Degenerative Disc Disease
Original Editor Amanda E Davidson, Colby Boers, &Lisa Pernet as part of the Vrije Universiteit Brussel's Evidence-based Practice project
Top Contributors - Lisa Pernet, Gaëlle Vertriest, Admin, Vanbeylen Antoine, Amanda E Davidson, Garima Gedamkar, Candace Goh, Laura Ritchie, Scott Cornish, Uchechukwu Chukwuemeka, Kim Jackson, WikiSysop, 127.0.0.1, De Maeght Kim, Eric Robertson, Lucinda hampton and Aya Alhindi
Definition/Description[edit | edit source]
Degenerative disc disease (DDD) represents a wide category of back pain resulting from or associated with the degeneration of intervertebral discs[1]. It involves a process where the intervertebral discs lose height and hydration. When this occurs, the discs are unable to fulfill their primary functions of cushioning and providing mobility between the vertebrae. Although the exact cause is unknown, it is thought to be associated with the aging process during which the intervertebral discs dehydrate, lose elasticity, and collapse. Despite its name, degenerative disc disease is not so much of a disease, but a natural occurrence that comes with ageing[2]. Degenerative disc disease may develop at any level of the spine, but is most common in the cervical and lower lumbar regions.[3][4]
Clinically Relevant Anatomy[edit | edit source]
Lumbar DDD is a condition that maybe a cause of lower back pain, which results from the co-existence of two different time scales, the slow dynamics of disc degeneration and the fast dynamics of pain recurrence. [5]
Lumbar DDD can also imply radiating pain from damaged discs in the spine. A lumbar spinal disc acts as a shock absorber between two vertebrae and allows the joints and the spine to move easily. The outer region of the disc, the annulus fibrosis, surrounds the soft inner core of the disc, the nucleus pulposus. Spinal discs undergo degenerative changes as we age, but not everyone will have symptoms as a result of these changes. Neural inflammation is one such possible cause of pain. When the outer section of the disc ruptures, the inner core can leak out, releasing proteins that irritate the neural tissue. Another cause is when discs can no longer absorb stress as well, leading to abnormal movement around the vertebral segment and causing back muscles spasm as they try to stabilise the spine. In some cases the segment may collapse, causing nerve root compression and radiculopathy. Pain often reduces with time as the inflammatory proteins dissipate and the disc collapse settles into a stable position. [6]
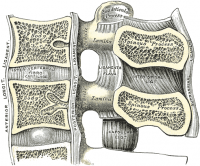
Intervertebral Discs
Degenerative Disc Disease is thought to begin with changes to the annulus fibrosis, intervertebral disc, and subchondral bone. The process of degeneration is divided into three classifications: early dysfunction, intermediate instability, and final stabilisation.
- Early dysfunction is the start of degenerative changes which can occur as early as 20 years old.
- Intermediate instability is classified by a loosening of the annulus fibrosis, which can cause lower back pain.
- Final stabilisation is where fibrosis develops in the posterior structures and osteophytes form. Pain and motion both decrease. [7]
Epidemiology /Etiology[edit | edit source]
Degenerative disc disease is fairly common and it is estimated that at least 30% of people aged 30-50 years old will have some degree of disc space degeneration, although not all will have pain or ever receive a formal diagnosis.[8] The pain is frequently caused by simple wear and tear as part of the general ageing process. It can also be as a result of a twisting injury to the lower back.
The process that leads to DDD begins with structural changes. The annulus fibrosis loses water content over time which makes it increasingly unyielding toward the daily stresses and strains placed on the spine. The loss of compliance in the discs contributes to forces being redirected from the anterior and middle portions of the facets to the posterior aspect, causing facet arthritis. Hypertrophy of the vertebral bodies adjacent to the degenerating disc also results. These overgrowth are known as bony spurs or osteophytes.
Characteristics/Clinical Presentation[edit | edit source]
DDD commonly occurs with other diagnoses such as:
- idiopathic low-back pain
- lumbar radiculopathy .
- myelopathy .
- lumbar stenosis.
- spondylosis[4].
- osteoarthritis .
