Assessment and Management of Cervicogenic Headaches Post Traumatic Injury
Top Contributors - Liam Cook, Cindy John-Chu, Tyler Harriott, Khloud Shreif, Grace Cain, Angeliki Chorti, Nupur Smit Shah, Adam Townsend, Trishia Baharani and Carina Therese Magtibay
Introduction[edit | edit source]
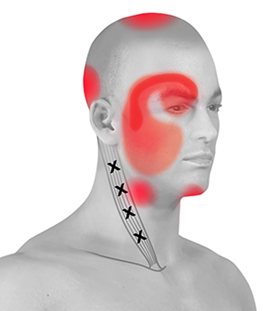
A cervicogenic headache (CGH) is a chronic headache that presents as unilateral pain starting at the neck and is perceived in one or more regions of the head/face. [2]
A common cause of CGH is trauma that has led to the dysfunction of the neck. Types of trauma that can cause CGH most commonly include: whiplash caused by car accidents, contact sport injuries causing neck injury, and falls leading to upper cervical structural damage. [3]
Mechanisms of injury caused by these types of trauma may include structural damage, development of myofascial pain and interaction of the trigeminal nociceptive system with the occipital nerves. There are also emotional and psychological factors to consider post-trauma which can impact rehabilitation and healing outcomes. [4]
Epidemiology[edit | edit source]
Studies focused solely on CGH post-trauma is limited. Current research states:
- Prevalent in 30 to 44 year-olds
- Accounts for 1-4% of headaches
- Equally prevalent between males and females
- Onset is said to be the early 30s and diagnosis is 49.4 years old (due to seeking medical attention late)
- Compared to other headache patients, CGH has peri cranial muscle tenderness on the painful side
Relevant Anatomy[edit | edit source]
The cervical spine is made up of 7 vertebrae (C1 to C7), cranial nerves (C1 to C8), muscles and ligaments. Trauma to any of these structures may cause an CGH. [3]
Joints[edit | edit source]
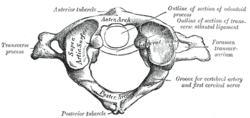
- C1 (atlas) supports the skull and articulates superiorly with the occiput to form the atlanto-occipital joint. This allows for 33% of flexion and extension at the C-spine
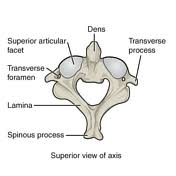
- C2 (axis) articulates with the atlas to form the atlantoaxial joint. Its main function is to provide 60% of cervical rotation
- C3-C7 are similar to one another and make up the rest of the movement in the C-spine
Muscles innervated by C1-C3[edit | edit source]
- Rectus capitis and lateralis
- Longus capitis
- Prevertebral muscle
- Sternocleidomastoid
- Levator scapulae
- Upper trapezius
- Scalenus Medius
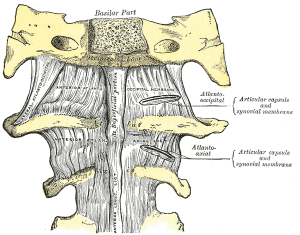
Ligaments (C1-C3)[edit | edit source]
- Apical
- Alar
- Cruciform
- Anterior/posterior longitudinal
- Ligamentum flavum
- Interspinous
- Nuchal ligament
- Transverse ligament
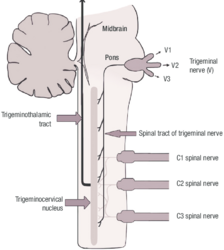
Trigeminocervical nucleus[edit | edit source]
- Located at the lower brainstem near the upper cervical spine.
- Vertical cluster of cell bodies in the medullary region.
- Within this area a convergence of the trigeminal sensory nerves and the C1 - C3 spinal nerves occurs.
- Sensory nerve fibers in the descending tract of the trigeminal nerve are believed to interact with sensory fibers from the upper cervical roots.
- Converged information then passes on to the somatosensory cortex.
