Electrical Stimulation - Its role in upper limb recovery post-stroke
Original Editor - Grant Burns as part of the Queen Margaret University Contemporary and Emerging Issues in Physiotherapy module
Top Contributors - Rebecca Graham, Grant Burns, Craig Philip, Joshua Tan, Hannah Little, Rucha Gadgil, Kim Jackson, 127.0.0.1, Admin, Rachael Lowe, Venugopal Pawar, Cindy John-Chu, Carina Therese Magtibay, Dinu Dixon, Evan Thomas and Jane Hislop
Introduction[edit | edit source]
Stroke has a large impact and burden on society [1]. The UK has approximately 1.2 million stroke survivors, with half experiencing disability and 77% with upper limb difficulties [1]. In the UK the over 65’s population is estimated to rise by over 40% in the next 17 years [2], and stroke incidence is higher in this demographic. [1]. This could potentially lead to even greater numbers of stroke survivors requiring support and rehabilitation from healthcare professions such as physiotherapy. Additionally, 36% of 65+ live alone [2] underpinning the importance of successful rehabilitation of upper limb function if people are to maintain independence, due to its impact on performance of activities of daily living [3].
Electrical Stimulation (ES) also Known as Electrical muscle stimulation, has a developing evidence base that supports its use for upper limb recovery after stroke [4] and the number of trials has quadrupled over the last decade [5]. However, current practice is varied and research shows that a lack of knowledge and skills are a key barrier to its use [4]. This learning package aims to addresse this contemporary issue by introducing and synthesizing key literature and translating this into practical recommendations that can support clinical practice.
What is Electrical Stimulation?[edit | edit source]
ES is an assistive technology that can be used to aid the recovery of upper limb after stroke. It uses electrical current to stimulate muscle contraction via electrodes, facilitating movement of a weakened or paralysed limb. It has been used since the mid 1960’s, traditionally to aid mobility through addressing dropped-foot, however, more recently it has been considered as a promising treatment modality for upper-limb recovery . ES has also been used in the treatment of other upper motor neuron impairments including people with Cerebral Palsy, Parkinson’s Disease, Multiple Sclerosis and spinal cord injury .
ES Uses[edit | edit source]
Several uses and benefits have been investigated regarding ES use in stroke upper limb recovery. These include strengthening weak muscles, increasing range of motion, reducing spasticity, improving motor control, reducing shoulder subluxation, reducing pain associated with shoulder subluxation and spasticity, improving sensory and proprioceptive awareness, and improving effects of botulinum toxin for management of spasticity [6], [7].
Neuroplasticity is a key concept underpinning stroke recovery and it is the ability of the brain to adapt and form new neuroconnections [8][9]. Following stroke there is evidence that the brain has a period of hyper-excitability within the first weeks after stroke[10] and it is hypothesised that central reorganisation can be enhanced by stimulation through movement which ES may be able to facilitate [11].
Figure 3. ES device and electrodes.
Terminology[edit | edit source]
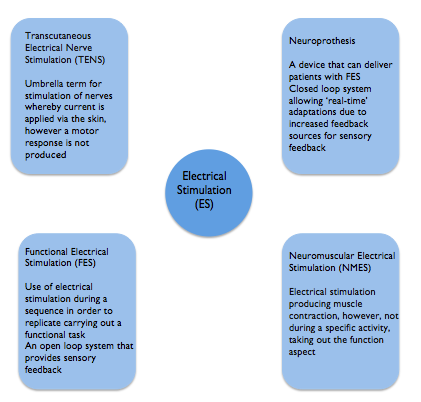
There are various terms used within the literature for Electrical Stimulation following Stroke. Although each has a different meaning they are frequently used synonymously which can make understanding and comparing different studies challenging [12]. Highlighted below are the most common terms encountered in the literature. This learning resource has focused on ES used to support upper limb recovery which is delivered predominantly by Neuromuscular Electrical Stimulation (NMES). Functional electrical stimulation (FES) is a form of NMES. Transcutaneous electrical nerve stimulation (TENS) has traditionally been used for analgesic purposes and does not elicit a motor response [13][4] although it has also been used in studies to promote sensory feedback[4].
One distinction made is the use of ES for therapeutic purposes such as to aid motor learning with the intention of a carryover effect beyond treatment. Functional electrical stimulation on the other hand is aimed at providing direct benefit to aid a task at the time of wearing and is used in an orthotic manner [5].
Figure 4. Terminology [13] [4] [14]
Physiology[edit | edit source]
ES uses electrodes to activate contraction and relaxation of muscles that have been affected by an upper motor neuron lesion. Motor-units are electrically stimulated by depolarization of motor axons, or terminal motor nerve branches. When depolarization reaches threshold, an action potential occurs due to sodium flowing from the extracellular to intracellular space. This action potential propagates along the nerve fibre axon to the muscle via the neuromuscular junction resulting in muscle contraction [14]. As an intact connection between the ventral horn and muscle is required for successful action potential propagation to reach the muscle, ES is not suitable for lower motor neuron lesions [15]. In ES it is the nerves that are stimulated rather than muscle, as they require a lesser current of that needed to trigger muscles directly. Influencing factors include distance from electrode to nerve fibre, size of motor unit, and surrounding tissue will all impact the number and type of motor units activated[16]
ES engages the patient and delivers feedback of a sensory and visual nature, beneficial for stroke patients during their recovery and promotes motor re-learning [17] [18][19]. Disuse atrophy is a common secondary complication of stroke and can result in muscle fibre changes [16]. ES may also be able to reverse this by bringing about muscle fibre changes over the course of treatment, with type II glycolytic fibres reverting to type I oxidative skeletal muscle fibres [16]. Type II fibres generate greater forces but fatigue more quickly, whereas type I fibres produce lesser force but are more fatigue-resistant [20]
Motor unit recruitment and Fatigue[edit | edit source]
In normal physiology, nerve fibre recruitment occurs as described by the Henneman size principle of voluntary motor unit recruitment, whereby nerves with the smallest diameter will be recruited first. When using ES, the reverse of this happens, with the largest diameter neurons being recruited first due to a lower nerve stimulus threshold [20] which can lead to fatigue. However, it has recently been established that it is in fact an uncoordinated method of recruitment, with no order, that is evident in ES [5].
One limitation of ES is that muscles can fatigue [21] and that the higher frequency selected, the quicker muscle fatigue will set in. Clinical judgement is required to determine the lowest frequency possible to achieve tonic muscle contraction[4]. Once fatigue has set in, there is argument for and against whether muscle strengthening can occur. It has been suggested that when fatigued, that no advantages can be gained from additional stimulation and therefore fatigue should be prevented if possible. Opposing arguments suggest that strengthening can only be achieved if the muscle fibre is worked to its maximum. Clarifying the cause of fatigue is important. Strengthening can occur if fatigue is within the muscle fibres due to cellular processes being activated, however if fatigue has resulted from neurotransmitter depletion or propagation failure, then the muscle will not be strengthened as the fibre is not being stimulated sufficiently [13].
