Endometriosis
Original Editors - Rebecca Clark from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Rebecca Clark, Admin, Kim Jackson, Lucinda hampton, Aya Alhindi, Ilona Malkauskaite, Nicole Hills, Eugenie Lamprecht, Franca Ebomah, Rishika Babburu, 127.0.0.1, Elaine Lonnemann, Laura Ritchie, Wendy Walker and WikiSysop
Introduction[edit | edit source]
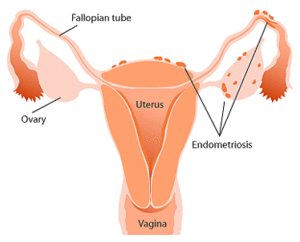
Endometriosis is a chronic gynecologic disease characterized by the development and presence of histological elements like endometrial glands and stroma in anatomical positions and organs outside of the uterine cavity.
The main clinical manifestations of the disease are chronic pelvic pain and impaired fertility.
The localization of endometriosis lesions can vary, with the most commonly involved focus of the disease the ovaries followed by the posterior broad ligament, the anterior cul-de-sac, the posterior cul-de-sac, and the uterosacral ligament.
Endometriotic nodules also affect the intestinal tract and the urinary system like the ureter, the bladder, and the urethra. Nevertheless, endometriosis is not limited to the pelvis but can damage extra pelvic structures like the pleura, the pericardium, or the central nervous system[1].
Epidemiology[edit | edit source]
The exact prevalence cannot be easily defined since the definitive diagnosis of the disease (a laparoscopic examination) needs to be conducted.
- It is estimated that endometriosis affects approximately 10% to 15% of women of reproductive age, whereas this prevalence increases by up to 70% in women with chronic pelvic pain.
- Usually it takes up to 8 to 10 years to reach the diagnosis of this disease .
- In the U.S., according to a more recent survey of the National Hospital Discharge Survey, 11.2% of all women between 18 and 45 years old hospitalized for genitourinary causes were diagnosed with endometriosis, and approximately 10.3% of the women undergone gynecologic surgeries have endometriosis.
- Endometriosis is a disease with a high burden in Europe and the U.S. It has a considerable impact on worldwide economics as well—it costs the world over 80 billion USD per year. [2]
- There are limited epidemiologic data on the prevalence of endometriosis in sub-Saharan Africa. In Nigeria, a prevalence of 4-8% has been reported among women.[3][4]In South Africa, a prevalence of 2-6% has been reported.[5]
- This low prevalence among African women have been attributed to: early age at first pregnancy, short inter pregnancy intervals with large size families, taboos around menstruation and pain, increased risk of pelvic inflammatory disease and blocked fallopian tube[6][7]Some others have further attributed this low prevalence to: low awareness of the disease in sub-Saharan Africa, the poor access to diagnostic and therapeutic facilities and the limited training available for the management of endometriosis in the region[8]
Theories on the Pathogenesis of Endometriosis[edit | edit source]
The exact cause of endometriosis is unknown, yet several theories have been developed that explain the existence of displaced endometrial tissue:
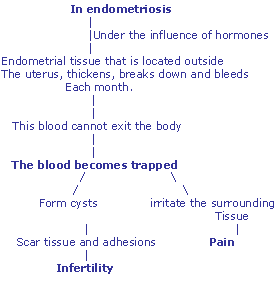
Retrograde Menstruation
It is also known as Sampson's theory. It has remained relevant since it was first described in 1925.
The basic idea is that menstrual blood containing endometrial cells regurgitates into the peritoneal cavity via patent fallopian tubes, where implantation of these cells may occur.[9][10]
Angiogenesis promotes the development and growth of the lesion after implantation. Which makes it possible because activated peritoneal macrophages produce angiogenic factors such as vascular endothelial growth factor (VEGF).
