Pathophysiology of Traumatic Brain Injury
Original Editor - Anna Ziemer
Top Contributors - Naomi O'Reilly, Kim Jackson, Lucinda hampton, Rachael Lowe, Tony Lowe, Simisola Ajeyalemi and Stacy Schiurring
Introduction[edit | edit source]
A traumatic brain injury (TBI) causes brain damage via primary and or secondary injury mechanisms.[1]
"Primary injury causes local brain damage, while secondary damage begins with inflammatory activity followed by disruption of the blood–brain barrier (BBB), peripheral blood cells infiltration, brain edema, and the discharge of numerous immune mediators including chemotactic factors and interleukins." [1]
Primary traumatic brain injury insult triggers complex cellular and molecular processes leading to further neuronal dysfunction and death (secondary injury). The variety of processes involved contributes to the traumatic brain injury complexity but also creates various therapeutic targets.[2] Multiple factors contribute to those pathophysiological mechanisms of secondary injury and their contribution to the severity of the secondary injury might vary.
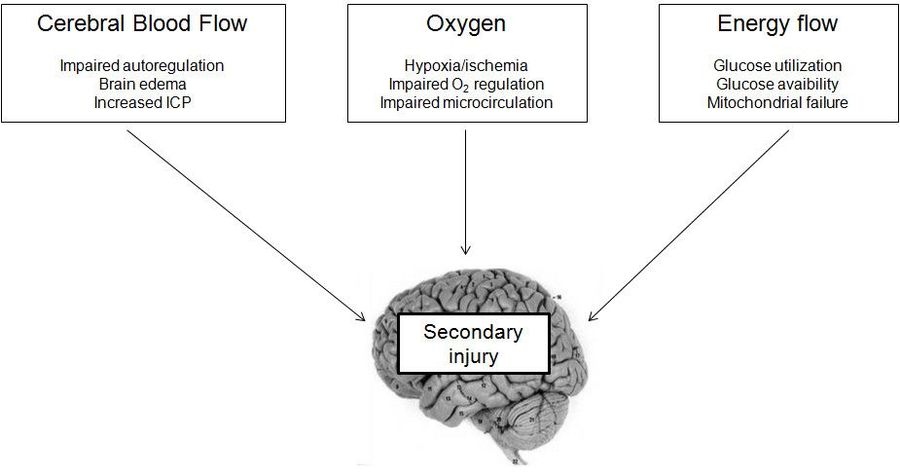
General pathophysiological features of traumatic brain injury and mechanism following primary onset might include:
- Diffuse axonal injury
- Degradation of the cytoskeleton
- Cortical and subcortical neuronal injury/death
- Vascular-related changes (barrier breakdown, vasospasm, oedema)
- Haemorrhage
- Ischemia
- Glutamate excitotoxicity
- Changes in neurotransmitters
- Seizures
- Physiological disturbances
- Free radical generation
- Disruption of calcium homeostasis
- Mitochondrial disturbances
- Metabolic disturbances
- Altered brain metabolism
- Altered gene expression
- Pro-inflammatory state
These pathophysiological events impair cell function impacting movement, memory and learning ability as well as potentially causing damage to white matter structure. They are described in more detail below.
Vascular Autoregulation[edit | edit source]
The normal brain vascular autoregulation includes a pressure and volume monitoring mechanism allowing continuous cerebral blood flow (CBF) and optimal oxygen supply. The main mechanism involved in maintaining consistent cerebral pressure in response to changing systemic arterial pressure are vasoconstriction and vasodilatation of brain vessels. Traumatic brain injury impairs or even abolishes cerebrovascular autoregulation immediately after the trauma or over time.
Multiple factors can initiate these vasodilation or vasoconstriction cascades, including; [5]
- Systemic arterial pressure
- Systemic blood volume
- Blood viscosity
- Oxygen level delivery
- Metabolism
- Hypo / hypercapnia
- Pharmacologic agents [5]
Autoregulatory vasoconstriction is more problematic than autoregulatory vasodilation and leads to greater brain tissue sensitivity to decreased cerebral perfusion pressure.