ES Devices and Parameters [edit | edit source]
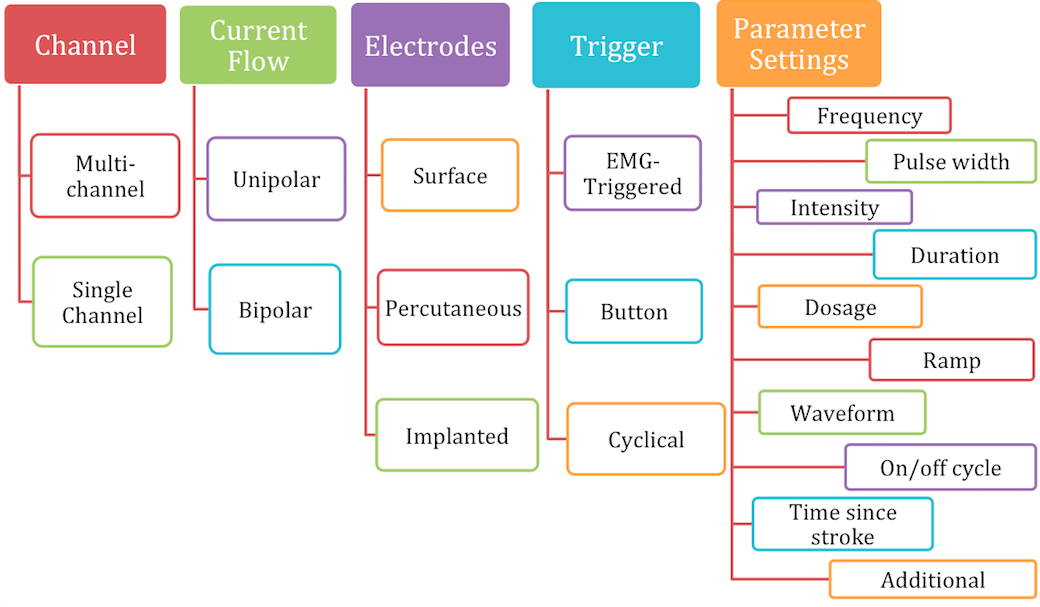
ES systems include three components: the control, an electrical stimulator and electrodes which connects the ES with the nervous system [16]. There are several different ways ES devices can be configured as depicted and described below. This can vary by supplier and intended use.
Figure 5. Overview of ES device and configuration.
Electrodes[edit | edit source]
Application of ES is done via electrodes, which generate the electrical field through either surface or percutaneous electrodes [22].ES can be provided via single channel or multichannel devices. Multichannel systems can be used when targeting multiple muscles to replicate a functional activity such as reaching and grasping, whereas a single channel device is used for less complex movements such as rectifying shoulder subluxation. Although single channel systems are mainly used for simple movements, they are potentially more portable, making them more practical for home use.
Surface electrodes are placed directly onto the skin over the nerves and are the most common [20]. As well as stimulating muscle, surface electrodes may also be used to achieve a reflex action[22]. Parameters of ES needed when using surface electrodes can differ depending on factors such as material of the electrodes, placement and surface area [20]. They are non-invasive and relatively inexpensive and are suitable for use across a wide variety of settings and by therapists and patients, promoting independence and self-management [4]. However, targeting contraction of small individual muscles can be difficult, and often activation of deeper muscles requires superficial muscles to be activated first.
Surface electrodes have also been reported to cause pain for some patients when compared with percutaneous electrodes [23]. Percutaneous electrodes penetrate through the skin into the muscle via hypodermic needles or can be completely implanted whereby stimulation is received from an external unit [20]. They can address the difficulties faced with surface electrodes by being able to target deep muscles and as they use lower currents, are reported to be less painful [20]. However, factors of cost and practicality need to be taken into consideration [22]. In line with the SSAF [4] recommendations, this resource focuses on the surface mounted electrodes as they are inexpensive and most commonly used, therefore best suited to this learning resources target audience as it covers the equipment they are most likely to encounter in practice.
Unipolar vs Bipolar[edit | edit source]
Two electrodes are required (active and indifferent) to generate a flow of current, however these can be unipolar or bipolar in configuration. Unipolar is when one electrode is more active than another due to their sizes. The active electrode is typically smaller and placed near the nerve to be stimulated with the indifferent electrode placed over less excitable tissue such as fascia. In multi-channel configurations there are several active electrodes but only one indifferent electrode is required [13]. Bipolar electrodes are both the same size meaning the current at each site will be equal. Both active and indifferent electrodes are placed close to the stimulated nerve and in multi-channel configurations there is an indifferent electrode for each active. Bipolar systems enable greater targeting of muscles [13].
Cyclical, button & EMG-triggered[edit | edit source]
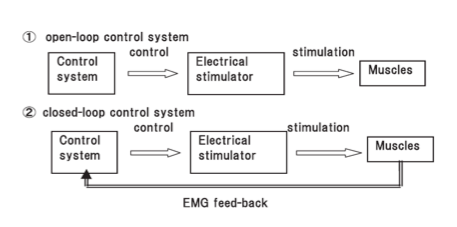
One other variable is whether the timing of when electrical stimulation is active is via a button, cyclical or EMG-triggered system. The button method is a manual form of ES and requires the user to actively press a switch to activate the stimulation. Cyclical means that the timing and sequence is predetermined by selection of an appropriate programme on the device and this is sometimes just referred to at NMES [4]. Alternatively, the EMG triggered system uses sensors built in to the active electrode to determine when voluntary muscle contraction is above a preset-threshold which then triggers the ES [4]. It has been suggested that this method promotes greater user involvement promoting neuroplastic changes [5]and leads to better outcomes [24][25]. Typically this is called EMG-triggered ES to differentiate from cyclical [4]. Cyclical or predetermined programs are considered open loop systems that rely on the user to turn-on/off whereas EMG-triggered are classified as closed-loop systems as the EMG trigger determines on/off timing.
Figure 6. Open and closed loop ES systems. [25]
Parameters[edit | edit source]
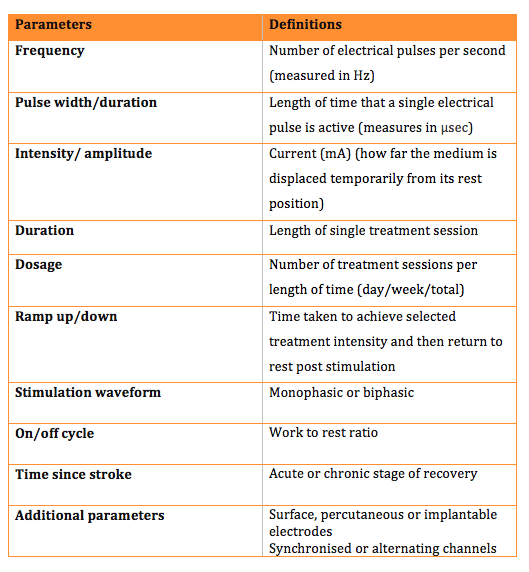
There are various parameters that can be adjusted on ES devices to help tailor the electrical field effects to the patient and its intended application. Table 1. below outlines each parameter and its definition. It is worth noting that not all ES devices allow individual control of all parameters and many offer a choice of pre-determined programmes.