The problem with this theory is that it only explains ovarian and superficial peritoneal endometriosis, not deep infiltrating endometriosis or lesions outside the peritoneum.[11][12][9][10]
Benign Metastasis
This theory holds that during menstruation, a small amount of endometrial tissue can be disseminated through the uterine-draining lymph vessels. This theory is based on the discovery of an endometrial polyp which had protruded into the lumen of a lymph vessel . Because lymphatic capillaries are found in almost all organs, a benign metastasis theory can explain the occurrence of endometriosis in lymphatic nodes and distant locations such as the lungs.
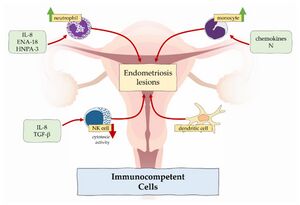
Immune Dysregulation
Inflammation, caused by immune dysregulation, is one of the primary mechanisms involved in diseases characterised by cell proliferation and infiltration. In the case of endometriosis, proinflammatory pathways prevent apoptotic mechanisms from working, and potentially harmful cells adhere to distant sites. Macrophages, neutrophils, NK cells, dendritic cells, and T cells are immune cells that play a role in the formation and progression of endometrial lesions.[13]
Coelomic Metaplasia
In 1924, Robert Meyer proposed the theory of coelomic metaplasia. It is based on the development of the female reproductive tract: it arises from a pair of Müllerian ducts, which arise from coelomic epithelial cells of mesodermal origin . According to this theory, the original coelomic membrane metaplasias and forms endometrial stroma and glands. It is the most likely explanation for cases of endometriosis in men who have received high doses of oestrogen for prostatic carcinoma treatment, as well as Rokitansky-Kuster-Hauser syndrome patients who do not have functioning endometrial tissue due to uterine and vaginal aplasia .
Ovarian endometrioma is the most common type of endometriosis that could be explained by this theory. The mesothelium, which develops from the ovarian coelomic epithelium, has high metaplastic potential and can invaginate into the ovarian cortex . Metaplasia could transform these mesothelial inclusions into endometriosis. The factors influencing this phenomenon are still unknown.[14]
Clinical Presentation[edit | edit source]
Clinical presentation of endometriosis varies in women. The peak of the disease incidence is greater in women between 25 and 29 years old and lowest in women over 44 years old, whereas the prevalence in whites is higher than the African-Americans.
Clinicians usually suspect and are more likely to diagnose the disease in females presenting with the typical symptomatology such as: [1][15][16]
- intermenstrual bleeding
- dysmenorrhea- commonly identified as the chief complaint if implants are located over the uterosacral ligaments
- dyspareunia (painful intercourse) - local adhesions may be irritated by penile penetration
- dyschezia or painful defecation - adhesions may be present over the large bowel. As faecal matter moves through the intestines these adhesions can be stretched causing local irritation.
- dysuria ( painful or difficult urination)
- Abdominal pain, fatigue and mood change beginning 1-2 days before menstruation and continuing for the duration
- Constant/intermittent, or cyclical pelvic and/or low back pain (unilateral or bilateral)
- Infertility - often first diagnosed in women who are seeking treatment for infertility
- History of ectopic pregnancy or miscarriage
- Low-grade fever
- Diarrhoea, constipation, rectal bleeding
- Referred pain to the low back/sacral groin, posterior leg, upper abdomen, or lower abdominal.suprapubic areas
- Menorrhagia/menometrorrhagia - excessive or occasional heavy periods may be experienced, along with bleeding between periods
Less Common Signs and Symptoms[17]
- Chest pain/hemoptysis - due to endometrial implants in the lungs
- Headache/seizures - due to endometrial implants in the brain
Associated Co-morbidities[edit | edit source]
- Several health problems have been shown to exist in combination with endometriosis.