- zygapophyseal joint degeneration. [7]
Activities that typically increase pain include:
- Sitting for extended periods of time.
- Rotating, bending, or lifting.
Activities that typically decrease pain include:
- Frequent changes in positions.
- Lying down.
- Staying active.[9]
Differential Diagnosis[edit | edit source]
People with DDD will often present with low back pain[10] with varying levels of severity between individuals. Pain is often chronic, but can also be acute on chronic with varying episodes of exacerbation [9]
There are different degrees of annular disruption[3] which are classified into 5 grades. These grades are differentiated by means of a contrast medium injection.
- Grade 0: no disruption
- Grade 1: the contrast medium passes into the cartilage endplate through a tear
- Grade 2: the contrast medium flows into the bony endplate
- Grade 3: the contrast medium enters into the cancellous bone of vertebra under endplate
- Grade 4: the contrast medium leaks completely in the cancellous bone.
Diagnostic Procedures[edit | edit source]
Provocation discography is a diagnostic test to identify a painful disc. To evaluate the degree of disruption, a combination of discogram and CT scan after discography is used.[3] X-ray findings can also be used to diagnose DDD. Anterior-Posterior and lateral views are taken where presence of osteophytes, narrowing of the disc joint space, or a “vacuum sign” is noted. [11]
Outcome Measures[edit | edit source]
None of the literature has reported a uniform system of outcome measures. The most common form of outcome measure for DDD is the Oswestry Disabilty Index (ODI) in combination with other forms of outcome measures, such as: Short Form 36 (or SF-12) questionnaire, self-paced walk, timed up-and-go test (TUG), Visual Analogue Scale (VAS) and the Roland-Morris disability index (RMDI) [12][13] .
Carreon found that the Oswestry Disability Index is a good primary outcome measure for lumbar fusion and nonsurgical interventions for various symptomatic degenerative spine disorders, [14] although further research is needed.
Examination[edit | edit source]
The patient’s history is a valuable tool for identifying the intervertebral disc as the nociceptive source. Patients may present with a history of chronic low back pain as well as symptoms in the gluteal region and stiffness in the spine which worsens with activity and tenderness on palpation over the involved area.[11]
Mood and anxiety disorders are associated with neurological deficits and more commonly seen in patients with lumbar or cervical disc herniation than in those without herniation.[15] No relationship was detected between pain severity and mood or anxiety disorders, however. These disorders can be diagnosed using the Structured Clinical Interview of the Diagnostic and Statistical Manual of Mental Disorders.
MRI is the most commonly used method of specifically assessing intervertebral disc degeneration. Based on proton density, water content and chemical environment, MRI depicts disc hydration and morphology. Pfirrmann et al devised a grading system for disc degeneration based on MRI signal intensity, disc structure, distinction between nucleus and annulus, and disc height.[16] This useful grading system has been accepted and applied clinically.
The modified system comprises 8 grades for lumbar disc degeneration. [17] Sagittal T2 weighted images were used for classification as they provide a comprehensive perception of disc structure and good tissue differentiation. The 8 grades represent a progression from normal disc to severe disc degeneration with Grade 1 corresponding to no disc degeneration and Grade 8 corresponding to end stage degeneration. As well as the 8 grade table there is also an image reference panel. [17]
Medical management[edit | edit source]
The preferred treatment protocol for patients with chronic low back pain, as a result of disc degeneration, is conservative management of physical therapy and medication.[9]
Conservative treatment includes rest, adequate stimulation for motor activity, regular physical activity, muscle strengthening, analgesic medication, physiotherapy, rehabilitation programs and lifestyle adjustments, such as weight loss. [18]
Medications such as non-steroidal anti-inflammatory drugs (NSAIDs) and acetaminophen (e.g. Tylenol) help patients to feel confident enough to engage in their regular activities. Stronger prescription medications such as oral steroids, muscle relaxants or narcotic pain medications may also be used to manage intense pain episodes on a short-term basis only and some patients may benefit from an epidural steroid injection. Epidural steroid injections can provide low back pain relief by delivering medication directly to the painful area to decrease inflammation.