Table 1. Parameters and definitions. [4]
When should I use Electrical Stimulation?[edit | edit source]
Guidelines [edit | edit source]
In order to synthesise the current evidence for the use of ES for upper limb rehabilitation after stroke this section will focus on the three main guidelines used in the United Kingdom for guiding physiotherapy practice:
Table 2. SIGN118 Guidelines [12]
Table 3. NICE CG162 Guidelines [26]
RCP Guidelines[27][27][27][27][27][27][27][16][21]
Table 4. RCP Guidelines [27]
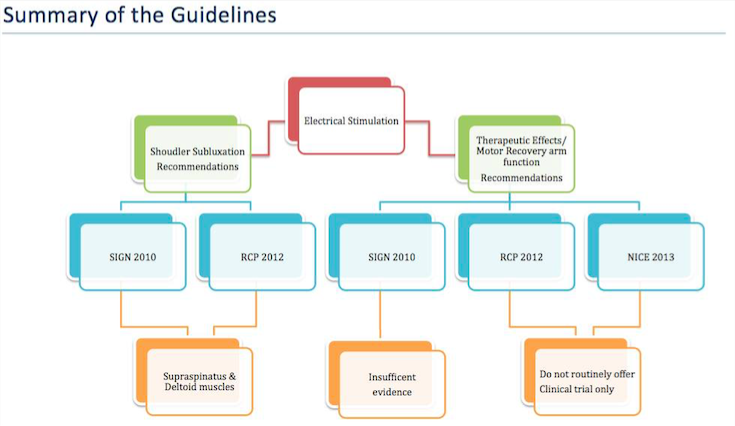
Overall there is general consensus and robust evidence to support the use of ES in the management of shoulder subluxation and this features in both the SIGN and RCP guidelines. However the findings for ES are not ubiquitous for other uses, including motor control, spasticity and shoulder pain. While ES to improve motor control does appear to have a growing evidence-base in the underpinning literature, it is unclear whether the gains reported translate across to meaningful arm function improvements such as activities of daily living or improved participation. Current evidence is not sufficiently robust to recommend routine use of ES therapeutically to improve motor control and arm function but as side-effects are minimal its use as an adjunct in a trial approach is merited.
Figure 8. Summary of the Guidelines.
Evidence[edit | edit source]
The recommendations in the guidelines were formulated based on review of the evidence base. Of the three guidelines bodies, only two have consistency in recommending ES for treatment of those with, or at risk of, shoulder subluxation [12] [27] This is supported by a robust evidence base [4]. Additionally, both the NICE [26] and RCP [27] guidelines state that therapeutic ES, to improve arm function by promoting motor recovery, is not routinely recommended [26]and should only be considered as a clinical trial. [26] [27] SIGN have stated that there is insufficient evidence to support its use in this manner also [12].
Although several trials reviewed in the guidelines have shown positive effects for ES, caution has been advised in interpreting the results due small sample sizes, poor methodological quality and large heterogeneity across studies inhibiting pooling of the data.[12] [26][27] Where benefits have been reported, they tended to demonstrate improvement in outcomes such as reduced impairment and increased function, but this did not always translate to improved activities of daily living or activities so benefits may not be meaningful to patients. This is an important point to note when considering the ICF framework which aims to support a holistic view [28]. Few studies provided sufficient follow up period also, so where data supporting ES for shoulder subluxation has been more robust, there is question as to whether this benefit can be maintained long term without continual treatment. [29]
One consideration is that the guidelines range in age of publication (2010-2013) and the evidence used within them to base recommendation may not be considered upto date, particularly as there has been an 4 fold increase in ES RCTs over the last decade [30]
Systematic Reviews[edit | edit source]
Systematic reviews (SR) are considered at the top of the evidence hierarchy[31]and Cochrane are considered by many as the gold standard [32].
A Cochrane overview reviewing the effectiveness of interventions for the recovery of upper limb function after stroke was recently produced [3]. The Cochrane review concluded that there was only low-quality evidence related to the use and effectiveness of ES. Difficulties due to the large variation in trials limited the ability to combine results to gain greater confidence in findings [3]. Overall the conclusion was that there was insufficient high quality evidence to support a change in clinical practice and further research was required to verify some of the beneficial treatment effects reported.
Howlett [19]investigated FES in improving activity after stroke compared with training alone and is the most up to date SR found in our literature search. Statistically significant standardised mean differences (SMD) were reported in favour for ES for activity when compared with no treatment or placebo. FES was also beneficial to activity when compared with training alone. However, the authors were unable to determine whether FES activity benefits improved participation or if activity benefits were long-lasting. Using SMD was also necessary as studies used different outcome-measures which means that although large effects-sizes were found for upper limb, it was difficult to translate this into real term, therefore clinical significance for FES in upper limb was unable to be verified.
Conclusion[edit | edit source]
ES has a developing evidence base and is recommended for use within the national guidelines although this is not consistently applied. Its use is generally considered as an adjunct to standard treatment rather than a stand-alone intervention such as promoting greater task based practice. Evidence supporting the use of ES for reduction of shoulder subluxation is robust for treatment in the early phase. Evidence supporting its use is less conclusive for improving motor control to aim improved arm function. While benefits have been shown they do not always transfer to meaningful improvements in activities or ADL’s and limited studies have investigated benefits with long-term follow up. The evidence base is currently limited by the small sample sizes, limitations in methodological quality and large variation in protocols and dosage prohibiting pooling. That said, there is a trend emerging that ES can offer a therapeutic effect and benefits may extend beyond treatment, however, further high-quality research is required to validate this.
How do I use Electrical Stimulation?[edit | edit source]
General Considerations[edit | edit source]
ES can be applied by qualified health professionals; including physiotherapists and occupational therapists who are competent in its use. Although this learning package gives a theoretical overview of ES we advise that practical training is performed before applying this as a treatment [33].
The checklist below details what training is required prior to use of ES.
ES training checklist [33]
Contraindications and Precautions:[edit | edit source]
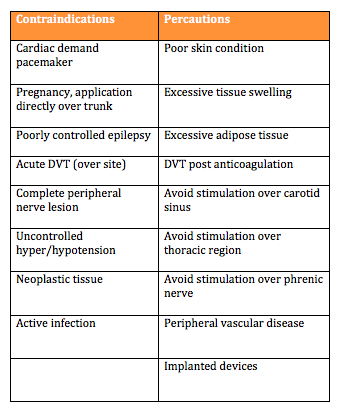
To ensure patient safety it is important to consider the contraindications and precautions before using an ES device. Understanding these will help make an informed decision regarding the use of ES as a treatment.
Table 5. Summary of the main contraindications and precautions.[4]
What do Patients need to know?[edit | edit source]
Prior to commencing treatment the patient needs to give verbal consent. After they have given consent it is important to provide the patient with information regarding ES. They should be informed of the expected skin sensation and that it may uncomfortable, but they should not experience any pain. Patients should also be told what to do if they experience these sensations and how to work the ES device if a patient or carer is able to adjust it. Providing an instruction manual in lay terms may be beneficial for the patient. Contraindications and precautions should be highlighted to the patient with relevant information explained to them. After providing the patient with this information, it should be documented in the patient notes [33].
Outcome Measures[edit | edit source]
There are a variety of outcome measures that can be used in the monitoring of upper limb recovery and function post stroke, which could be used to track progress of patients using ES. Two of these outcome measures will be outlined below.
Motor Assessment Scale(MAS):
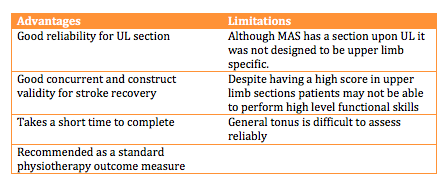
The MAS is an outcome measure which focuses upon functional motor activities. It has specific sections for Supine to side lying, Supine to sitting over side of bed, Balanced sitting, Sitting to standing, Walking, Upper-arm function, Hand movements, Advanced hand activities and general tonus. Each of these are scored from 0-6. A score of six is the optimal motor score in each area, with a score of 48 being available in total [34].