- A large percentage of women experience co-morbidities such as[18][19]:
- Fibromyalgia
- Hypothyroidism
- Chronic fatigue syndrome
- Allergies
- Asthma
- Rheumatoid arthritis
- Multiple sclerosis
- Systemic lupus erythematosus
- Auto-immune disorders
Associated Risk Factors[edit | edit source]
The following factors may place you at greater risk for developing endometriosis:[16][20][19]
- Early-onset of menstruation
- Never giving birth or postponed pregnancy
- Family history of endometriosis
- Frequent menstrual cycles with a duration greater than eight days
- Medical conditions which block the flow of menstruation (such as a closed hymen)
- Low body mass index (BMI)
- Sedentary lifestyle
Diagnostic Tests/Lab Tests/Lab Values[edit | edit source]
The following are common tests used to identify physical clues leading to the diagnosis of endometriosis:
- Laparoscopy: Accurate diagnosis of endometriosis requires direct visualization of endometrial tissue through laparoscopic procedures.[15]This is accomplished by distending the abdomen through the injection of carbon dioxide for adequate visualization of the reproductive organs. An instrument with a camera (laparoscope) is inserted through a tiny incision near the navel in order to determine if reproductive and abdominal organs possess endometrial implants. Laparoscopy is a valuable tool guiding the course of treatment for endometriosis by identifying the severity, size and location of foreign implants.[16]
- Pelvic Exam: Manual palpation of the pelvic region to identify abnormalities such as cysts or scar tissue surrounding reproductive organs. Although this exam may reproduce pain in large areas of implantation, it is often not possible to feel small areas, making this test less reliable at identifying the presence of endometriosis.[16]
- Ultrasound: Ultrasounds work by using sound waves from a transducer wand to create a video image of organs. Vaginal ultrasounds are performed by inserting a wand into the vagina to view images of the reproductive organs, while abdominal ultrasounds are done by moving a wand over the abdominal-pelvic region. Ultrasound is done in order to identify the presence of cysts associated with endometriosis, but it is important to note that this is not a definitive test used to diagnose endometriosis.[16]
- MRI: An MRI is used to view endometrial implants but proves to be more sensitive in comparison to the use of ultrasound.[15]
- Blood Labs: Cancer antigen 125 (CA 125) is a blood test that has been used to detect common proteins found in those with endometriosis. This is the same test that is used to tumour markers for various types of cancer. Although this blood test may identify proteins in advanced cases of endometriosis, it is not sensitive to the detection of mild or moderates disease. Due to the fact that this test is not sensitive to early disease detection, it is not recommended as a screening test for endometriosis.[16]
Differential Diagnosis[edit | edit source]
The most common symptoms of endometriosis are infertility and chronic cyclic pelvic pain; therefore, other conditions should be ruled out from the differential diagnosis.
Chronic pelvic pain is a usual symptom derived from pathologic conditions of the urologic, reproductive, and gastrointestinal system.
Pelvic inflammatory disease, adhesions, endometritis, primary dysmenorrhea, and second dysmenorrhea due to adenomyosis, myoma, cervical stenosis should be excluded from the pathologies of the genital system.
Other diseases that should be excluded come from the gastrointestinal system like irritable bowel syndrome, constipation, inflammatory bowel disease, and the urinary system, such as interstitial cystitis or chronic urinary inflammation.
Medical Management[edit | edit source]
Endometriosis is a non-curable disease.[15]The objectives of medical treatment focus on:
- Restoring normal pelvic anatomy.
- Removal of endometriotic implants.
- Pprevention of reoccurrence to decrease pain.
- Increase fertility.
In order to achieve these goals, the course of medical management depends heavily on:
- The severity of symptoms.
- The severity of disease.
- Age.
- Future child bearing plans.[20]
Generally, conservative treatment is recommended to control symptoms prior to surgical management.[16]
Treatment Options[edit | edit source]
The main aspect of medical management is aimed at reducing or blocking ovarian function due to the fact that endometriosis is related to hormone function,including:[17]
- Medications to control pain
- Hormonal Therapy
- Surgical management - less common approach due to unchanged aetiology and rapid implant regrowth.[15]
- Non-traditional management
Common medical treatments and their functions are listed in the medication section. In the instance that symptoms are severe and disabling, or conservative medical treatment fails, surgical procedures can be useful in removing/destroying implants and decreasing or ceasing symptoms.