Successful outcomes have been demonstrated by animal experiments with mesenchymal stem cells.[18][3] Surgical intervention include disc arthroplasty and lumbar spine fusion to reduce the chronic low back pain[18][20][3].
The Device for Intervertebral Assisted Motion (DIAM) is another option of chirurgical management for the treatment of DDD. The DIAM is a polyester encased silicone interspinous dynamic stabilisation device that can unload the anterior column and re-establish the functional integrity of the posterior column. This device is designed for preservation of the functional spinal unit. [21]
In cases where patients are not responsive to nonsurgical treatment, a lumbar total disc replacement (TDR) is an option. Patients presenting with symptomatic single level lumbar DDD who have failed at least 6 months of nonsurgical management were randomly allocated to treatment with an investigational TDR device (also called: TDR activL device) or FDA approved control devices. After 2 years of research, these devices are deemed to be safe and effective for the treatment of symptomatic lumbar DDD. [22]
Physical Therapy management[edit | edit source]
One of the main aims of physical therapy is to reduce pain. Various physical modalities are used, including heat and cold application, traction, spinal manipulations [23] [24] [25], exercise programs and electrical stimulation such as ‘TENS’ and ‘pulsed radiofrequency (PRF)’ treatment[26] and lifestyle modifications (e.g., weight reduction, smoking cessation).[24] Amongst exercise approaches, unloaded movement facilitation exercises of McKenzie, core strengthening, and core stabilisation exercises are all effective in pain reduction for degenerative disc disease.[27]
Spinal manipulations
Spinal manipulations have traditionally been used to relieve lower back pain, but the effects are generally only temporary. The HVLA (High-Velocity, Low-amplitude) is a manipulation that includes many different techniques and may involve preliminary preparation of the joint and its surrounding tissues, using stretching, assisted motion and other methods. Loads, both forces and moments, are applied to the joint, and it is moved to its end range of voluntary motion. An impulse is then applied, the effective load is the summation of forces applied by the therapist, with the inertial forces generated by the motion of body segments, and the internally generated tensions from client muscle reactions. [28]
This technique can immediately improve self-perceived pain, spinal mobility in flexion and hip flexion during the passive SLR test, [29] but Paige et al reported only moderate improvements in pain and with (transient) minor musculoskeletal harms. [30] Before Spinal Manipulation Therapy (SMT) can be considered as a treatment option, patients with LBP need to be screened for possible serious pathology. There are two reasons for this: some conditions, such as a fracture, affect the mechanical integrity of the spine and would make SMT clearly dangerous. In other conditions, a failure to recognise the condition delays commencement of more appropriate care. For example, early detection and treatment of spinal malignancy is important to prevent the spread of metastatic disease and the development of further complications such as spinal cord compression. Application of SMT with the presence of any red flags are considered as contraindications to SMT until further investigation has excluded other pathologies.
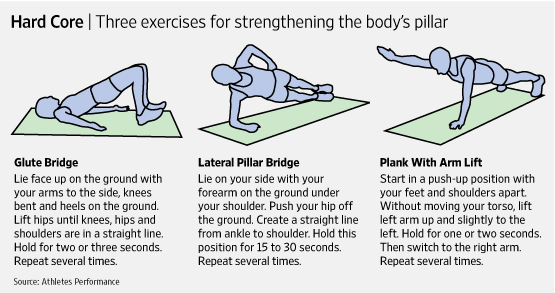
Core Stability
Strength training aims to enhance core stability by strengthening and improving the coordination between the abdominal and back muscles.[31] Stabilisation exercises will increase a patient’s capacity to resist higher loads in the degenerative discs[32]. This is a key element for prevention and treatment of injury as muscle tissue reduces at a rate of 1 kg/year after the age of 40.[33] A posterior dynamic stabilisation programme will have result in a significant improvement in pain and disability.[34] [35] Training exercises performed 1-3 times a week reduces pain and should be continued once pain has subsided and patients are able to return to their jobs and hobbies/activities and are able to cease the use of analgesics.[36]
Exercises are performed to reduce pain and to ensure stability by strengthening the hip extensors, hip flexors, abdominal muscles and the sacrospinalis muscles.[37]Other important exercises include engaging of the pelvis musculature to restore body symmetry, such as the back extensors and abdominal muscles. The Williams method suggests stretching of the back extensors and strengthening of the abdominal muscles to relieve some of the pressure placed on the lumbar intervertebral discs.[38] [39]
Sample examples of the Williams flexion exercises plan are:
- Pelvic tilt - lying on the back with bent knees, the back is pressed into the floor and held in this position for up to 10 seconds.