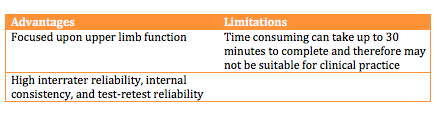
Table 6. Advantages and limitations of MAS. [34]
Wolf Motor Function Test:
The Wolf Motor Function Test is a timed outcome measure which aims to assess how quickly functional upper limb tasks can be performed. Patients are given 120 seconds to complete each task before the task is marked as incomplete. Each of these are scored from 0 to 5. Five is the best score achievable for each task, with 75 being the maximum score available [35].
Table 7. Advantages and limitations of Wolf Motor Function Test. [35]
Uses of ES[edit | edit source]
Shoulder Subluxation[edit | edit source]
Inferior glenohumeral joint displacement, commonly known as Shoulder subluxation is a secondary musculoskeletal impairment amongst patients with extreme muscle weakness and limb inactivity [36]. Incidence rates are reported between 17%-81% of people who suffer a stroke, with greater paralysis related to higher incidence [37]. Weakness of the shoulder musculature can result after a stroke, leading to subluxation due to the muscles being unable to hold the humerus within the glenohumeral fossa against the pull of gravity [36].
How does ES aid in Shoulder Subluxation?
UK national guidelines recommend ES of the upper limb post stroke to prevent shoulder subluxation [12][27]. Linn and Fil [38] [39]stated that early application of FES, preferably within the first 48 hours post stroke is vital in achieving positive results. While benefits have also been shown when applied within the first 2-3 weeks, they are less effective [4] and it has been suggested that once the capsule and soft-tissues around the shoulder are stretched then may persist [38]. Therefore early application of ES is recommended and has shown to improve function, muscle tone, joint alignments and sensory deficits [40]. Caution has also been advised for those with more severe paresis, however, as some patients have shown a delay in restoration of motor control as a result of treatment, therefore the benefits need to be weighted appropriately [41].
Some benefits for patients in the chronic stages of stroke have been reported however they were not statistically significant [42] [4][43] although limited evidence exists in this stage, therefore further evidence is required for a definite conclusion [29].
Guidelines and evidence advocate stimulation of the supraspinatus and deltoid muscles [29] [27] [26]. Stimulation of the supraspinatus alone has been shown to be inadequate in maintaining the humeral position of the shoulder [44]. A more recent study by Manigandan [37] found that additional stimulation of the long head of biceps improved effects on preventing shoulder subluxation. This could be an alternative treatment option if stimulation of the supraspinatus and deltoid muscles prove ineffective.
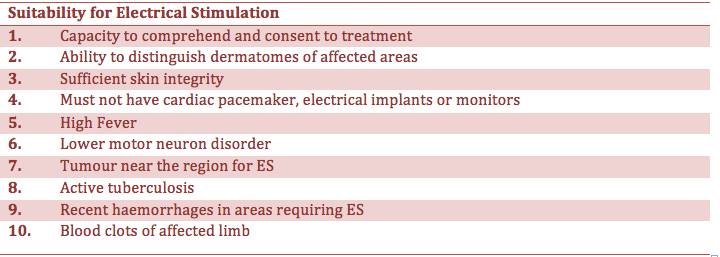
Another audit by Larkin [4] presents a table to address some of the concerns about the suitability of ES for patients. The table below shows the suitability for ES.
Table 9. Suitability for Electrical Stimulation[4]
Application:
Before applying ES to the upper limb to assist reduction of shoulder subluxation, you should consider the movement you wish to illicit and the structures involved in this movement. Below are some illustrations of ES being used with slight variations.
The settings below are by Odstock Medical Limited who are one of the main distributors of ES in the UK.
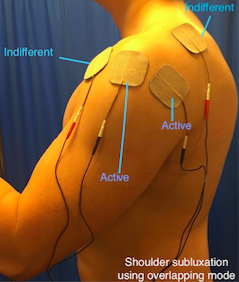
Reducing Shoulder Subluxation
- 2 pairs of electrodes required
o Placement of pair 1: Supraspinatus & Middle fibres of Deltoids
o Placement of pair 2: Anterior & Posterior fibres of Deltoids
Figure 10. Shoulder subluxation (overlapping mode).
This is using a dual channel device. A quick guide would be for all 4 electrodes fitting under the hand of the clinician over the patient’s shoulder. When adjusting the current to relocate the position of the humerus, extensive shoulder abduction should be avoided.
If a single channel device is only available, place electrodes over middle deltoids and supraspinatus. Polarity to which is active can be adjusted, if too much shoulder abduction is observed, switch polarity of active to supraspinatus instead of middle deltoids.
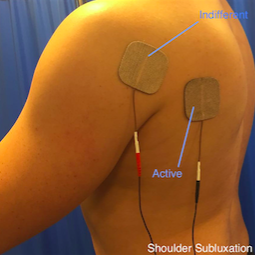
Reduction of shoulder subluxation with external rotation
This is suitable for patients with significant anterior subluxation/ internal rotation of the humerus.
- 2 pairs of electrodes required
o Placement of pair 1: Supraspinatus & Middle fibres of Deltoids
o Placement of pair 2: Teres Minor & Posterior fibres of Deltoids
You can adjust which electrodes are active or indifferent depending on your patient’s needs. This set up should relocate the humerus more posteriorly.
The current levels may be too high when the shoulder is brought into elevation and you should adjust the current where appropriate to achieve humeral relocation.
Figure 11. Shoulder Subluxation
Recommendations:
If the shoulder is internally rotated, place one electrode on the posterior fibres of deltoid and the other on teres minor. If further external rotation is required, electrodes can be placed on teres
minor and infraspinatus.[45]
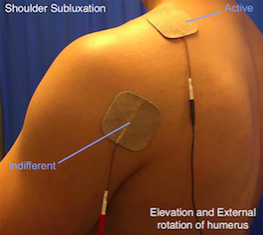
Figure 12. Elevation and external rotation of humerus
Recommendations:
If the shoulder is subluxed without any rotation, place the electrodes over the middle fibres of deltoid and supraspinatus.
Stimulation of the supraspinatus may be challenging due to the lack of stimulation to the trapezius which would result in shoulder elevation. If that occurs, replace the electrodes over the middle and posterior deltoids.
Dosage and Parameters:
Before commencing with ES treatment on a patient, it is important to consider the parameter settings available and decide upon the most appropriate settings for your patient. The different types of settings used can provoke various responses from patients.
Summary of main parameters outlined below:[edit | edit source]
Frequency:
A diverse range of frequencies has been reported in the literature. Salisbury [46]reported that 40hz is sufficient to elicit a contraction, however Ada and Foongchomcheay [36] reported that any frequency range above 30Hz would be sufficient for muscle activity. Furthermore, Baker and Parker [47] stated that a range between 10 – 60Hz could also be sufficient. Therefore, there is inconclusive evidence towards a specific stimulation frequency and clinicians should use their own clinical judgment to achieve a tetanic contraction. Although 20 – 30 Hz is the most commonly reported.
Pulse amplitude and pulse width:
Pulse amplitude, width and frequency have been described as the most important factors in achieving a visible contraction. Although many trials do not state or justify their amplitude and width settings a standard pulse width of 300µs has been suggested as a starting point [46]. However the general consensus of various authors reports values between 100-350µs [4]. Key factors to consider during selection of pulse amplitude and width are muscle fatigue and patient comfort [24]. The quality of underpinning research however is low [4] and therefore further research is warranted to uncover the full effects of pulse amplitude and pulse width.