It is important to note that implant regrowth may occur following removal and may only be a temporary source of symptom relief.[17]
Medications[edit | edit source]
- Anti Inflammatories (NSAIDs): Over the counter or prescribed NSAIDs work by decreasing the pain and amount of inflammation in the region of disease caused by displaced endometrial tissue or scaring. NSAIDs also decrease pain associated with menstrual bleeding by blocking the protein prostaglandin.[21]
- Birth Control Hormones:creates hormone levels in the body similar to those during pregnancy. This may slow or cease the growth of endometrial implants, and stop the shedding and discharge of menstrual fluid which will lead to a decrease in pain. This treatment may be used in the form of a pill, patch or ring and has been shown to have the least amount of side effects.[21]
- Gonadotropin Releasing Hormone Agonist (GnRH-a): This form of hormone treatment lowers estrogen levels in the body producing a state which mimics menopause. Typically this form of treatment is utilized in more severe cases where the pain is not relieved by birth control hormones and NSAIDs. GnRH-a works by stopping the growth-shrinking implants which in turn reduces pain.[21]
- Progestin (pill or shot): This form of hormone therapy works to create levels of progestin in the body similar to pregnancy. By mimicking the hormonal characteristics of pregnancy ovulation ceases and estrogen levels lower which causes shrinkage of endometrial growths and reduced pain.[21]
- Danazol: Increases androgen levels and decreases estrogen levels to create a menopause-like state. Similar to previous hormone therapy, this shrinks and stops the growth of endometrial implants which typically reduces pain. Treatment effects last approximately 6-12 weeks and may produce adverse signs/symptoms.[21]
- Aromatase Inhibitors: May provide relief in women who have not had improvements with hormone therapy. Aromatase works by inhibiting the amount of estrogen the tissue can produce. Studies have shown that aromatase inhibitors are effective at reducing pain and preventing the return endometrial growths. This form of treatment is often used in conjunction with hormone therapy such as birth control pills or progestin.[21]
Surgical Management[edit | edit source]
The major advantage of surgical treatment compared to pharmaceutical options is the ability to enhance fertility capability, but it can also provide pain relief simultaneously.
Surgery should be considered in cases of superficial endometriosis unresponsive or with contradictions to medical treatment.
When laparoscopic surgery is performed, then the recommendation is to excise all the endometriotic lesions and adhesions. By the ablation of the endometriotic tissue, the local inflammatory milieu decreases in the pelvic cavity, thus increasing the chances for conception.
The management of ovarian endometriomas is still challenging and poses a dilemma to clinicians. The reason behind this is the cumulative research evidence which shows that the removal of the capsule of the cyst can result in a decrease of the ovarian reserve and follicular loss. However, cystectomy is preferable compared to cyst drainage or ablation, due to its greater effectiveness in pain relief and to the lower recurrence rate.
The final decision about which treatment approach should be followed should be taken collaboratively with the patient, after a detailed explanation of all the possible risks and benefits related to each treatment option[1].
Physical Therapy Management[edit | edit source]
The use of physiotherapy in women with endometriosis has a conservative and complementary effect on the gynaecological, pharmacological and surgical treatment process.Treatment with physical methods can be an effective alternative to other forms of treatment.
The kinds of physical therapy used in women with endometriosis are mainly light therapy, laser therapy, electrotherapy and magnetotherapy, as well as:[22]
- Pelvic floor strengthening
- Internal and external trigger point management
- Myofascial manual therapy
- Stretching and flexibility exercises
- Spinal mobilizations
- Nerve glides
- Relaxation exercises
Secondary Musculoskeletal Impairments Caused by Endometriosis [23]
- Lumbar, sacroiliac, and pelvic floor pain
- Muscle spasms
- Trigger points (common in the pelvic floor and lumbar spine)
- Connective tissue dysfunction
- Urinary urgency
- Scar tissue adhesion
Physical therapy may prove to be an integral treatment option for the patient with endometriosis presenting with musculoskeletal impairments. By addressing this disease using a multidisciplinary approach, decreased pain and increased functional capabilities may be provided, allowing for a greater quality of life.