- Single or double Knee to chest - Lying on the back with bent knees, one is pulled up to the chest and held for up to 10 seconds. This can be done with one or two knees at a time.
- Partial sit-up - starting in a pelvic tilt position, the shoulders are then lifted off the floor. This is held for 2-10 seconds for each repetition
- Hamstring stretch
- Hip Flexor stretch
- Squat - performed correctly, this is a good overall exercise for the whole body and trunk control recruitment and lower back strength.
In combination with these exercises core stability exercises are recommended. Exercises to facilitate and engage the transversus abdominis are key to creating a stable base for performing other exercises, including strength exercises. [40]
Exercises can be progressed onto: curl ups, side plank, prone plank, bridging and performing alternated leg and arm raises in a four point kneeling position. This can be progressed to raising opposite arm and leg at the same time. During these exercises the spine should be maintained in a neutral position without any compensatory movements and the pelvis should not tilt. [40] Balance and coordination exercises relevant to an individual's sport can also be added into the programme.[40] [41]
In patients with degenerative disc disease it is also advised to add behavioural therapy to treatment programme due to the psychological effects of the disease, as patients can associate the constant pain and term degeneration with their backs becoming increasingly weak, This additional therapy has been shown to give better results.[7] These erroneous thoughts often elicit a fear of movement and can lead to the avoidance of movements of the spine. [32] Education and advice can also improve patient compliance,[42] helping them to overcome their fears and be adaptive with their coping strategies. Staying active and physical training will not have adverse effects on their spines[7] and performing low impact aerobic exercises is actually beneficial for the spine, increasing the flow of nutrients and blood to spinal structures and decreases pressure on the intervertebral discs. Prolonged single positions should also be avoided where possible. Back care is a life long process and patients should be encouraged to remain regularly active.[32][7]
Extension exercises are given to displace the pressure on the discs anteriorly. Some exercises are:
- Hollowing the back
- Making a sphinx posture
- Gym ball exercises
Physical therapies should aim to promote healing in the disc periphery, by stimulating cells, boosting metabolite transport and preventing adhesion and re-injury. Such an approach has the potential to accelerate pain relief in the disc periphery, even if it fails to reverse age-related degenerative changes in the nucleus.[43]
The use of low-level laser therapy is a further possible option in the conservative treatment of discogenic back pain, with positive clinical results of more than 90% efficacy, not only in the short-term, but also in the long-term, with lasting benefits. In their research, Ip and Fu conducted three sessions of treatment per week over a 12 week period. 49 of the 50 patients had significant improvement in their Oswestry Disability Index score [44], but this still needs more research.
Occupational Therapy[edit | edit source]
Occupational Therapy may be administered to teach body mechanics and postures. It can help patients gain proper manual handling skills and modify their regular tasks to avoid painful movement. [45]
Occupational therapy also can assist in locating essential equipment such as a pick-up stick, lumbar cushion, shower chair, swivel cushion for automobile transfer, and in some severe cases, a lifter and sling. [45]
Clinical Bottom Line[edit | edit source]
DDD is a is a condition in which the intervertebral discs lose height and hydration and as a result the discs are unable to fulfill their primary functions. Although the exact cause of DDD is unknown, it is thought to be primarily associated with the aging process. The most common location of this process is situated in the cervical and lower lumbar regions of the spine.
Diagnosis is via X-ray and to accurately identify the painful disc, a provocation discography test is used. Common outcome measures are the Oswestry Disability Index in combination with questionnaires like VAS and SF-36.
