sciences. 2014 Feb;19(2):164.Available from:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3999603/ (accessed 4.3.2021)
- ↑ Bullen JJ, Rogers HJ, Griffiths E. Role of iron in bacterial infection. Current topics in microbiology and immunology. 1978:1-35.Available from:https://link.springer.com/chapter/10.1007%2F978-3-642-66956-9_1 (accessed 5.3.2021)
- ↑ https://www.ucsfhealth.org/education/hemoglobin-and-functions-of-iron
- ↑ 5.0 5.1 Lucky fish Facts about iron Available from: https://luckyironfish.com/pages/iron-101 (accessed 4.3.2021)
- ↑ Very well health Phytic acid Available from: https://www.verywellhealth.com/phytic-acid-5088824 (accessed 4.3.2021)
- ↑ 7.0 7.1 The Conversation Why iron is such an important part of your diet Available from:https://theconversation.com/why-iron-is-such-an-important-part-of-your-diet-69974 (accessed 4.3.2021)
- ↑ Pieracci FM, Barie PS. Iron and the risk of infection. Surgical infections. 2005 Jun 1;6(S1):s41-6.Available from: https://pubmed.ncbi.nlm.nih.gov/19284357/(accessed 5.3.2021)
- ↑ Frost JN, Tan TK, Abbas M, Wideman SK, Bonadonna M, Stoffel NU, Wray K, Kronsteiner B, Smits G, Campagna DR, Duarte TL. Hepcidin-mediated hypoferremia disrupts immune responses to vaccination and infection. Med. 2020 Nov 19.Available from: https://www.cell.com/med/fulltext/S2666-6340(20)30021-0(accessed 5.3.2021)