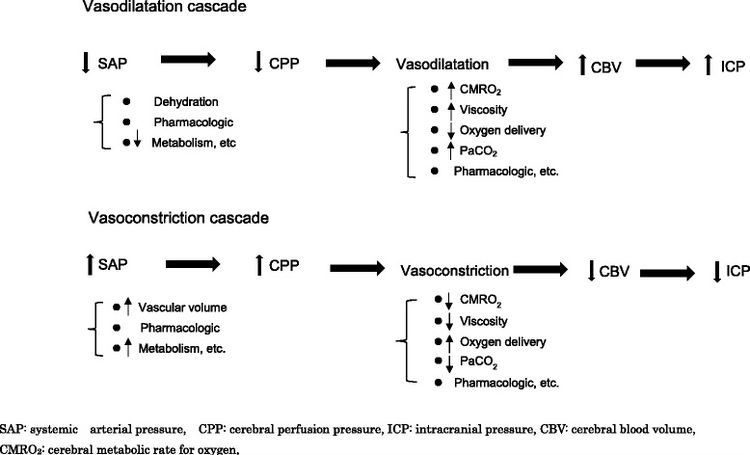
Following an initial insult, an ischemia like stage of traumatic brain injury triggers a cascade of processes characterised by direct brain tissue damage and cerebral blood flow (CBF) regulation impairment as well as metabolism impairment. This cascade might result in oedema formation, increase of intracranial pressure (ICP), and decreased cerebral perfusion pressure (CPP).
Cerebral Perfusion Pressure [edit | edit source]
Cerebral perfusion pressure is the difference between the systemic arterial pressure and the intracranial pressure. The normal range of this difference is between 50 and 70 mmHg.
The following clinical mechanisms contribute to the dysregulation of the mechanisms that usually maintain volume and pressure:
- Depolarisation and disturbance of ionic homeostasis
- Neurotransmitter release (e.g. glutamate excitotoxicity)
- Mitochondrial dysfunction
- Neuronal apoptosis
- Lipid degradation
- Initiation of inflammatory and immune responses
Cerebral blood flow (CBF) disruption can also be caused by mechanical displacement of brain structures, stretching and distorting brain vessels, arterial hypotension, vasospasm, changes in cerebral microvasculature.
Both hypoperfusion and hyperperfusion is related to the difference between cerebral blood flow and cerebral metabolism and oxygen consumption. Therefore, decreased CBF with a normal metabolic rate creates ischemic conditions.
Ischemia[edit | edit source]
Cerebral ischemia is a state of decreased blood supply of the brain (hypoperfusion) and leads primarily to metabolic stress and ionic perturbations.[7] Coexisting traumatic damage such as structural injury of cell bodies, astrocytes and microglia, cerebral vascular and endothelial damage intensify the brain tissue damage. The factors involved in post-traumatic vasospasm and contributing to resultant ischaemia include:
- Morphological damage due to mechanical displacement, i.e.vessel distortion
- Hypotension
- Depletion of nitric oxide and or cholinergic neurotransmitters
- Vascular smooth muscle depolarisation related to potassium channel reduced activity
- Potentiation of prostaglandin induced vasoconstriction
- Free radicals formation
TBI frequently leads to focal or global cerebral ischemia and its presence points towards poor clinical outcome like persistent vegetative state or death.
Oedema[edit | edit source]
The mechanism responsible for oedema formation and intracranial pressure increase is hyperaemia. Due to reduced proximal cerebrovascular resistance and increased cerebral blood volume and vessel dilatation the impaired brain blood barrier causes an excess of fluids to reside in vascular bed. This pathology is equally detrimental to brain tissue as ischemia causing an increase in intracranial pressure. Oedema is a common result of traumatic brain injury and can be vasogenic or cytotoxic and can cause ICP increase and secondary ischemia.
- Vasogenic brain oedema is caused by endothelial cells damage. The ion and protein flow through vascular walls to interstitial space causing an increased volume in extracellular space.
- Cytotoxic oedema results from intracellular water accumulation related to increased cell membrane permeability.