Multiple concepts of medical management are used to treat DDD. When pain persists despite efforts from conservative management, surgical techniques may be considered. A few examples of surgical management are:
- DIAM interspinous spacer[46][47]
- Disc arthroplasty
- Lumbar spinal fusion
- Total disc replacement
However, the preferred treatment protocol is conservative management, consisting of physical therapy and analgesic medications. The aim of physical therapy is to reduce pain, to enhance overall strength and core stability and to inform and advise the patient about the disease so that can then effectively self manage for the long term.
References[edit | edit source]
- ↑ Goel AP, Wang EJ, Bicket MC. Degenerative Disc Disease. In Spine Pain Care. Cham:Springer, 2020, p181-188.
- ↑ Medical News Today. All about degenerative disc disease. Available from: https://www.medicalnewstoday.com/articles/266630#diagnosis (Accessed 19 May 202)
- ↑ 3.0 3.1 3.2 3.3 3.4 Peng B. Pathophysiology, diagnosis, and treatment of discogenic low back pain. World J Orthop. 2013 April 18; 4(2): 42-52
- ↑ 4.0 4.1 McCormack BM, Weinstein PR. Cervical spondylosis. An update. West J Med 1996; 165(1-2): 43-51
- ↑ Zheng CJ, Chen J. Disc degeneration implies low back pain. Theor Biol Med Model. 2015 Nov 9;12:24. doi: 10.1186/s12976-015-0020-3.
- ↑ Spine-health, Degenerative Disc Disease Health Center. Available from: http://www.spine-health.com/conditions/degenerative-disc-disease (Accessed 18 May 2023)
- ↑ 7.0 7.1 7.2 7.3 7.4 Dutton M. Orthopaedic Examination, Evaluation, and Intervention. 2nd ed. New York, NY: McGraw-Hll; 2008.
- ↑ Ullrich, P. F. (2006 11 6). Lumbar Degenerative Disc Disease. Retrieved 06 02, 2009, from Degenerative Disc Disease: http://www.spine-health.com/conditions/degenerative-disc-disease/lumbar-degenerative-disc-disease
- ↑ 9.0 9.1 9.2 O'Halloran DM, Pandit AS. Tissue-engineering approach to regenerating the intervertebral disc. Tissue Eng 2007; 13(8):1927-1954.
- ↑ 7SecretsMedical. Degenerative Disc Disease. Available from: http://www.youtube.com/watch?v=UTBrVj7F6kI [accessed on 27/11/2023]
- ↑ 11.0 11.1 Thompson, J.C. MD. Netter's Concise Atlas of Orthopaedic Anatomy. (2002) Saunders Elsevier. p.36-7
- ↑ Bono CM, Lee CK. Critical analysis of trends in fusion for degenerative disc disease over the past 20 years: influence of technique on fusion rate and clinical outcome. Spine (Phila Pa 1976). 2004 Feb 15;29(4):455-63; discussion Z5. doi: 10.1097/01.brs.0000090825.94611.28.
- ↑ Kumar MN, Jacquot F, Hall H. Long-term follow-up of functional outcomes and radiographic changes at adjacent levels following lumbar spine fusion for degenerative disc disease. Eur Spine J. 2001 Aug;10(4):309-13. doi: 10.1007/s005860000207.
- ↑ Carreon LY, Glassman SD, Howard J. Fusion and nonsurgical treatment for symptomatic lumbar degenerative disease: a systematic review of Oswestry Disability Index and MOS Short Form-36 outcomes. Spine J. 2008 Sep-Oct;8(5):747-55. doi: 10.1016/j.spinee.2007.06.013.
- ↑ Kayhan F, Albayrak Gezer İ, Kayhan A, Kitiş S, Gölen M. Mood and anxiety disorders in patients with chronic low back and neck pain caused by disc herniation. Int J Psychiatry Clin Pract. 2016;20(1):19-23. doi: 10.3109/13651501.2015.1100314.
- ↑ Pfirrmann CW, Metzdorf A, Zanetti M, Hodler J, Boos N. Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine (Phila Pa 1976). 2001 Sep 1;26(17):1873-8. doi: 10.1097/00007632-200109010-00011.
