Pancoast Tumor
Original Editors - Paul Clark and Sam Holzknecht from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Paul Clark, Sam Holzknecht, Lucinda hampton, Admin, Kim Jackson, Wendy Walker, Riccardo Ugrin, Elaine Lonnemann, WikiSysop, 127.0.0.1, Evan Thomas and Adam Vallely Farrell
Introduction[edit | edit source]
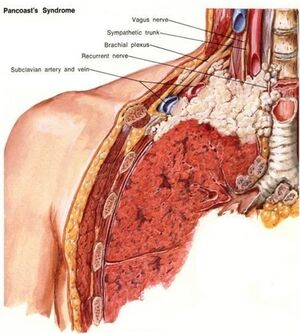
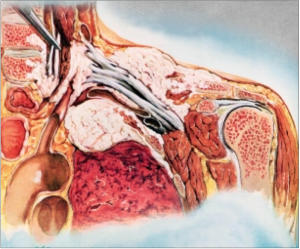
A pancoast tumour is a relatively uncommon primary lung cancer forming in the lung apex and invading the surrounding soft tissues. This is a tumour type is hard to difficult to treat because of its closeness, and frequent metastasis, to adjacent structures such as the subclavian vessels, lymphatic system, brachial plexus, spine, second and third ribs, stellate ganglion, and the sympathetic nervous system.[1] Pancoast tumors are often mis-diagnosed and/or diagnosed too late.[2]
Etiology[edit | edit source]
Common risk factors include (similar to lung cancer):
- Smoking
- Secondhand smoke
- Asbestos exposure
- Exposure to industrial elements like gold or nickel
- Diesel exhaust[3]
Epidemiology[edit | edit source]
Pancoast tumours account for 3-5% of all bronchogenic carcinomas and having demographics similar to other lung cancers (see link).
Characteristics/Clinical Presentation[edit | edit source]
Symptom commonly include: chest shoulder and/or arm pain; weight loss (frequently present). Pancoast syndrome occurs in 25% cases and includes: severe pain in the shoulder region radiating toward the axilla and scapula along the ulnar aspect of the muscles of the hand; atrophy of hand and arm muscles; Horner syndrome (ptosis, miosis, hemianhidrosis, enophthalmos); and compression of the blood vessels with edema."[4][5]
Diagnosis[edit | edit source]
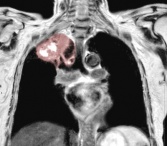
Diagnosis is often difficult and delayed due to the typical absence of lung cancer symptoms and includes a combination of CT scans and MRI followed by a biopsy confirmation [1].
Systemic Involvement[edit | edit source]
Pancoast tumors are often mis-diagnosed or diagnosed too late to prevent metastasis to adjoining structures. This form of lung cancer is very dangerous because of its close proximity to other structures that include[2]:
- Brachial plexus- this is commonly invaded at the ulnar distribution (C8-T1), thus atrohpy/parasthesia of the ulnar distribution is reported. This is often the patient's primary complaint/presentation to therapy with shoulder pain that radiates down to the 4th and 5th digit.[1]
- Subclavian vessels- metastasis to the subclavian artery or adjoining structure may lead to deterioration or compression of blood vessels causing edema.[6]
- Stellate ganglion- This is a large ganglion located at the bottom of the cervical spine in connection with the thoracic spine and is related to the sympathetic nervous system. Invasion of this may result in Horner's Syndrome.[3]
- Adjacent vertebral bodies- If the tumor is able to metastasize the vertebral bodies, this may lead to permanent paralysis of the affected area.[3]
Medical Management[edit | edit source]
For many years, Pancoast tumors were considered untreatable. In recent decades, the outlook for people with Pancoast tumors has greatly improved.Treatment for Pancoast tumors involves a combination of chemotherapy, radiation, and surgery. Treatment depends on the extent of involvement especially through the apex (these tumours usually involve the brachial plexus and subclavian vessels). In this case, radiotherapy is typically administered in an attempt to reduce the tumour sufficiently to allow for attempted resection. If the tumor has metastasized to areas beyond the chest surgery may not be appropriate.[7]
Complete resection is the major factor in determining survival
- complete resection achieved - 45% 5-year survival
- incomplete resection only - 0% 5-year survival[5]
Physical Therapy Management[edit | edit source]
A mutimodal management of the patient is needed. When it comes to pancoast tumors, the primary role of the Physical Therapist is post operative treatment to prevent respiratory complications including atelectasis and pneumonia, and control the patient’s pain levels. The patient is immobilized the first post op day with attention to chest Physiotherapy and bronchoscopic suctioning may be necessary to clear secretions in patients with ineffective cough [4].
Intensive respiratory care should be implemented and chest expansion measurements should be taken to ensure: [8]
- Adequate ventilation using mechanical support (if necessary)
- Satisfactory chest tube function
- Clearance of secretions by mobilizing, coughing, chest physiotherapy, nasotracheal or orotracheal or bronchoscopic suctioning, or a temporary tracheostomy
- Adequate analgesia
- Increased transpulmonary pressure with incentive spirometry or continuous positive airway pressure mask.
Avoid fluid overload and use diuretics judiciously to avoid acute respiratory distress. Chest tubes remain in place until all air leaks have stopped, there is complete lung expansion and almost no fluid drainage present [8].
The role of the Physical Therapist it also depends on the type of surgery approach used. Its focus could be the articular mobilisation of the shoulder complex, the neck and the thoracic spine according to the surgeon. Soft tissue mobilisation around the scar (it can block the normal clavicular or scapular range of motion) can be performed 2-3 weeks after operation. Early mobilisation of the gleno-humeral can prevent secondary shoulder limitation in the ADL as subluxation and adhesive capsulitis [9].