Length of treatment:
Evidence regarding the length of treatment is inconsistent due to various individual session durations and overall length of treatment varying between studies. Ada and Foongchomcheay [36] synthesised the evidence and recommended that ES be initially applied for1 hour per day and gradually increased to 6 hours per day. It is recommended patients should continue with the treatment until they have a score of more than four on the MAS outcome measure to reduce the reoccurrence of a subluxation[36]. Chantraine et. al.[48]reported that improvements could be seen within the first 12 months of treatment and none thereafter with progress being maintained when re-measured at 24 months.
Waveform, ramp times and on/off cycle time:
There are no definitive guidelines or evidence justifying the specific types of waveform, ramp times and on/off cycle time when treating a subluxed shoulder. However a slower ramp –up time of at least 2 seconds is recommended when spasticity is present as a sudden contraction would elicit a stretch reflex resulting in a reduced range of motion and a long ramp could also be beneficial towards reducing tone [45]. The general consensus is in conjunction with conventional therapy, ES should be considered in the acute stages, ideally the first few days post stroke, when flaccidity is present and shoulder subluxation risk is high.
Motor Control and Recovery[edit | edit source]
Electrical stimulation (ES) in patients with motor function impairment of the upper extremity has been employed as a rehabilitation modality for many years. In order for ES to beneficial as a treatment for motor control, it is reported that patients should have some degree of movement.
NICE [26] suggest that patients should be able to hold a contraction but may not be able to move their arm against resistance. The proposed mechanism for upper limb motor control ES is to strengthen the elbow, wrist and finger extensor muscles, reduce the spasticity of the antagonist muscles and help to promote neuroplastic changes [45][42]. However, there is a limited evidence indicating that repeated muscle activation using ES may lead to improvement in voluntary motor control and providing a carry over effect [5].
How does ES aid Motor control?
It is understood that when a muscle contraction is produced by electric stimulation, a whole range of sensory inputs are produced. This includes the direct sensation from the stimulation and proprioceptive feedback from joints, tendons, muscles and mechanoreceptors.
This causes a significant increase in the activity along the intact pathways to the cortex, stimulating the production of new synpatic connections [49]. The increased level of motor neuron excitation will also make it easier for weak descending inputs to activate the motor neuron and therefore help to produce a voluntary contraction [5]. When using ES to improve motor function it is often useful to combine muscles to produce a larger pattern of movement, similar to the combination of movements in ADL’s.
Application
Typically there are three main focuses of ES for upper limb:
• elbow extension
• wrist, finger and thumb extension
• reaching actions.
Overall the placement of the electrodes is key to achieving a comfortable, effective movement for the patient. It is important to ask the patient to assist with the movement, however this voluntary effort must not be so great that it causes a rise in spasticity and inhibits the desired movement. The images below indicate the electrode placement and the functional movement they help to produce.
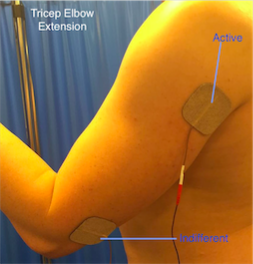
Figure 13. Tricep Elbow Extension
Elbow extension
• The triceps can be activated placing an active electrode over its motor point and the indifferent over the tendon at the elbow.
• Due to the size of the muscle it is useful to use larger electrodes, which may help produce a more effective movement.
• Practicing ‘table polishing’ by sliding the hand over a table using a cloth to reduce friction can be useful.
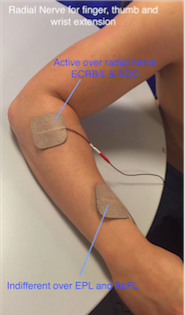
Figure 14. Wrist, finger and thumb extension
Wrist, finger and thumb extension
• This is best achieved by stimulation of the radial nerve, which produces an extensor pattern.
• It is often a problem to get good thumb extension so it is good practice to place the indifferent electrode over the motor points of extensor palmaris longus and abductor palmaris longus, about three fingerbreadths proximal to the wrist.
• If thumb extension is still not good, make this electrode the active, assuming this does not significantly reduce finger and wrist extension.
• Care should be taken to avoid either radial or ulna deviation of the wrist. If there is excessive ulnar deviation, move the active electrode towards the extensor carpi radialis brevis on the radial side of the arm. If radial deviation occurs, move the electrode towards the ulna side and the extensor carpi ulnaris.
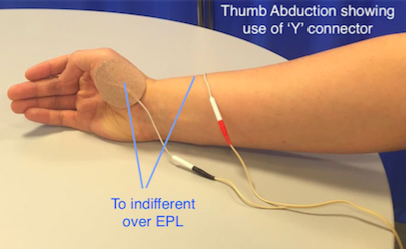
Figure 15. Thumb opposition
Thumb abduction and opposition
• Radial nerve stimulation can be effective at opening the hand but thumb extension alone can leave the thumb in a less than functional position.
• Abduction and opposition can be produced by stimulating the thenar eminence.
• Place the active electrode over the motor point of Abductor poliicis brevis or opponens pollicis and the indifferent over the back of the wrist. To combine this movement with a general extensor pattern it can be useful to use a ‘Y’ connector.
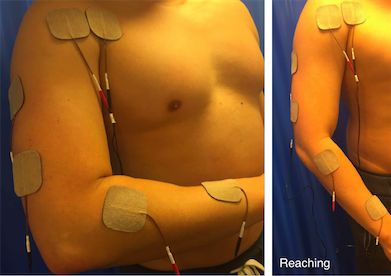
Figure 16. Reaching
Reaching
• It is often useful to combine muscles to produce a gross pattern of movement, similar to the combination movements used in every day life. In this way it may be possible to more effectively re-train function rather than by practicing individual muscle activity.
• Reaching is where finger, thumb and wrist extension from radial nerve stimulation are combined with elbow extension and shoulder flexion by all channels on together.
Dosage and Parameters
There are a wide variety of dosage and parameters in the literature. However, the SSAHPF [4]has been developed to provide guidance for the recommended dosage and parameters for upper limb motor control recovery after stroke. Before selecting ES as a treatment it is important that you are aware of the different dosage and parameter settings and how these will affect the treatment.
A summary of the main dosage and parameters are outlined below:[edit | edit source]
FrequencyIn order to achieve a muscle contraction and minimise patient discomfort and fatigue while maximising clinical benefits has been reported as 12.5Hz [50]. However, it has also been reported that somewhere between 20-50Hz is appropriate [24][51] with lower frequencies required for the upper limb [50].
Pulse amplitude and pulse width
To achieve greater muscle force generation through recruitment of neurons increasingly further from the electrode, pulse amplitude and pulse width may (usually 200-400 micro sec) need to be adjusted [50],[52]. It has been suggested that the intensity frequency and pulse width of electrical current should be adjusted in order to produce a visible contraction. Although there is agreement in this area, there is still variability in application and the final decision will fall to the clinician when addressing the individual patient.
Length of treatment
Common doses and duration of treatments delivered range from 30minutes once per day to one hour three times per day for two weeks to three months [24]although this was not substantiated or justified by the original authors. Hsu [52] randomised 95 participants to dosages of 0, 15, 30, 60 minutes of ES five times per week for four weeks and reported improved recovery in the upper limb with more intensive ES. However, de Kroon [24] suggested that the particular treatment parameters may not in fact be the critical element in the efficacy of ES within their study so it may be that individual patient treatment approaches may be sufficient.