Aetiology[edit | edit source]
A CGH is believed to be pain referring to dysfunction of cervical structures innervated by cervical nerves C1, C2, and C3. Trauma causing damage to joints, intervertebral discs, ligaments, and muscles can all be a source of a CGH. There is limited evidence to suggest that the lower cervical spine plays a role in referred pain causing a CGH. [7]
The most common cause of CGH has been attributed to whiplash injury. It is believed that this type of injury accounts for up to 53% of all CGH, with 15.2% of patients having a headache lasting longer 42 days and 4.6% developing chronic daily headaches. Research also suggests that around 50% of CGH originate from the C2-C3 zygapophysial joint. [8]
Pathophysiology[edit | edit source]
Activation of the trigeminal nerve and its connections are well established in regard to headaches. In terms of post-traumatic CGH, the activation occurs from disruption of structures innervated by spinal nerves C1-C3. [9]
Efferent innervation converges onto the second-order neuron at the dorsal horn of C1/C2. At the same time, the trigeminal nerve will send sensory information from the face. The trigeminal nerve converges in the second-order neuron in at the same spinal segment as C1/C2. This sensory information will be sent to the trigeminocervical nucleus within the brain stem. When afferent nociception stimulus from the upper cervical structures travels to the trigeminocervical nucleus, the information sent to the somatosensory cortex becomes corrupt. This is due to the higher number of nociceptive efferent nerves in the face compared to the upper cervical spine. The convergence within the trigeminocervical nucleus means the brain will process this as an error, as it will assume the pain originates from the area with the higher area of nociceptive innervation. As a result, pain originating from the upper C-spine will be referred to the head and present as a CGH. Lower pain thresholds caused by emotional changes and aseptic inflammation can increase CGH pain levels further. [2]
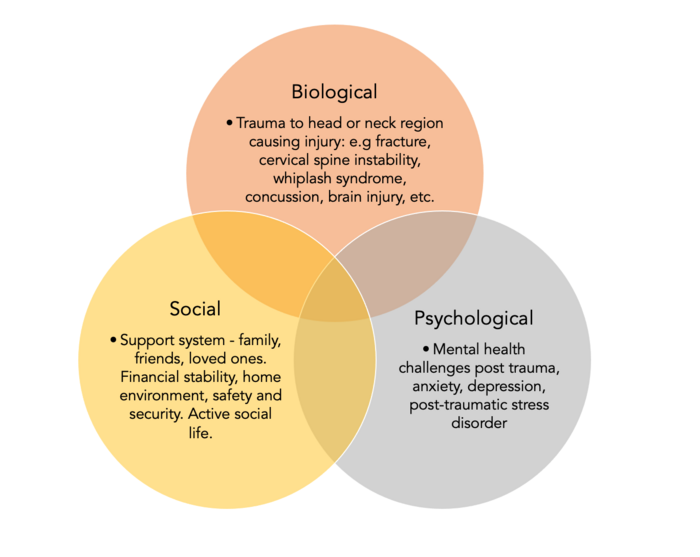
The Biopsychosocial Model[edit | edit source]
The biopsychosocial model, developed by George Engel in 1977 displays the complex relationship between biological, psychological, and social factors and the impact which it has on a patient's pain experience. It guides clinicians to develop a holistic approach to patient care, recognising that factors beyond biological changes alone may cause patients to feel pain, and highlights the importance of a detailed biopsychosocial assessment before treating this condition. [11]
Biological factors[edit | edit source]
Post-traumatic head injury may cause cervicogenic headaches (CGH) and they are often a common complication. [12] As mentioned above, structural damage, development of myofascial pain, and interaction of the trigeminal nociceptive system with the occipital nerves fall under the biological factors relating to this condition. It is vital when assessing and managing patients with CGH that we investigate the biological causes through thorough subjective and objective testing.
Psychological factors[edit | edit source]
Traumatic brain injuries can be particularly emotionally traumatic. Post-traumatic stress disorder is described as re-experiencing symptoms, avoidance behavior, and alterations in cognition and mood resulting from traumatic events. Although the mechanism of the association remains unclear, it is thought that TBI damages neural circuits which regulate fear responses, potentially explaining the heightened paranoia or stress. [13] Patients who have mild TBI are more likely to suffer from psychological symptoms and headaches compared with severe TBI (involving a loss of consciousness), suggesting a link between memory of trauma and headaches.
Some patients who have had a mild TBI misinterpret the clinical advice about their injury which often leads to catastrophising and fear avoidance. [14] Catastrophising, fear-avoidance, and depressive symptoms positively correlate with post-TBI symptoms, therefore during the treatment of patients that display these psychological symptoms – more emphasis on advice and education around pain is necessary.[15]
Patients with cervicogenic headaches often show lower spinal postural variability. When intrinsic variables such as stress and anxiety are higher, evidence suggests that this decreases the postural variability of the upper cervical spine, therefore putting emphasis on the effect that psychological symptoms have on cervicogenic headache symptom severity. [16]
Social factors[edit | edit source]
The social impacts of CGH are problematic, below are the possible social impacts of CGH: [17]
- Work absenteeism
- Decreased productivity
- Decreased ability to complete activities of daily living
- Reduced leisure time
- Personal relationships
Social commitments are the fundamental elements of a patient's quality of life; with the inability to contribute to what a patient values, comes detrimental effects psychologically, and as mentioned, this can alter the patient's pain experience. Person-centered assessment and management of CGH are vital to aid return to what they value and care for.
Clinical Presentation[edit | edit source]
- Restricted ROM in the cervical spine [2]
- Head pain with neck movement or awkward head positioning [18]
- Tenderness of the upper cervical or occipital region [19]
- Varying duration of episodes [18]
- Fluctuating, continuous pain [18]
- Pain is unilateral without side shift [2]
- Moderate to severe pain, not excruciating [2]
- Weakness in the deep neck flexors [3]
- Potential neck, shoulder or arm pain ipsilaterally [18]
- Confirmatory evidence by diagnostic anesthetic blockades [18]
- Less likely to present with autonomic symptoms, however still may be present e.g. vomiting, nausea, photophobia or phonophobia [2] [18]
Red Flags[edit | edit source]
Screening for red flags is crucial when assessing patients who have a cervical injury following trauma. It is important to identify and rule out serious pathology.