- ↑ 17.0 17.1 Griffith JF, Wang YX, Antonio GE, Choi KC, Yu A, Ahuja AT, et al. Modified Pfirrmann grading system for lumbar intervertebral disc degeneration. Spine (Phila Pa 1976). 2007 Nov 15;32(24):E708-12. doi: 10.1097/BRS.0b013e31815a59a0.
- ↑ 18.0 18.1 18.2 Drazin D, Rosner J, Avalos P, Acosta F. Stem cell therapy for degenerative disc disease. Adv Orthop. 2012;2012:961052. doi: 10.1155/2012/961052.
- ↑ uwaterloo. Waterloo's Dr. Spine, Stuart McGill. Available from: http://www.youtube.com/watch?v=033ogPH6NNE [Accessed on 03/05/13]
- ↑ Phillips FM, Slosar PJ, Youssef JA, Andersson G, Papatheofanis F. Lumbar spine fusion for chronic low back pain due to degenerative disc disease: a systematic review. Spine (Phila Pa 1976). 2013 Apr 1;38(7):E409-22. doi: 10.1097/BRS.0b013e3182877f11.
- ↑ Taylor J, Pupin P, Delajoux S, Palmer S. Device for intervertebral assisted motion: technique and initial results. Neurosurg Focus. 2007 Jan 15;22(1):E6. doi: 10.3171/foc.2007.22.1.6.
- ↑ Garcia R Jr, Yue JJ, Blumenthal S, Coric D, Patel VV, Leary SP, et al. Lumbar Total Disc Replacement for Discogenic Low Back Pain: Two-year Outcomes of the activL Multicenter Randomized Controlled IDE Clinical Trial. Spine (Phila Pa 1976). 2015 Dec;40(24):1873-81. doi: 10.1097/BRS.0000000000001245.
- ↑ Beattie PF. Current understanding of lumbar intervertebral disc degeneration: a review with emphasis upon etiology, pathophysiology, and lumbar magnetic resonance imaging findings. J Orthop Sports Phys Ther. 2008 Jun;38(6):329-40. doi: 10.2519/jospt.2008.2768.(level of evidence 1b)
- ↑ 24.0 24.1 Zhang Y, An HS, Tannoury C, Thonar EJ, Freedman MK, Anderson DG. Biological treatment for degenerative disc disease: implications for the field of physical medicine and rehabilitation. Am J Phys Med Rehabil. 2008 Sep;87(9):694-702. doi: 10.1097/PHM.0b013e31817c1945.
- ↑ Mirza SK, Deyo RA. Systematic review of randomized trials comparing lumbar fusion surgery to nonoperative care for treatment of chronic back pain. Spine (Phila Pa 1976). 2007 Apr 1;32(7):816-23. doi: 10.1097/01.brs.0000259225.37454.38.
- ↑ Rohof O. Intradiscal pulsed radiofrequency application following provocative discography for the management of degenerative disc disease and concordant pain: a pilot study. Pain Pract. 2012 Jun;12(5):342-9. doi: 10.1111/j.1533-2500.2011.00512.x.
- ↑ Slade SC, Keating JL. Unloaded movement facilitation exercise compared to no exercise or alternative therapy on outcomes for people with nonspecific chronic low back pain: a systematic review. J Manipulative Physiol Ther. 2007 May;30(4):301-11. doi: 10.1016/j.jmpt.2007.03.010.
- ↑ Triano JJ, McGregor, M, Skogsbergh DR. Use of chiropractic manipulation in lumbar rehabilitation. Journal of rehabilitation research and development. 1997; 34: 394-404.
- ↑ Vieira-Pellenz F, Oliva-Pascual-Vaca A, Rodriguez-Blanco C, Heredia-Rizo AM, Ricard F, Almazán-Campos G. Short-term effect of spinal manipulation on pain perception, spinal mobility, and full height recovery in male subjects with degenerative disk disease: a randomized controlled trial. Arch Phys Med Rehabil. 2014 Sep;95(9):1613-9. doi: 10.1016/j.apmr.2014.05.002.
- ↑ Paige NM, Miake-Lye IM, Booth MS, Beroes JM, Mardian AS, Dougherty P, et al. Association of Spinal Manipulative Therapy With Clinical Benefit and Harm for Acute Low Back Pain: Systematic Review and Meta-analysis. JAMA. 2017 Apr 11;317(14):1451-1460. doi: 10.1001/jama.2017.3086.