Phototherapy
In the field of phototherapy, the main action is to: accelerate the absorption of exudates and improve blood circulation and regeneration after surgery.[24]
Low-Level Laser (Light) Therapy (LLLT):
Endometriosis treatments with laser therapy have a profound effect on tissue regeneration and restoration. Deep infrared action has been shown in studies to increase endometrium proliferation and functional (functional) cellular capacity.
At the cellular level, laser irradiation causes:
- increased adenosine triphosphate (ATP) production
- increased activity of membrane enzymes
- increased synthesis of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA)
- accelerated electrolyte exchange between the cell and its environment.
At the tissue level, there is:
- an increase in blood and lymph circulation
- a decrease in intra-capillary pressure
- an increase in nerve ending excitability threshold.
- stimulation of the immune system
Magnetotherapy
The application of an electromagnetic field causes endogenous heat, or internal tissue overheating. The treatment is administered to the lower abdomen. The treatment method, duration, and number of treatments in a series are all individually tailored to the patient's condition and severity of symptoms. It is used to treat chronic conditions in which deep overheating and hyperaemia are indicated, and thus should not be used in women of childbearing age. It is preferable to use a pulse wave in this group of patients because a similar effect is obtained with almost complete elimination of the heat factor.[24]
Electrotherapy
Electrotherapy, which has an analgesic and hyperemic effect, is used in the physiotherapeutic management of endometriosis patients. Currents of medium and low frequency are used. Interference currents and Transcutaneous Electrical Nerve Stimulation (TENS) both can be used to relieve pain in the pelvic area.
Kinesiotherapy
Kinesiotherapy is an important part of the treatment for endometriosis patients, and it involves selecting an appropriate exercise programme with individually selected loads. Appropriately chosen exercises in the post-operative period and during inflammation treatment have a significant impact on patients' recovery and functioning. Kinesiotherapy is also appropriate for patients whose medical condition forbids surgical treatment.[24]
A study entitled Treating Fallopian Tube Occlusion with a Manual Pelvic Physical Therapy was performed in 2008 by Wurn, BF. et al. This study described the efficacy of a non-invasive soft tissue physical therapy treatment in opening bilateral occluded fallopian tubes in infertile women with a history of abdominal-pelvic adhesions.[25] Through this study it was discovered that non-invasive therapy such as manual techniques may be useful as an adjunct therapy in treating tubal occlusion caused by adhesions.[25]
Resources[edit | edit source]
- The International Endometriosis Association (IEA): Established by Mary Lou Ballweg, RN, PhD. The IEA functions as an online support and advocacy outlet for women diagnosed with endometriosis.
- Endometriosis Research Center: A lobbying organization working to support those with endometriosis through education.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Tsamantioti ES, Mahdy H. Endometriosis. [Updated 2021 Feb 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-.Available from: https://www.statpearls.com/ArticleLibrary/viewarticle/21107(accessed 5.4.2021)
- ↑ Horne, A.W.; Saunders, P.T.K. SnapShot: Endometriosis. Cell 2019, 179, 1677.
- ↑ Strathy JH, Molgaard CA, Coulam CB, Melton III LJ. Endometriosis and infertility: a laparoscopic study of endometriosis among fertile and infertile women. Fertility and sterility. 1985 Aug 1;44(2):83-8.
- ↑ Otolorin EO, Ojengbede O, Falase AO. Laparoscopic evaluation of the tuboperitoneal factor in infertile Nigerian women. International Journal of Gynecology & Obstetrics. 1987 Feb;25(1):47-52.
- ↑ Wiswedel K, Allen DA. Infertility factors at the Groote Schuur hospital fertility clinic. South African Medical Journal. 1989;76(1):65-6.
- ↑ Kyama CM, Mwenda JM, Machoki J, Mihalyi A, Simsa P, Chai DC, D'Hooghe TM. Endometriosis in African women. Women’s Health. 2007 Sep;3(5):629-35.
- ↑ D'Hooghe TM. Special focus: the challenge of endometriosis. Women’s Health. 2007 Sep;3(5):615-.
- ↑ Signorello LB, Harlow BL, Cramer DW, Spiegelman D, Hill JA. Epidemiologic determinants of endometriosis: a hospital-based case-control study. Annals of epidemiology. 1997 May 1;7(4):267-74.
