Basal Ganglia
Introduction[edit | edit source]
The “basal ganglia” refers to a group of subcortical nuclei within the brain responsible primarily for motor control, as well as other roles such as motor learning, executive functions, emotional behaviours, and play an important role in reward and reinforcement, addictive behaviours and habit formation.
The basal ganglia are located at the base of the forebrain (cerebrum) and have attracted attention in medicine for various disturbances that appear with dysfunctions caused by diseases or trauma. Disruption of the basal ganglia network forms the basis for several movement disorders e.g. Parkinson's Disease, Huntington Disease.
Proposed more than two decades ago, the classical basal ganglia model shows how information flows through the basal ganglia back to the cortex through two pathways with opposing effects for the proper execution of movement[1].
Much of the classical model has remained today, however the model has been modified and amplified with the emergence of new data. The basal ganglia network is now viewed as multiple parallel loops and re-entering circuits whereby motor, associative, and limbic territories are engaged. These circuits deal mainly in the control of movement, behaviour, and emotions. These parallel circuits subserve the other functions of the basal ganglia engaging associative and limbic territories.[1]
Structure[edit | edit source]
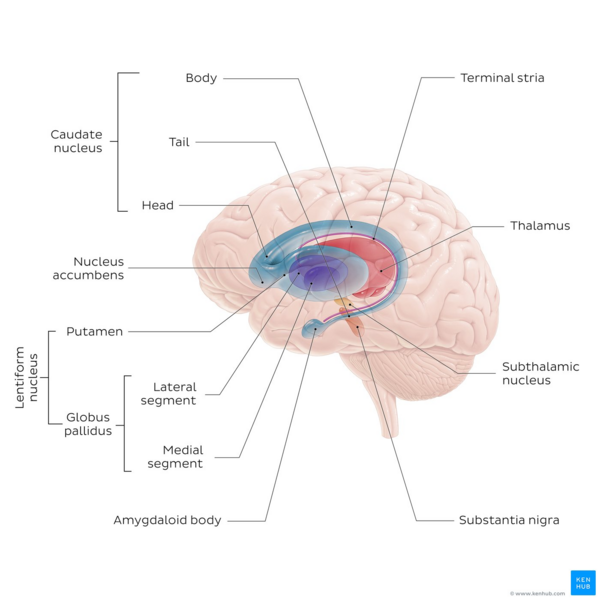
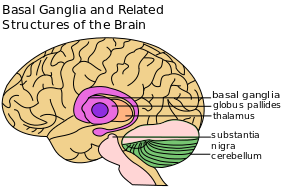
The basal ganglia (BG) are a cluster of subcortical nuclei deep to cerebral hemispheres. The largest component of the BG is the corpus striatum. The corpus striatum is made up of the caudate nucleus and lenticular nuclei, which in turn is made up of the putamen, globus pallidus externus, and globus pallidus internus. The subthalamic nucleus (STN), and the substantia nigra (SN) are also components of the BG. These structures intricately synapse onto one another to promote or antagonize movement.[2]
Divisions of the BG i.e. subcortical nuclei:
- Corpus Striatum- Has a volume of approximately 10 cm. It is a heterogeneous structure that receives afferents from several cortical and subcortical structures and projects to various BG nuclei.[1] Within the striatum, there are two main divisions:
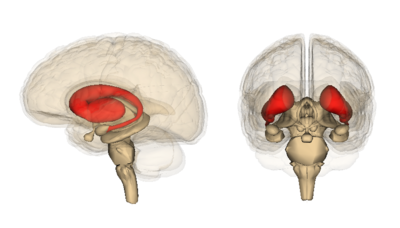
- Dorsal striatum (DS) see image, shown in red. Primarily involved in control over conscious motor movements and executive functions. The dorsal striatum consists of the caudate nucleus and the putamen. A white matter nerve tract (the internal capsule) in the dorsal striatum separates the caudate nucleus and the putamen.