- ↑ Brox JI, Sorensen R, Friis A. Randomized Clinical Trial of Lumbar Instrumented Fusion and Cognitive Intervention and Exercises in Patients with Chronic Low Back Pain and Disc Degeneration. Spine 2003; 28(17):1913-1921
- ↑ 32.0 32.1 32.2 Beattie P. Current understanding of lumbar intervertebral disc degeneration: a review with emphasis upon etiology, pathophysiology, and lumbar magnetic resonance imaging findings. 2008; 38(6):329-340
- ↑ Adam Gąsiorowski et al, Strength training in the treatment of degeneration of lumbar section of vertebral column, Annals of Agricultural and Environmental Medicine, Vol 20, No 2, 203–205, 2013
- ↑ Zagra A., et al; Prospective study of a new dynamic stabilisation system in the treatment of degenerative discopathy and instability of the lumbar spine, Eur Spine J, 2012, 21, suppl 1: S83-S89
- ↑ Canbay S., et al, Posterior dynamic stabilization for the treatment of patients with lumbar degenerative disc disease: long-term clinical and radiological results, Turkish Neurosurgery 2013, vol. 23, No. 2, 188-197
- ↑ 41. Adam Gąsiorowski et al, Strength training in the treatment of degeneration of lumbar section of vertebral column, Annals of Agricultural and Environmental Medicine, Vol 20, No 2, 203–205, 2013
- ↑ Johnson Olubusola E. Therapeutic Exercises in the Management of Non-Specific Low Back Pain, Low Back Pain, ISBN: 978-953-51-0599-2, 2012
- ↑ Lee M.,The effects of core muscle release technique on lumbar spine deformation and low back pain.J Phys Ther Sci.;27(5):1519-22, 2015
- ↑ Mircea MOLDOVAN, Therapeutic Considerations and Recovery in Low Back Pain: Williams vs McKenzie, Timisoara Physical Education and Rehabilitation Journal. Volume 5, Issue 9, 2012
- ↑ 40.0 40.1 40.2 AKUTHOTA V., Core Stability Exercise Principles Akuthota, Curr. Sports Med. Rep., Vol. 7, No. 1, pp. 39Y44, 2008
- ↑ COSIO-LIMA, Effects of Physioball and Conventional Floor Exercises on Early Phase Adaptations in Back and Abdominal Core Stability and Balance in Women. J Strength Cond Res.17(4):721-5. 2003
- ↑ Mirza SK, Deyo RA. Systematic Review of Randomized Trials Comparing Lumbar Fusion Surgery to Nonoperative Care for Treatment of Chronic Back Pain. Spine 2007; 32(7): 816-823
- ↑ Adams MA, Stefanakis M, Dolan P. Healing of a painful intervertebral discfckLRshould not be confused with reversing disc degeneration: implications forfckLRphysical therapies for discogenic back pain. Clin Biomech (Bristol, Avon). 2010fckLRDec;25(10):961-71.
- ↑ Ip D, Fu NY. Can intractable discogenic back pain be managed by low-level laser therapy without recourse to operative intervention? J Pain Res. 2015 May 26;8:253-6
- ↑ 45.0 45.1 australiaot. Occupational Therapy Proforma - Format & Example (Lumbar Disc Bulge) 디스크 [Internet]. Australia OT. 2021 [cited 2023 Aug 10]. Available from: https://australiaot.com/en/occupational-therapy-proforma-format-example-lumbar-disc-bulge-디스크/
- ↑ Hrabalek L, Machac J, Vaverka M. The DIAM spinal stabilisation system to treat degenerative disease of the lumbosacral spine. Acta chirurgiae orthopaedicae et traumatologiae Cechoslovaca. 2009 Oct;76(5):417-23.
- ↑ Bruneau A, Anderson KM, Lim R, Lange EC, Carls T, Trieu HH, inventors; Warsaw Orthopedic Inc, assignee. Interspinous process brace. United States patent US 8,105,357. 2012 Jan 31.