Cushing's Syndrome
Original Editors - Jessica Stevenson from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Jessica Stevenson, Admin, Lucinda hampton, Aminat Abolade, Elaine Lonnemann, Laura Ritchie, Dave Pariser, Tony Lowe, Wendy Walker, Kirenga Bamurange Liliane, 127.0.0.1 and Kim Jackson
Introduction[edit | edit source]
Cushing’s syndrome is a general term for increased secretion of cortisol by the adrenal cortex.
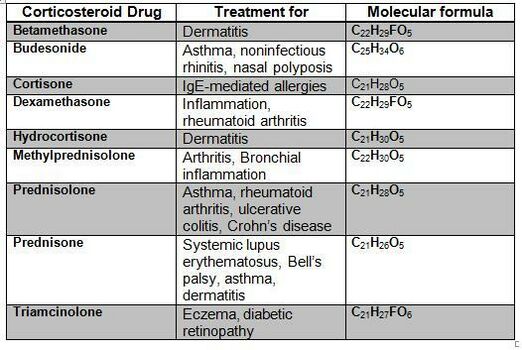
- When corticosteroids are administered externally, a condition of hypercortisolism called iatrogenic Cushing’s syndrome occurs.
- When the hypercortisolism results from an oversecretion of Adrenocorticotropic hormone (ACTH) from the pituitary, the condition is called Cushing’s disease.
The clinical presentation is the same for all of these conditions.[1] [2]
Epidemiology[edit | edit source]
The actual incidence and prevalence of Cushing syndrome are not known. The prevalence of the disease is highly variable across different ethnic and cultural groups depending upon the frequency and spectrum of the medical conditions requiring steroid-based therapy.
- Of the known cases, iatrogenic hypercortisolism outweighs the endogenous causes,
- Of the endogenous causes, pituitary-mediated ACTH production accounts for up to 80% of cases of hypercortisolism, followed by adrenals, unknown source, and ectopic ACTH production secondary to malignancies[3]
Etiology[edit | edit source]
There are two main etiologies of Cushing syndrome:
- Endogenous hypercortisolism: results from excessive production of cortisol by adrenal glands and can be ACTH-dependent and ACTH-independent. ACTH-secreting pituitary adenomas (Cushing disease) and ectopic ACTH secretion by neoplasms are responsible for ACTH-dependent Cushing. Adrenal hyperplasia, adenoma, and carcinoma are major causes of ACTH-independent Cushing syndrome.
- Exogenous hypercortisolism: the most common cause of Cushing syndrome, mostly iatrogenic and results from the prolonged use of glucocorticoids[3].
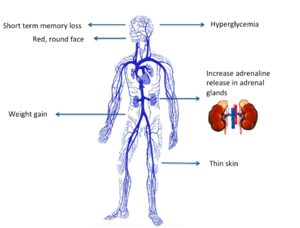
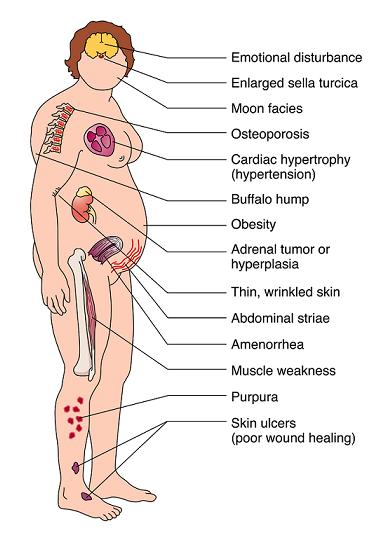
Characteristics/Clinical Presentation[edit | edit source]
Clinical manifestations include:
- “moon” face (very round),
- buffalo hump (truncal obesity with prominent supraclavicular and dorsal cervical fat pads) [4],
- protuberant abdomen with accumulation of fatty tissue and stretch marks with purple striae,
- muscle wasting and weakness,
- thin extremities,
- decreased bone density (especially spine),
- kyphosis and back pain (secondary to bone loss),
- easy bruising and poor wound healing due to thin and atrophic skin [4],
- acne,
- psychiatric or emotional disturbances,
- impaired reproductive function (decreased libido and changes in menstrual cycle, and
- diabetes mellitus.