Cerebral Metabolic Dysfunction[edit | edit source]
Cerebral metabolic disfunction relates to oxygen and glucose depletion as well as reduced cerebral energy state at the period of post traumatic hypermetabolic demand. Hypermetabolism is pathophysiological phenomenon following TBI and occurs as a result of transmembrane ionic influx leading to overexcitation and uncoupling with cerebral blood flow. The extent of primary injury is reflected by the extent of pathophysiological processes like mitochondrial dysfunction related to decreased respiratory rates and ATP production, depletion of the nicotinic co-enzyme pool, intramitochondrial accumulation of calcium ions leading to metabolic failure.[7] The overload of excitatory amino acid neurotransmitters results in overstimulation of ionotropic and metabotropic glutamate receptors with consecutive calcium, sodium and potassium ions flow triggering brain blood barrier breakdown and cellular compensatory ATPase activity increase resulting in aggravated metabolic demand. The cascade of mismatched processes of overflow and metabolism creates excitotoxicity. TBI metabolic failure is also related to imbalance between oxygen supply and oxygen consumption and leads to hypoxia. The extent of the hypoxia and its duration determines the clinical outcome. Therefore, measuring brain oxygenation is one of the standard measurements along with ICP and CPP. The oxidative stress related to imbalance of free radicals and endogenous antioxidants availability can lead to immediate cell death or inflammatory processes or apoptosis.
Neuroinflammation[edit | edit source]
Damage to blood vessel endothelium following TBI triggers a neuroinflammation process with a release of cytokines, free radicals, prostaglandins and complements mobilising an active response from immune system to eliminate the damaged cells and format scar tissue. Proinflammatory enzymes like interleukin-1, interleukin -6 intensify the activity within the first hours from initial insult. The leucocytosis increasing cell debris and phagocytosis response may further increase the inflammatory response and tissue destruction affecting undamaged tissue extending the injury and decomposing dead tissue.
Cell Death[edit | edit source]

Cell death and axonal injuries contribute to the extent of the traumatic brain injury. The main death processes are known as necrosis and apoptosis. Apoptotic and necrotic neurons are present even in mild injuries and can be found in areas distant from the injury impact area. Degenerating oligodendrocytes and astrocytes are present in the white matter of primary injury area.
Necrosis (cell death) occurs after the first few hours following an insult to brain tissue, mechanical or hypoxic, and is related to cell membrane damage and uncontrolled release of cell death products. The blood brain barrier becomes impaired and white matter injury usually increases. The resulting detritus is interpreted as an ‘antigen’ and triggers inflammatory process and scaring.
Apoptosis is a naturally programmed cell death and affects undamaged neurons. The imbalance of pro and anti-apoptotic proteins triggers the cell death mechanism hours post primary insult. Apoptosis is triggered by cell surface receptor engagement, growth factor withdrawal and DNA damage.
Several groups of proteins and biochemical transitional pathways are involved in cell death mechanisms and their tracking might create new therapeutic opportunities limiting neurodegeneration and resulting disabilities especially with apoptosis providing the window of opportunity for therapy due to its delayed nature.
Axonal Damage[edit | edit source]
Morphologically, axons with their long structure are at significant mechanical risk during the impact of external forces. Sheering or stretching of axons results in primary axotomy or when damage incomplete they trigger secondary axon degeneration. There are some localities of the brain with greater vulnerability such as the junction of grey and white matter. Myelination might provide some protective features and enhance recovery. The pathophysiological mechanisms involved in axonal damage also include swelling of intact axons and “retraction bulbs”. [2]
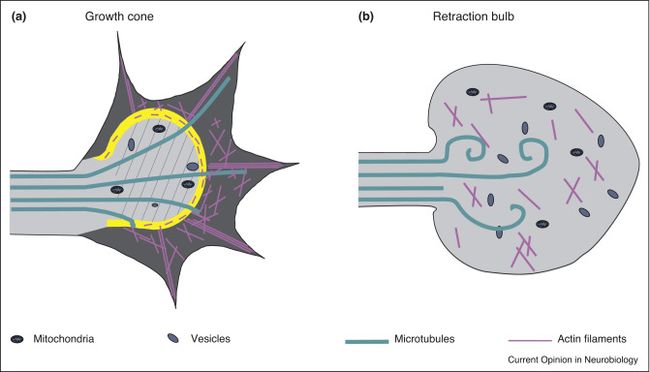
Axonal damage due to deafferentation interrupts established pathways and can cause focal and diffused injury immediately after or even after several years from the primary insult. The extent of deafferentation in mild to severe injuries and axonal damage impacts the ability of synaptic sprouting of undamaged axons. The more severe the injury with extensive secondary damage, the less possible axonal reconnection and function recovery.