- Ventral striatum, responsible for limbic functions of reward and aversion. Consists of nucleus accumbens and the olfactory tubercle.[2]
- Internal and External segments of Globus Pallidus (NB until the first half of the 19th century the globus pallidus and putamen were considered one structure, collectively referred to as the lentiform or lenticular nucleus[3])
- Subthalamic Nucleus (STN) - a lens-shaped cell group that makes up the largest part of the subthalamus
- Substantia Nigra (SN) - (“black substance” in Latin) is a long nucleus located in the midbrain but considered functionally a part of the BG because of its reciprocal connections with other brainstem nuclei. It consists of two components, the pars compacta and the pars reticulata, which have different connections and use different neurotransmitters. [4]
The 2 videos below outline BG concepts.
Basal Ganglia - Current Concepts[edit | edit source]
The original functional organization of the BG was conceived as a loop, in which cortical afferent activity is dispatched to and modulated by the basal ganglia, which subsequently sends back a signal to the cortex to facilitate (or inhibit) motor activity. The BG were featured as a “go through” station within the motor loop. Current thinking now is that the BG has several loops, where cortical and subcortical projections interact with internal re-entry loops forming a complex network, ideally designed for selecting and inhibiting simultaneously occurring events and signals[1].
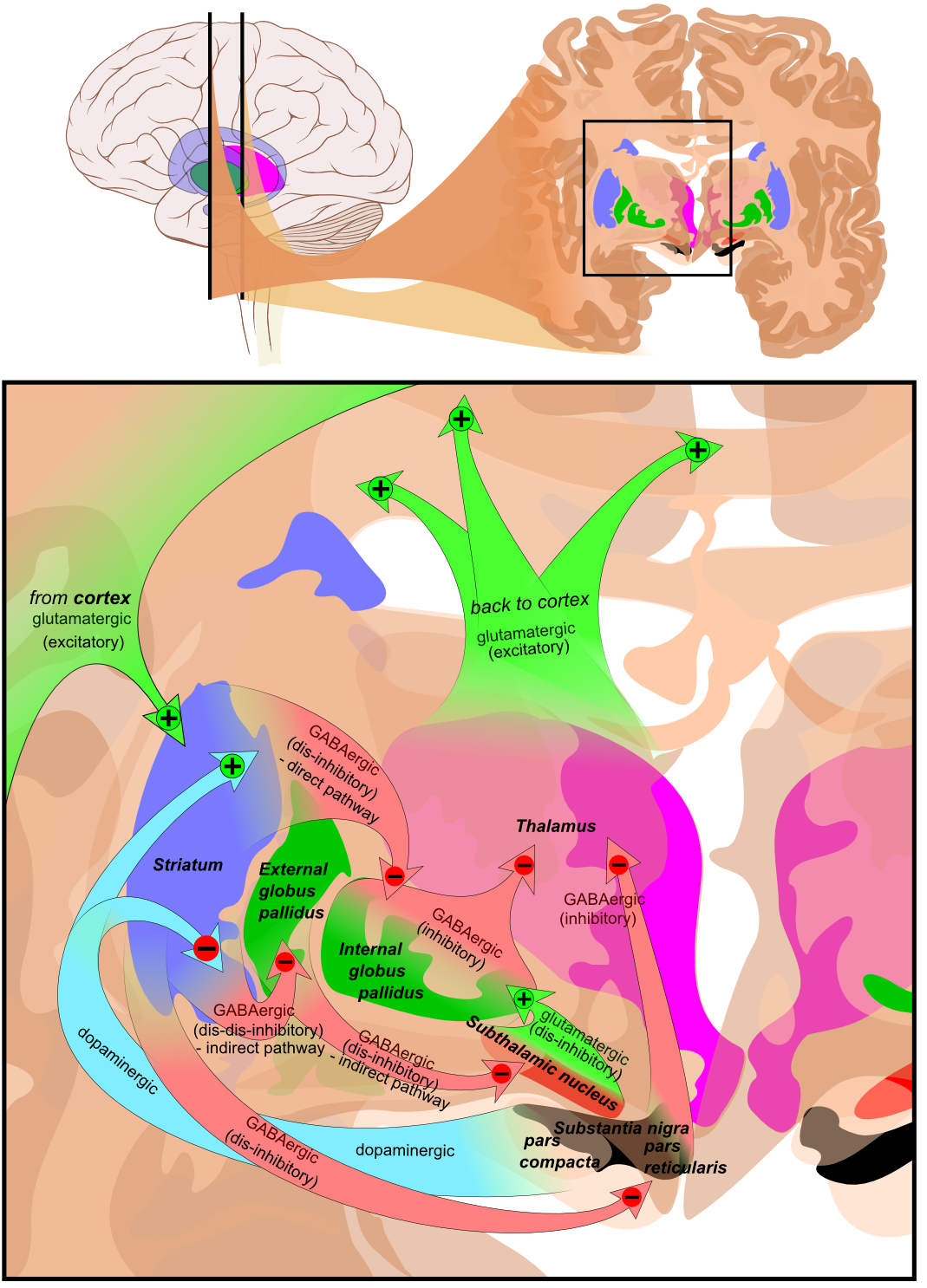
Main circuits of the BG are shown below. (Very complicated and involved circuits exist)
Coronal slices that have been superimposed to include the involved BG structures. The' +' and '-' signs at the point of the arrows indicate respectively whether the pathway has an excitatory or inhibitory effect. Green arrows refer to excitatory glutamatergic pathways, red arrows refer to inhibitory GABAergic pathways and turquoise arrows refer to dopaminergic pathways that are excitatory on the direct pathway and inhibitory on the indirect pathway. Note that dis-inhibitory pathways in effect are excitatory on the feedback to the cortex, while dis-dis-inhibitory pathways in effect are inhibitory.
Pathophysiology[edit | edit source]
The BG are particularly associated with movement disorders. Associated with damage to the BG are: tremors; involuntary muscle movements; abnormal increase in tone; difficulty initiating movements; abnormal posture.