- ↑ 9.0 9.1 Yovich, J.L.; Rowlands, P.K.; Lingham, S.; Sillender, M.; Srinivasan, S. Pathogenesis of endometriosis: Look no further than John Sampson. Reprod. Biomed. Online 2020, 40, 7–11.
- ↑ 10.0 10.1 Sampson, J.A. The development of the implantation theory for the origin of peritoneal endometriosis. Am. J. Obstet. Gynecol. 1940, 40, 549–557
- ↑ Wang, Y.; Nicholes, K.; Shih, I.M. The origin and pathogenesis of endometriosis. Annu. Rev. Pathol. 2020, 15, 71–95.
- ↑ Yovich, J.L.; Rowlands, P.K.; Lingham, S.; Sillender, M.; Srinivasan, S. Pathogenesis of endometriosis: Look no further than John Sampson. Reprod. Biomed. Online 2020, 40, 7–11.
- ↑ Kapoor, R.; Stratopoulou, C.A.; Dolmans, M.M. Pathogenesis of endometriosis: New insights into prospective therapies. Int. J. Mol. Sci. 2021, 22, 11700
- ↑ Konrad, L.; Dietze, R.; Kudipudi, P.K.; Horné, F.; Meinhold-Heerlein, I. Endometriosis in MRKH cases as a proof for the coelomic metaplasia hypothesis? Reproduction 2019, 158, R41–R47.
- ↑ 15.0 15.1 15.2 15.3 15.4 Goodman C., Fuller K. Pathology: Implications for the Physical Therapist. St. Louis, Missouri: Saunders Elsevier; 2009.
- ↑ 16.0 16.1 16.2 16.3 16.4 16.5 16.6 Endometriosis [Internet]. Mayo Clinic. 2018 [cited 2023 Mar 26]. Available from: http://www.mayoclinic.com/health/endometriosis/DS00289
- ↑ 17.0 17.1 17.2 Edwards, M. Understanding Endometriosis. Practice Nurse. December 2009; 38, 10: Proquest Nursing and Allied Health Source. pgs 22-25. Accessed: February 17, 2010.
- ↑ Goodman and Snyder. Differential Diagnoses in Physical Therapy. Saunders (W.B.) Co Ltd; 3rd Revised edition edition (28 Jan 2000)
- ↑ 19.0 19.1 Troyer MR. Differential diagnosis of endometriosis in a yound adult woman with nonspecific low back pain. Physical Therapy. 2007;87:801-810.
- ↑ 20.0 20.1 Endometriosis [Internet]. Nih.gov. [cited 2023 Mar 26]. Available from: http://www.nlm.nih.gov/medlineplus/ency/article/000915.htm
- ↑ 21.0 21.1 21.2 21.3 21.4 21.5 Endometriosis Health Center. WebMD. July 28, 2009. Available at: http://women.webmd.com/endometriosis/endometriosis-medications. Accessed March 7, 2010.
- ↑ ennfjord, M.K.; Gabrielsen, R.; Tellum, T. Effect of physical activity and exercise on endometriosis-associated symptoms: A systematic review. BMC Womens Health. 2021, 21, 355.
- ↑ Troyer MR. Differential diagnosis of endometriosis in a yound adult woman with nonspecific low back pain. Physical Therapy. 2007;87:801-810.
- ↑ 24.0 24.1 24.2 Wójcik M, Szczepaniak R, Placek K. Physiotherapy Management in Endometriosis. International Journal of Environmental Research and Public Health. 2022; 19(23):16148. https://doi.org/10.3390/ijerph192316148
- ↑ 25.0 25.1 Wurn, Belinda F; Wurn, Lawrence J; King, Richard C; Heuer, Marvin A; Roscow, Amanda S; Hornberger, Kimberly; Scharf, Eugenia S. Treating Fallopian Tube Occlusion with a Manual Pelvic Physical Therapy. Alternative Therapies in Health and Medicine: Jan/Feb 2008; 14, 1: pg 18.