Hsu [52]reported cycles of 10 seconds on 10 seconds off in the first two weeks and 10 seconds on and 5 seconds off in the second two weeks.It has been suggested in a review of ES, that it is the adjustment of these parameters which determines the nature of the evoked action potential response and thus impacts on the amount of muscle force generated as well as patient comfort and safety. [24]
It is important to take into consideration whether the patient has any upper limb tone or spasticity. The dosage and parameters may need to be adjusted for these. Sailsbury [45] suggested that if spasticity is present then a slower stimulation with a longer ramp time may be beneficial.
Evidence base[edit | edit source]
Currently there is a limited amount of evidence to support the use of ES for upper limb motor control. A number of articles have been synthesised and below are their detailed findings.A systematic review by Vafander [29], found that the use of ES does not have any significant benefits over conventional treatment. A number of the articles included focused on early intervention after stroke, and concentrated on methods to measure impairments (spasticity, strength and joint motion), not function or activity. These articles showed positive benefits of ES for motor control, however it is unclear if improvements in muscle activity and joint motion can be translated to improvement in motor function. The studies that found no superiority of ES over a conventional treatment tended to be of higher quality and mostly used methods for measuring function or activity instead of impairment.
Hara [25] found that individuals that receive motor, proprioceptive, and cognitive inputs through the daily use of ES may demonstrate significantly greater improvements in voluntary movement and functional use of the hand and arm.
Another article by McCabe [53] , suggested that for severely impaired stroke survivors with upper limb dysfunction the use of ES combined with motor learning (5 hours per day partial and whole-task practice of complex tasks) helped to improve coordination and functional task performance. However, when analysing the effectiveness of motor learning and ES, compared to motor learning alone or in conjunction with robotics there were no significant differences.
At this stage it is hard to say whether ES is an effective treatment due to the limited literature. Future research is needed with a greater consistency throughout the studies. More studies need to be undertaken with larger sample sizes, the use of ES in early and late stages of rehabilitation after stroke being explored. Furthermore, the use of standardised outcome measures for function and activity will strengthen the generalisabilty for the use of ES for upper limb recovery post stroke.
Conclusion[edit | edit source]
ES is commonly used in clinical practice in lower limbs however the same frequency of use has not been adopted for upper limbs, although loss of upper limb function is a common consequence affecting a large proportion of the stroke population. There is now robust evidence supporting the use of ES for the management of patients with or who are at risk of shoulder subluxation, supporting its use in clinical practice. There is a developing evidence base for the effects of ES in improving motor control of the upper limbs post stroke, and it is the quality of the evidence available that limits ES being recommended for this purpose. Further research should therefore be carried out in this area to determine the true effect of ES for upper limb within this population.
Useful Resources[edit | edit source]
- Allen K; Goodman C. Using Electrical Stimulation: A Guideline for Allied Health Professionals. 2014 http://www.aci.health.nsw.gov.au/__data/assets/pdf_file/0004/211819/Using-Electrical-Stimulation-January-2014.pdf;
- Scottish Stroke AHP Forum. Use of Electrical Stimulation Following Stoke: A Consensus Statement. http://www.chss.org.uk/documents/2014/10/electrical-stimulation-consensus-statement-ssahpf-pdf.pdf;
- Scottish Intercollegiate Guidelines Network. SIGN 118 Management of Patients with stroke: Rehabilitation, Prevention and Management of Complications and Discharge Planning. A national clinical guideline. http://www.sign.ac.uk/pdf/sign118.pdf
- National Institute for Health and Care Excellence. NICE 162 Long- term Rehabilitation after stroke. http://www.nice.org.uk/guidance/cg162;
- Intercollegiate Stroke Working Party. National Clinical Guideline for Stroke Fourth Edition. Homepage of Royal College of Physicians. 2012. https://www.rcplondon.ac.uk/guidelines-policy/stroke-guidelines;
- Ada L, Foongchomcheay A. Efficacy of electrical stimulation in preventing or reducing subluxation of the shoulder after stroke: A meta-analysis. Australian Journal of Physiotherapy. 2002; 48(4): 257-267. http://www.sciencedirect.com/science/article/pii/S0004951414601653;
- Manigandan JB, Ganesh GS, Pattnaik M, Mohanty P. Effect of electrical stimulation to long head of biceps in reducing gleno humeral subluxation after stroke. Neurorehabilitation. 2014;34(1): 245-252. http://eds.a.ebscohost.com/ehost/pdfviewer/pdfviewer?sid=e2743008-f009-4d62-8281-bc2a335d2026%40sessionmgr4004&vid=8&hid=4203;
- McCabe J, Monkiewicz M, Holcomb J, Pundik S, Daly J. Comparison of Robotics, Functional Electrical Stimulation, and Motor Learning Methods for Treatment of Persistent Upper Extremity Dysfunction After Stroke: A Randomised Controlled Trial. Archives of Physical Medicine and Rehabilitation 2015; 96(6): 981-990. http://www.archives-pmr.org/article/S0003-9993(14)01228-3/pdf;
- Hara Y. Neurorehabilitation with new functional electrical stimulation for hemiparetic upper extremity in stroke patients. Journal of Nippon Medical School 2008; 75(1): 4-14. https://www.researchgate.net/publication/5492468_Neurorehabilitation_with_New_Functional_Electrical_Stimulation_for_Hemiparetic_Upper_Extremity'_in_Stroke_Patients;
- Odstock website for the uses of Electrical Stimulation http://www.odstockmedical.com/category/knowledge-base/applications/upper-limb
- Salisbury fes newsletter. 1. Upper Limb Electrical Stimulation Exercises. 2002. http://www.salisburyfes.com/pdfs/upper limb.PDF;
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Stroke Association. State of the Nation: Stroke statistics. https://www.stroke.org.uk/sites/default/files/stroke_statistics_2015.pdf (accessed 26 January 2016).
- ↑ 2.0 2.1 Age UK. Later Life in the United Kingdom. http://www.ageuk.org.uk/Documents/EN-GB/Factsheets/Later_Life_UK_factsheet.pdf?dtrk=true (accessed 8 January 2016).
- ↑ 3.0 3.1 3.2 Pollock A, Farmer SE, Brady MC, Langhorne P, Mead GE, Mehrholz J, van Wijck F. Interventions for improving upper limb function after stroke (Review). http://onlinelibrary.wiley.com/doi/10.1002/14651858.CD010820.pub2/abstract (accessed 8 January 2016).
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 4.18 4.19 4.20 Scottish Stroke AHP Forum. Use of Electrical Stimulation Following Stoke: A Consensus Statement. http://www.chss.org.uk/documents/2014/10/electrical-stimulation-consensus-statement-ssahpf-pdf.pdf (accessed 5 January 2016).
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 Quandt F, Hummel F. The Influence of Functional Electrical Stimulation on Hand Motor Recovery in Stroke Patients: A Review. Experimental and Translational Stroke Medicine 2014; 6:9. http://www.etsmjournal.com/content/6/1/9 (accessed 18 December 2015).
- ↑ Scottish Intercollegiate Guidelines Network. SIGN 118 Management of Patients with stroke: Rehabilitation, Prevention and Management of Complications and Discharge Planning. A national clinical guideline. http://www.sign.ac.uk/pdf/sign118.pdf (accessed 24 January 2016).
- ↑ Foley N, Mehta S, Jutai J, Staines E, Teasell R. Upper Extremity Interventions. http://www.ebrsr.com/sites/default/files/module-10-upper-extremity_final_16ed.pdf (accessed 8 January 2016).