Movement disorders comprise a variety of motor problems, not all of which are associated with dysfunction of the BG. Those that have a clearly established pathological basis and are caused by pathophysiological mechanisms directly involving the BG include[2]
Parkinson's[edit | edit source]
Parkinson's is the most notorious disease of the BG. Classic clinical symptoms include:
- Bradykinesia
- Resting tremor
- Postural instability
- Shuffling gait
This disease is a result of neurodegeneration of the SNpc dopaminergic neurons. Often found in the Parkinsonian striatum, alpha-synuclein protein aggregates form toxic “Lewy bodies,” which are inclusions within neurons. The substantia nigra, due to degeneration, loses its grossly visible dark pigmentation, a concomitant sign of dopamine biosynthesis dysfunction. This loss of dopamine depresses the nigrostriatal pathway. With decreased dopaminergic input the striatum exerts less positive motor activity and more negative motor inhibition. This gives the characteristic hypokinetic dysfunction found in these patients.[2]
Huntington Disease[edit | edit source]
Huntington disease is a hyperkinetic movement disorder. Its cause is a genetic defect manifesting as a CAG repeat on chromosome 4p on the HTT gene. This creates an abnormally long Huntington gene which leads to neuronal death in the caudate and the putamen. The indirect pathway is interrupted and leads to a hyperkinetic presentation. Symptoms include involuntary movements such as chorea, cognitive degeneration, and psychiatric dysfunction.[2]
Hemiballism[edit | edit source]
Hemiballism (from the Greek “to throw”) is used to describe hyperkinetic, involuntary, forceful movements of the ipsilateral arm and leg. Commonly, a lesion in the contralateral subthalamic nuclei causes hemiballism. Given that the subthalamus is part of the indirect pathway this lesion reduces or eliminates indirect pathway signaling, leading to a relative overabundance of activity in the direct pathway. Such causes include stroke, traumatic brain injury, amyotrophic lateral sclerosis, nonketotic hyperglycemia, neoplasm, vascular malformation, and other causes.[2]
Tourette Syndrome[edit | edit source]
Tourette syndrome has been shown to have a significant neurological BG component which manifests as sudden, repetitive uncontrolled movements and vocalizations, called “tics.” These tics have been associated with dysfunction of the GABAergic projections from the striatum, leading to a relative increase in dopaminergic activity much like in hemiballism and Huntington’s disease.