Diffuse Axonal Injury[edit | edit source]
The primary insult of axonal injury leads to disconnection and/or neuron connections malfunction resulting in functional areas impairment. Mechanically disrupted axons present cytoskeletons malfunction resulting in proteolysis, swelling, and other microscopic and molecular changes to the neuronal structure. Neurological presentation of Diffuse Axonal Injury includes bilateral neurological examination deficits frequently affecting the frontal and temporal white matter, corpus callosum, and brainstem.
The Adams Diffuse Axonal Injury Classification:
Grade 1:[edit | edit source]
A mild diffuse axonal injury with microscopic white matter changes in the cerebral cortex, corpus callosum, and brainstem
Grade 2[edit | edit source]
A moderate diffuse axonal injury with gross focal lesions in the corpus callosum
Grade 3[edit | edit source]
A severe diffuse axonal injury with finding as Grade 2 and additional focal lesions in the brainstem.
The Diffuse Axonal Injury is a severe form of brain injury and is usually diagnosed after a traumatic brain injury with Glasgow Coma Scale (GCS) < 8 for more than six consecutive hours. Diffuse Axonal Injury features in Computed Tomography (CT) and present as small punctate haemorrhages to white matter. However, CT Head has a low yield in detecting Diffuse Axonal Injuries and Magnetic Resonance Imaging (MRI), specifically Diffuse Tensor Imaging (DTI), is the imaging modality of choice for diagnosis of diffuse axonal injury.
References[edit | edit source]
- ↑ 1.0 1.1 Thapa K, Khan H, Singh TG, Kaur A. Traumatic brain injury: mechanistic insight on pathophysiology and potential therapeutic targets. Journal of Molecular Neuroscience. 2021 Sep;71(9):1725-42.
- ↑ 2.0 2.1 Hill CS, Coleman MP, Menon DK. Traumatic Axonal Injury: Mechanisms and Translational Opportunities. Trends in Neuroscience. 2016. 39(5): 311-324 doi.org/10.1016/j.tins.2016.03.002
- ↑ Bouzat P, Sla N, Payen JF, Oddo M. Beyond intracranial pressure: optimization of cerebral blood flow, oxygen, and substrate delivery after traumatic brain injury. Annals of Intensive Care. 2013.3:23https://doi.org/10.1186/2110-5820-3-23
- ↑ OPENPediatrics. "Introduction to Traumatic Brain Injury" by Lisa DelSignore, MD for OPENPediatrics. Available from: https://youtu.be/2JNTJKaz-aM[last accessed 30/08/19]
- ↑ 5.0 5.1 Kinoshita K. Traumatic brain injury: pathophysiology for neurocritical care. Journal of Intensive Care. 2016. 4:29-39. DOI 10.1186/s40560-016-0138-3
- ↑ Kinoshita K. Traumatic brain injury: pathophysiology for neurocritical care. Journal of Intensive Care. 2016. 4:29-39. DOI 10.1186/s40560-016-0138-3
- ↑ 7.0 7.1 Werner C., Engelhard K. Pathophysiology of traumatic brain injury. British Journal of Anaesthesia. 2007 (1): 4–9 doi:10.1093/bja/aem131
- ↑ Kinoshita K. Traumatic brain injury: pathophysiology for neurocritical care. Journal of Intensive Care. 2016. 4:29-39. DOI 10.1186/s40560-016-0138-3
- ↑ Kinoshita K. Traumatic brain injury: pathophysiology for neurocritical care. Journal of Intensive Care. 2016. 4:29-39. DOI 10.1186/s40560-016-0138-3
- ↑ Brainline. Understanding Diffuse Axonal Injury. Available from: https://youtu.be/k_MktbTuEdI[last accessed 30/08/19]
- ↑ Brainline. What is Diffuse Axonal Brain Injury?. Available from: https://youtu.be/cw-T99xFwoI[last accessed 30/08/19]