Specific scapular mobilisation could be address to the limited range of motion and to the local pain after surgery. To obtain better outomes the passive and active self mobilisation of the thoracic spine and of the cervical spine could be useful in the early stages of rehabilitation.
The muscular strengthening of the arm and of the shoulder girdle can be performed in the early stages of rehabilitation. At the beginning isometric contraction could be useful to avoid excessive pain caused by repetitive active mobilisation. The simultaneous respiratory and surgical complication must be assessed before starting the strenghtening program.
Pancoast tumors with brachial plexus involvement use to underwent to neurosurgical nerve resection of C8-T1 roots. The tumor resection and the neurolysis could both outcome in a significant loss of hand function in many patients. In those cases the PT could prevent the muscluar retraction of hand and wrist with splints or orthesis. Exercises address to mobility and strenght can improve the quality of life and the arm and shoulder function.
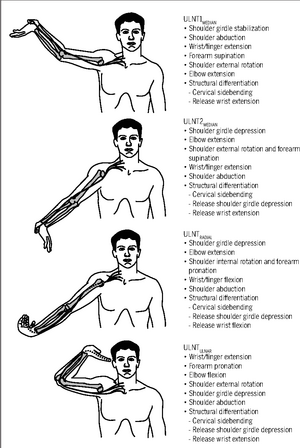
The use of the Upper Limb Tension Test 3 (ULTT3) as a neurodynamic slider mobilisation could be a good exercise in the first stages of rehabilitation. The passive neurodynamic mobilisation could be address to make feel the patient comfortable in the positione: then self ulnar nerve mobilisation in contribution to a specific home exercise program could improve range of motion both acting on the mechanosensitivity of the nerve and on the articular active movements of scapula and gleno-humeral. In that cases in which the abduction of the shoulder is limited some variation could be performed.
Laser therapy in combination with and individualized wellness program is also effective in treating this condition and can improve the patients overall health and wellbeing. [9] Ultrasound therapy could be locally contraindicated in those patient who risk bones or pulmunar metastasis.
Differential Diagnosis[edit | edit source]
Lung Cancer; Non-Small Cell or Oat Cell (Small Cell) [6]
Thoracic Outlet syndrome
Pulmonary echinococcosis [10]
Pulmonary leiomyosarcoma [11]
C8-T1 radiculopathy [12]
Primary Ewing's sarcoma [6]
Case Reports/ Case Studies[edit | edit source]
Paravertebral Cervical Nerve Block in a Patient Suffering from a Pancoast Tumor
Pancoast Tumour: a case report
Alleviation of Pancoast tumor by Ultrasound-guided Percutaneous Ablation of Cervical Nerve Roots
Superior Pulmonary Sulcus Tumor
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Archie V, Thomas C. Superior sulcus tumors: a mini-review. The Oncologist [serial on the Internet]. (2004), [cited April 4, 2012]; 9(5): 550-555. Available from: MEDLINE.
- ↑ 2.0 2.1 Aigner C, Klepetko W. Current treatment concepts of Pancoast tumors. European Surgery: ACA Acta Chirurgica Austriaca [serial on the Internet]. (2010, Oct), [cited April 4, 2012]; 42(5): 214-219. Available from: Academic Search Premier.
- ↑ 3.0 3.1 3.2 Muscolino G, Valente M, Andreani M. Pancoast tumours: clinical assessment and long term results of combined radio surgical treatment. Thorax. 1997; 52:284-6
- ↑ 4.0 4.1 Shabir Bhimji, Md, May SK. Emedicinehealth: Pancoast tumor. [homepage on the Internet]. April 2, 2011 [cited 2012 Mar 30]. Available from: http://www.emedicinehealth.com/pancoast_tumor/article_em.htm#Pancoast%20Tumor%20Overview
- ↑ 5.0 5.1 Radiopedia Pancoast Tumour Available:https://radiopaedia.org/articles/pancoast-tumour (accessed 24.1.2023)
- ↑ 6.0 6.1 6.2 D'silva KJ. Medscape: Pancoast syndrome, [homepage on the Internet]. Aug 15, 2011 [cited 2012 Mar 30]. Available from: http://emedicine.medscape.com/article/284011-medication
- ↑ Healthline Pancoast tumours Available:https://www.healthline.com/health/pancoast-tumor#treatment (accessed 24.1.2023)
- ↑ 8.0 8.1 Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011. CA: A Cancer journal for clinicians. 2011;61:212–36.
- ↑ 9.0 9.1 9.2 Petraglia, Md JF. International Pain Medical Group. [homepage on the Internet]. 2008 [cited 2012 Mar 30]. Available from: http://www.gotpaindocs.com/pancsts_tmor_syndrm.htm
- ↑ Kim P, Hains F, Wallace M, Mior S. Pancoast tumour: a case report. Journal Of The Canadian Chiropractic Association [serial on the Internet]. (1993, Dec), [cited April 4, 2012]; 37(4): 214. Available from: Health Source: Nursing/Academic Edition.
- ↑ Shoji F, Yoshino I, Takeshita M, Sumiyoshi S, Sueishi K, Maehara Y. Pulmonary leiomyosarcoma presenting as a pancoast tumor. Pathology - Research & Practice [serial online]. October 18, 2007;203(10):745-748.
- ↑ Abdul Khader S, Kumar A. Pancoast tumor presenting as shoulder pain and C8-T1 radiculopathy: a case report. (Poster Session). Archives Of Physical Medicine & Rehabilitation [serial online]. November 2002;83(11):1663.