Additionally, parts of the basal ganglia play a key role in reward and reinforcement, addictive behaviours and habit formation. Pathophysiological processes underlying psychiatric disorders such as depression and obsessive-compulsive disorder (OCD) involve the BG and their connections with many other structures (particularly to the prefrontal cortex and the limbic system).[7] In terms of cognitive disorders, BG abnormalities have been found in individuals with schizophrenia[8] and may explain the presence of learning deficits associated with the disorder.
Silent Cerebral Infarcts - of interest a 2008 study found that approx. 5% of healthy middle-aged adults have microlesions in their BG. [9]
Physiotherapy - Implications From Recent Studies[edit | edit source]
The importance and value of exercise is becoming more and more apparent for a whole raft of health conditions. Here are a few of the latest findings regarding exercise and BG function.
- A 2016 study by Becker et al[10] into cognitive performance and BG changes concluded that physical activity, especially motor fitness level training, might be a promising tool that leads to structural changes in the basal ganglia. This might have the potential to diminish the cognitive decline in older adults and to support academic success in children and young adults.
- Exercise is beneficial and should be routinely prescribed in the management of PD (may aid in BG function)[11].
- Exercise effects on BG damage:[11]
- The adult brain possesses a tremendous capacity for experience-dependent neuroplasticity, even in the context of ageing and neurodegenerative disorders including PD, where the activation of neurotrophic factors may play a key role.
- Animal models of dopamine depletion are beginning to reveal the underlying mechanisms by which exercise can remodel the brain through alteration in neuronal synaptic connections, especially dopaminergic and glutamatergic neurotransmission within the basal ganglia.
- Epidemiological studies, clinical observations, and animal research indicate that appropriately dosed physical activity and exercise may not only reduce the risk of developing PD in vulnerable populations but also benefit PD patients by potentially protecting the residual nigrostriatal dopamine neurons or directly restoring the dysfunctional cortico-basal ganglia motor control circuit, and these benefits may be mediated by exercise-triggered production of endogenous neuroprotective molecules such as neurotrophic factors[12].