In women, masculinizing effects such as hypertrichosis, breast atrophy, voice changes, and other signs of virilism are noted. Cessation of linear growth is characteristic in children. [1] [4]
Complications[edit | edit source]
Complications include:
- Excess hair growth
- Osteoporosis
- Susceptibility to infections
- Type 2 diabetes
- Peptic ulcer disease
- Hypertension[3]
Diagnosis[edit | edit source]
Taking glucocorticoid medications is the most common cause of Cushing syndrome. If this is the case other tests are not needed. Cushing syndrome from endogenous cortisol production can be difficult to diagnose because other conditions have similar signs and symptoms. Diagnosing Cushing syndrome can be a long and extensive process. The below tests can help find the cause:
- Urine and blood tests: measure hormone levels and show whether person is producing excessive cortisol. The doctor might also recommend other specialized tests that involve measuring cortisol levels before and after using hormone medications to stimulate or suppress cortisol.
- Saliva test. Cortisol levels normally rise and fall throughout the day. In people without Cushing syndrome, levels of cortisol drop significantly in the evening. By analyzing cortisol levels from a small sample of saliva collected late at night, doctors can see if cortisol levels are too high.
- Imaging tests. CT or MRI scans can provide images of the pituitary and adrenal glands to detect abnormalities, eg tumors.
- Petrosal sinus sampling. This test can help determine whether the cause of Cushing syndrome is rooted in the pituitary or somewhere else. For the test, blood samples are taken from the veins that drain the pituitary gland (petrosal sinuses).
Management[edit | edit source]
Treatments for Cushing syndrome are designed to lower the high level of cortisol in the body. The best treatment for you depends on the cause of the syndrome. Options include:
- The best therapy for iatrogenic Cushing syndrome is to taper exogenous steroids. Chronic exposure to steroids potentially suppresses the function of the adrenal and it can take several months for normal adrenal functioning to recover. Steroids should be slowly tapered allowing adrenal functioning to recover.
- Hypercortisolism due to Cushing disease or adrenal tumor or ectopic tumor is treated with surgical resection. After the operation, cortisol replacement medications are needed to provide the body with the correct amount of cortisol. In most cases eventually normal adrenal hormone production returns. If the surgeon can not totally remove a pituitary tumor, radiation therapy as well as surgery will be needed. Additionally, radiation may be used for people who aren't suitable candidates for surgery[3].[5]
- Medications can be used to control cortisol production when surgery and radiation don't work. Medical therapy may not completely improve all of the symptoms of excess cortisol. Medications to control excessive production of cortisol at the adrenal gland include ketoconazole, mitotane (Lysodren) and metyrapone (Metopirone).
In the management of Cushing syndrome, it is crucial to treat comorbidities such as diabetes mellitus, hypertension, osteoporosis, psychiatric issues, and electrolyte disorders.[3]
Physical Therapy Management[edit | edit source]
Therapists are more likely to treat people who have developed medication-induced Cushing’s syndrome. Exogenous steroids are administered for a number of inflammatory and other disorders eg asthma or rheumatoid arthritis. [1]
Educate Clients with regard to:
- Getting regular exercise to help maintain muscles and bone mass and prevent weight gain. To maintain muscle and bone mass, weight-bearing exercises eg push-ups, sit-ups, or lifting weights are helpful. To prevent weight gain, aerobic exercise are good. eg fast walking, jogging, cycling, and swimming. [6]
- Education on avoiding falls and removing loose rugs and other hazards in the home. Falling may lead to broken bones and other injuries. [6]
- Pay close attention to all wounds. Too much cortisol slows wound healing. Education on proper wound healing and cleansing is important. Clean all wounds immediately with antibacterial soap and use antibiotic ointment and dressings to prevent infection. [6]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Goodman CC, Snyder KS. Differential Diagnosis for Physical Therapists: Screening for Referral. Philadelphia : W.B. Saunders Company; 2006: 473-475
- ↑ Goodman CC, Fuller KS. Pathology: Implications for the Physical Therapist 3rd ed. St. Louis: Saunders Elsevier; 2009: 481-483.
- ↑ 3.0 3.1 3.2 3.3 3.4 Chaudhry HS, Singh G. Cushing syndrome. InStatPearls [Internet] 2021 Jul 30. StatPearls Publishing. Available:https://www.ncbi.nlm.nih.gov/books/NBK470218/ (accessed 12.5.2022)
- ↑ 4.0 4.1 4.2 Beers MH, Porter RS, Jones TV, Kaplan JL, Berkwits M. The Merck Manual of Diagnosis and Therapy 18th ed. Whitehouse Station:Merck Research Laboratories; 2006: 1212-1214.
- ↑ Mayo clinic Cushing Syndrome Available:https://www.mayoclinic.org/diseases-conditions/cushing-syndrome/diagnosis-treatment/drc-20351314 (accessed 13.5.2022)
- ↑ 6.0 6.1 6.2 Caroline Rea. Cushing's Syndrome. YahooHealth. last updated 4/29/2008. http://health.yahoo.com/hormone-living/cushing-s-syndrome-home-treatment/healthwise--hw71687.html