- ↑ Van Wijck F, McBean D, The brain-behaviour relationship: an introduction. In: Applied Neuroscience for the Allied Health Professions. Edinburgh: Churchill Livingstone/Elsevier, 2013. P33-52.
- ↑ Schmidt R, Lee T. Motor control and learning : a behavioral emphasis. Champaign, IL : Human Kinetics, 2011.
- ↑ Butefisch CM, Netz J, Wessling M, Seitz RJ, Homberg V. Remote changes in cortical excitability after stroke. Brain 2003; 126 (2): 470-81. http://brain.oxfordjournals.org/content/126/2/470 (accessed 11 January 2016).
- ↑ Meilink A, Hemmen B, Seelen H, Kwakkel G. Impact of EMG-triggered neuromuscular stimulation of the wrist and finger extensors of the paretic hand after stroke: a systematic review of the literature. Clinical Rehabilitation 2008; 22: 291-305. http://cre.sagepub.com/content/22/4/291.long (accessed 11 January 2016).
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 12.6 Scottish Intercollegiate Guidelines Network. SIGN 118 Management of Patients with stroke: Rehabilitation, Prevention and Management of Complications and Discharge Planning. A national clinical guideline. http://www.sign.ac.uk/pdf/sign118.pdf (accessed 24 January 2016).
- ↑ 13.0 13.1 13.2 13.3 13.4 Robertson V, Ward A, Low J, Reed A. 4th ed. Electrotherapy Explained – Principles and Practice. Edinburgh: Butterworth Heinemann Elsevier. 2006.
- ↑ 14.0 14.1 Neo Stroke Network. Funcitonal Electrical Stimualtion: “To stim or not to stim”. https://www.neostrokenetwork.com/newportal/Portals/0/Education%20Documents/Everything%20Stroke/Rehabilitation/09-Related%20Presentations/Functional%20Electrical%20Stimulation_a%20discussion%20paper%20by%20L%20Taipalus.pdf (accessed 5 January 2016).
- ↑ Bear MF, Connors BW, Paradiso MA. Neuoscience: Exploring the Brain, 3rd ed. Baltimore: Lippincott Williams & Wilkins; 2007.
- ↑ 16.0 16.1 16.2 16.3 Gorman PH, Peckham PH, Functional electrical stimulation in neurorehabilitation. In: Selzer ME, Clarke S, Cohen LG, Miller RH editors. Textbook of Neural Repair and Rehabilitation. United Kingdom: Cambridge University Press, 2014, pp. 120-134.
- ↑ Dobkins BH, Dorsch A. New Evidence for Therapies in Stroke Rehabilitation. Current Atherosclerosis Reports 2013; 15 (6): 1-9. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3679365/ (accessed 5 January 2016).
- ↑ Kawashima N, Popovic MR, Zivanovic V. Effect of Intensive Functional Electrical Stimulation Therapy on Upper-Limb Motor Recovery after Stroke: Case Study of a Patient with Chronic Stroke. Physiotherapy Canada 2013; 65 (1): 20-28. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3563372/ (accessed 8 January 2016).
- ↑ 19.0 19.1 Howlett OA, Lannin, NA, Ada, L, Mckinstry, C. Functional Electrical Stimulation Improves Activity After Stroke: A Systematic Review With Meta-Analysis. Archives of Physical Medicine and Rehabilitation 2015; 96 (5):1-9. http://www.archives-pmr.org/article/S0003-9993(15)00044-1/abstract (accessed 8 January 2016).
- ↑ 20.0 20.1 20.2 20.3 20.4 20.5 Sheffler LR, Chae J. Neuromuscular electrical stimulation in neurorehabilitation. Muscle and Nerve 2007; 35(5): 562-590. http://onlinelibrary.wiley.com/doi/10.1002/mus.20758/abstract;jsessionid=33B6E38FCCF503F611C557F5541E8788.f03t01 (accessed 4 January 2016).
- ↑ Thrasher A, Graham GM, Popovic MR. Reducing muscle fatigue due to functional electrical stimulation using random modulation of stimulation parameters. Artificial Organs 2005; 29 (6): 453-458. http://onlinelibrary.wiley.com/doi/10.1111/j.1525-1594.2005.29076.x/abstract (accessed 17 December 2015).
- ↑ 22.0 22.1 22.2 Ewins D, Durham S, Functional Electrical Stimualtion. In: Watson T editor. Electrotherapy: evidence based practice. Edinburgh: Churchill Livingstone, 2008, pp. 317-326.
- ↑ Popavic DB. Neural Prostheses for Movement Restoration. In: Moore J, Zouridakis G editors. Biomedical Technology and Devices Handbook. United States of America: CRC Press, 2003. p 9-16.
- ↑ 24.0 24.1 24.2 24.3 24.4 24.5 De Kroon JR, Ijzerman MJ, Chae J, Lankhorst GJ, Zilvold G. Relation between stimulation characteristics and clinical outcome in studies using electrical stimulation to improve motor control of the upper extremity in stroke. Journal of Rehabilitation Medicine 2005;37(2): 65-74. http://www.medicaljournals.se/jrm/content/?doi=10.1080/16501970410024190 (accessed 24 January 2016).
- ↑ 25.0 25.1 25.2 Hara Y. Neurorehabilitation with new functional electrical stimulation for hemiparetic upper extremity in stroke patients. Journal of Nippon Medical School 2008; 75(1): 4-14. https://www.researchgate.net/publication/5492468_Neurorehabilitation_with_New_Functional_Electrical_Stimulation_for_Hemiparetic_Upper_Extremity_in_Stroke_Patients (accessed 14 December 2016).
- ↑ 26.0 26.1 26.2 26.3 26.4 26.5 26.6 26.7 National Institute for Health and Care Excellence. Stroke and transient ischaemic attack in over 16s: diagnosis and initial management. https://www.nice.org.uk/guidance/cg68 (accessed 19 December 2015).
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 27.6 27.7 Intercollegiate Stroke Working Party. National Clinical Guideline for Stroke Fourth Edition. Homepage of Royal College of Physicians. 2012. https://www.rcplondon.ac.uk/guidelines-policy/stroke-guidelines (accessed 24 January 2016).
- ↑ World Health Organisation. Towards a common language for function, disability and health: ICF Geneva: WHO. http://www.who.int (accessed 9 January).
- ↑ 29.0 29.1 29.2 29.3 Vafadar AK, Cote JN, Archambault PS. Effectiveness of Functional Electrical Stimulation in Improving Clinical Outcomes in the Upper Arm following Stroke: A Systematic Review and Meta-Analysis. BioMed Research International. 2014;2015(1): 1-14. http://eds.a.ebscohost.com/ehost/pdfviewer/pdfviewer?sid=25c0498f-13ec-4520-8d45-310ed8491799%40sessionmgr4005&vid=0&hid=4203 (Accessed 24 January 2016).
- ↑ Veerbeek JM, Van Wegen E, Van Peppen R, Van Der Wees P, Hendriks E, Rietberg M, Kwakkel G. What Is the Evidence for Physical Therapy Poststroke? A Systematic Review and Meta-Analysis. PLOS ONE 2014; 9(2) http://www.plosone.org (accessed 10 January 2016).