- Animal studies have been instrumental in providing evidence for exercise-induced neuroplasticity of cortico-striatal circuits that are profoundly affected in Parkinson’s disease. Exercise has been implicated in modulating dopamine and glutamate neurotransmission, altering synaptogenesis, and increasing cerebral blood flow. In addition, recent evidence supports that the type of exercise may have regional effects on brain circuitry, with skilled exercise differentially affecting frontal-striatal related circuits to a greater degree than pure aerobic exercise (with skilled exercise differentially affecting frontal related circuits more so than pure aerobic exercise). These effects are not mutually exclusive e.g. peddling on a recumbent bicycle may be considered predominantly aerobic with minimal skill or cognitive engagement whilst at the other end of the spectrum juggling may represent a highly skilled task with minimal aerobic involvement. However, many exercises such as swimming and running involve a combination of both skilled and aerobic exercise[13]. These results indicate that the community Parkinson Dance classes that exist are a great option, as are many other structured classes.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 Lanciego JL, Luquin N, Obeso JA. Functional neuroanatomy of the basal ganglia. Cold Spring Harbor perspectives in medicine. 2012 Dec 1;2(12):a009621.Available from:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3543080/ (last accessed 12.1.2020)
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Young CB, Sonne J. Neuroanatomy, basal ganglia. InStatPearls [Internet] 2018 Dec 28. StatPearls Publishing.Available from:https://www.ncbi.nlm.nih.gov/books/NBK537141/ (last accessed 12.1.2020)
- ↑ Neuroscientificallychallenged Know your brain Globus Pallidus 9.1.2019 Available from: https://www.neuroscientificallychallenged.com/blog/know-your-brain-globus-pallidus (last accessed 14.1.2020)
- ↑ Jacobs LK, Sapers BL. Neurological Disease. InPerioperative Medicine 2011 (pp. 343-359). Springer, London.Available from:https://www.sciencedirect.com/science/article/pii/B9780443068980000335 (last accessed 13.1.2020)
- ↑ Neuroscientifically Challenged Basal ganglia . Available from: https://www.youtube.com/watch?v=OD2KPSGZ1No [last accessed 14/01/2020]
- ↑ Armando Hasudungan. The Basal Ganglia Clinical Anatomy. Available from: https://www.youtube.com/watch?v=mNc-6q6YAAw [last accessed 05/01/2024]
- ↑ Stathis P, Panourias IG, Themistocleous MS, Sakas DE. Connections of the basal ganglia with the limbic system: implications for neuromodulation therapies of anxiety and affective disorders. InOperative Neuromodulation 2007 (pp. 575-586). Springer, Vienna. Available from: https://www.ncbi.nlm.nih.gov/pubmed/17691350/ (last accessed 13.1.2020)
- ↑ Kéri S. Interactive memory systems and category learning in schizophrenia. Neuroscience & Biobehavioral Reviews. 2008 Jan 1;32(2):206-18. Available from: https://www.ncbi.nlm.nih.gov/m/pubmed/17854895/ (last accessed 13.1.2020)
- ↑ Das RR, Seshadri S, Beiser AS, Kelly-Hayes M, Au R, Himali JJ, Kase CS, Benjamin EJ, Polak JF, O'Donnell CJ, Yoshita M. Prevalence and correlates of silent cerebral infarcts in the Framingham offspring study. Stroke. 2008 Nov 1;39(11):2929-35.Available from:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2712254/ (last accessed 13.1.2020)
- ↑ Becker L, Kutz DF, Voelcker-Rehage C. Exercise-induced changes in basal ganglia volume and their relation to cognitive performance. Available https://www.jneurology.com/articles/exerciseinduced-changes-in-basal-ganglia-volume-and-their-relation-to-cognitive-performance.html (last accessed 13.1.2020)
- ↑ 11.0 11.1 Petzinger GM, Fisher BE, Akopian G, Holschneider DP, Wood R, Walsh JP, Lund B, Meshul C, Vuckovic M, Jakowec MW. The role of exercise in facilitating basal ganglia function in Parkinson’s disease. Neurodegenerative disease management. 2011 Apr;1(2):157-70. Available from:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3691073/ (last accessed 13.1.2020)
- ↑ Hou L, Chen W, Liu X, Qiao D, Zhou FM. Exercise-induced neuroprotection of the nigrostriatal dopamine system in Parkinson's disease. Frontiers in aging neuroscience. 2017 Nov 3;9:358. Available from: https://www.frontiersin.org/articles/10.3389/fnagi.2017.00358/full (last accessed 14.1.2020)
- ↑ Petzinger GM, Holschneider DP, Fisher BE, McEwen S, Kintz N, Halliday M, Toy W, Walsh JW, Beeler J, Jakowec MW. The effects of exercise on dopamine neurotransmission in Parkinson’s disease: targeting neuroplasticity to modulate basal ganglia circuitry. Brain plasticity. 2015 Jan 1;1(1):29-39.Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4621077/ (last accessed 14.1.2020)