- ↑ Greenhaulgh T. How to read a paper: Getting your bearings (deciding what the paper is about). BMJ 1997 ;315 243-246 http://www.bmj.com (accessed 10 January 2016)
- ↑ Smith R. The Cochrane Collaboration at 20. BMJ 2013 http://www.bmj.com (accessed 10 January 2016)
- ↑ 33.0 33.1 33.2 Allan K, Goodman C. Using Electrical Stimulation A Guideline for Health Professionals. http://www.aci.health.nsw.gov.au/__data/assets/pdf_file/0004/211819/Using-Electrical-Stimulation-January-2014.pdf (accessed 7 January 2016).
- ↑ 34.0 34.1 Carr J, Shepherd RB and Nordholm L nvestigation of a new motor assessment scale for stroke patients. Physical Therapy 1985 65:175-180. http://www.ncbi.nlm.nih.gov/pubmed/3809245 (accessed 7 January 2016)
- ↑ 35.0 35.1 Wolf SL, Thompson PA, Morris DM, Rose DK, Winstein CJ, Taub E, Giuliani C and Pearson SL. Wolf Motor Function Test in Subacute Stroke The EXCITE Trial: Attributes of the Wolf Motor Function Test in Patients with Subacute Stroke Neurorehabilitation and Neural Repair 2005; 19 (3):194-205. http://nnr.sagepub.com/content/19/3/194.long?hwshib2=authn%3A1453766034%3A20160124%253Adbcb3854-3ac8-458b-a4c3-0ae27a7bf028%3A0%3A0%3A0%3AVr%2B4aUt5CRoWfs4DNZJ3Tw%3D%3D (accessed 7 January 2016).
- ↑ 36.0 36.1 36.2 36.3 36.4 Ada L, Foongchomcheay A. Efficacy of electrical stimulation in preventing or reducing subluxation of the shoulder after stroke: A meta-analysis. Australian Journal of Physiotherapy. 2002; 48(4): 257-267. http://www.sciencedirect.com/science/article/pii/S0004951414601653 (Accessed 24 January 2016).
- ↑ 37.0 37.1 Manigandan JB, Ganesh GS, Pattnaik M, Mohanty P. Effect of electrical stimulation to long head of biceps in reducing gleno humeral subluxation after stroke. Neurorehabilitation. 2014;34(1): 245-252. http://eds.a.ebscohost.com/ehost/pdfviewer/pdfviewer?sid=e2743008-f009-4d62-8281-bc2a335d2026%40sessionmgr4004&vid=8&hid=4203 (Accessed 24 January 2016).
- ↑ 38.0 38.1 Linn SL, Granat MH, Lees KR. Prevention of Shoulder Subluxation After Stroke With Electrical Stimulation. Strokeahajournals.1999;30(1): 963-968. http://stroke.ahajournals.org/content/30/5/963.full.pdf+html (Accessed 24 January 2016).
- ↑ Fil A, Armutlu K, Atay AO, Kerimoglu U, Elibol B. The effect of electrical stimulation in combination with Bobath techniques in the prevention of shoulder subluxation in acute stroke patients. Clinical Rehabilitation. 2011;25(1): 51-59. http://cre.sagepub.com/content/25/1/51.full.pdf+html (Accessed 24 January 2016).
- ↑ Price CIM, Pandyan AD. Electrical stimulation for preventing and treating post stroke shoulder pain: a systematic Cochrane review. Clinical Rehabilitation. 2001;15(1): 5-19. http://cre.sagepub.com/content/15/1/5.long (Accessed 24 January 2016).
- ↑ Church C, Price C, Pandyan A, Huntley S, Curless R, Rodgers H. Randomized controlled trial to evaluate the effect of surface neuromuscular stimulation to the shoulder after acute stroke. Stroke. 2006; 37(10), pp. 2995-3001. http://stroke.ahajournals.org/content/37/12/2995.full.pdf+html (Accessed 14 January 2016).
- ↑ 42.0 42.1 Foley N, Mehta S, Jutai J, Staines E, Teasell R. Upper Extremity Interventions. http://www.ebrsr.com/sites/default/files/module-10-upper-extremity_final_16ed.pdf (accessed 8 January 2016).
- ↑ Paci M, Nanneti L, Rinaldi, L A. Glenohumeral subluxation in hemiplegia: An over view. Journal of Rehabilitation Research Development. 2005. 42(4): 557-568. http://www.rehab.research.va.gov/jour/05/42/4/pdf/paci.pdf (Accessed 18 January 2016).
- ↑ Kobayashi H, Onishi H, Ihashi K, Yagi R, Handa Y. Reduction in subluxation and improved muscle function of the hemiplegic shoulder joint after therapeutic electrical stimulation. Journal of Electromyography and Kinesiology. 1999;9(5): 327-336. http://www.sciencedirect.com/science/article/pii/S1050641199000085 (Accessed 24 January 2016).
- ↑ 45.0 45.1 45.2 45.3 45.4 Salisbury fes newsletter. 1. Upper Limb Electrical Stimulation Exercises. http://www.salisburyfes.com/pdfs/upper limb.PDF (accessed 24 January 2016).
- ↑ 46.0 46.1 http://www.salisburyfes.com/pdfs/products%20and%20services.PDF (accessed 26 October 2015)
- ↑ Baker LL, Parker K. Neuromuscular Electrical Stimulation of the Muscles Surrounding the Shoulder. Journal of the American Physical Therapy Association. 1986;66(12): 1930-1937. http://ptjournal.apta.org/content/66/12/1930.long (Accessed 24 January 2016).
- ↑ Chantraine A, Baribeault A, Uebelhart D, Gremion G. Shoulder pain and dysfunction in hemiplegia: Effects of functional electrical stimulation. Archives of Physical Medicine and Rehabilitation. 1999;80(3): 328-331. http://www.sciencedirect.com/science/article/pii/S0003999399901466 (Accessed 24 January 2016).
- ↑ Taylor P. Referral Criteria OML Learning Through Technology Precautions.fckLRhttp://www.odstockmedical.com/sites/default/files/referral-criteria_0.pdf (accessed 7 January 2016).
- ↑ 50.0 50.1 50.2 Sheffler LR, Chae J. Neuromuscular electrical stimulation in neurorehabilitation. Muscle and Nerve 2007; 35(5): 562-590. http://onlinelibrary.wiley.com/doi/10.1002/mus.20758/abstract;jsessionid=33B6E38FCCF503F611C557F5541E8788.f03t01 (accessed 4 January 2016).
- ↑ Sujith OK. Functional electrical stimulation in neurological disorders European Journal of Neurology 2008 15(5): 437–444. http://onlinelibrary.wiley.com/doi/10.1111/j.1468-1331.2008.02127.x/abstract (accessed 8 January 2016)
- ↑ 52.0 52.1 52.2 Hsu S, Hu M, Luh J, Wang Y, Yip P, Hsieh C. Dosage of neuromuscular electrical stimulation: is it a determinant of upper limb functional improvement in stroke patients? Journal of Rehabilitation Medicine 2012; 44(2): 125 -130. http://www.medicaljournals.se/jrm/content/?doi=10.2340/16501977-0917&html=1 (accessed 8 January 2016).
- ↑ McCabe J, Monkiewicz M, Holcomb J, Pundik S, Daly J. Comparison of Robotics, Functional Electrical Stimulation, and Motor Learning Methods for Treatment of Persistent Upper Extremity Dysfunction After Stroke: A Randomised Controlled Trial. Archives of Physical Medicine and Rehabilitation 2015; 96(6): 981-990.fckLRhttp://www.archives-pmr.org/article/S0003-9993(14)01228-3/pdf (accessed 20 November 2015).