- Cervical fractures.
- Intracranial pathology.
- Cervical instability.
- Cervical myelopathy.
- Vascular pathologies
Red flags for headaches post-trauma that will require further investigation: [20]
- New severe or unexpected headache.
- Progressive or persistent headache, or headache that has changed dramatically.
- Comorbidities such as current or past malignancy.
- Current or recent pregnancy.
Associated features such as:
- Fever, impaired consciousness, seizure, neck pain/stiffness, or photophobia.
- New focal neurological deficit, cognitive impairment, and/or altered consciousness, personality change.
- Atypical aura (duration >60 mins, motor weakness involvement, diplopia, one sided visual symptoms, or decreased balance)
- Papilloedema.
- Dizziness.
- Visual disturbance.
- Vomiting.
These may be indicative of CNS infection, malignancy, intracranial pathologies such as subdural hematoma, stroke, or vascular pathologies such as giant cell arteritis. See this physiopedia page for more insight.
Differential Diagnosis[edit | edit source]
Post-traumatic headaches can present with similarities of symptoms indicative of primary and secondary type headaches so it is important to differentiate between these. [21] See headaches and cervicogenic headaches.
| Migraine | Tension Headache | Cluster | |
|---|---|---|---|
| Gender ratio(M:F) | 1:3 | 5:4 | 3:1 |
| Age of onset | 15-55 | 25-30 | 28-30 |
| Prevelance | 18%F- 6%M | 30%-> 78% | 0.9% |
| Quality | Throbbing | Non-throbbing | Stabbing- Sharp |
| Intensity | Moderate to sever | Mild to moderate | Severe to very severe |
| Location | Unilateral | Bilateral | Unilateral |
| Duration of attack | 4-72h | 30 min- 7 days | 15-180 min |
| Symptoms | Nausea, vomiting, photophobia
phonophobia |
photophobia
phonophobia |
Autonomic dysfunction |
| Triggers | Physical activity | Stress | Laying down, or sleep |
Examination[edit | edit source]
A comprehensive assessment needs to be undertaken when a patient presents with neck pain and headache post-trauma. This will include a full subjective and objective assessment of the cervical spine.
Subjective Assessment[edit | edit source]
It is important to rule out serious pathology and red flags for patients who have had trauma to the neck. In order to do this, a thorough subjective history should be taken.
To get a detailed subjective history the patient should be asked about the trauma in more detail. For example:
- History of trauma (onset, previous trauma, etc)
- Mechanism of the injury caused by the trauma
- Change in symptoms since the trauma occurred (i.e., new or worsening symptoms)
To rule out cervical fracture you would complete the Canadian C-Spine Rule.
Check out the assessment section of this Physiopedia page for further investigation of cervicogenic headaches.
Objective Assessment[edit | edit source]
Trauma to the neck may cause damage to the cranial nerves. A neurological assessment may be completed if the patient presents with paraesthesia. A cranial nerve assessment may be completed as part of the comprehensive assessment if patients present with neurological symptoms in the face and neck and alterations in sensory and motor function. See Cranial Nerves.
Once serious pathologies have been ruled out, physical examination for cervicogenic headaches post-trauma should assess your usual objective assessment with a focus on neck ROM and strength, passive accessory intervertebral movements (PAIVMs) tests and the cervical flexion-rotation test. [23]
Cervical flexion-rotation test:
In the supine position passively but maximally flex the cervical spine and passively rotate left and right. Reported range of motion is determined by the onset of pain or firm resistance encountered by the therapist. The therapist should visually estimate the rotation range of motion. A visually estimated range reduced by 10° or more from the normal range of 44° is indicative of a positive test. [24]
Vestibular and Oculomotor System:
A patient may present with vestibular and oculomotor dysfunction-related symptoms following mild traumatic brain injury such as concussion. These symptoms may include: [25]
- Dizziness
- Nausea
- Movement related blurry vision
- Headache
- Brain fog
- Unsteadiness
- Fatigue and eyestrain
This may warrant an examination using the Vestibular Ocular Motor Screening (VOMS) Tool to measure the extent of symptoms against tests including smooth pursuits, horizontal and vertical saccades, convergence, horizontal and vertical VOR, and visual motion sensitivity. See VOMS Assessment for further detail.
Other tests for concussion can be found assessment and management of concussion|here.
Outcome Measures[edit | edit source]
- Neck Disability Index (NDI)
- In cervicogenic headache, NDI showed excellent reliability (ICC = 0.92; [95 % CI: 0.46–0.97]) [27]
- Numerical Pain Rating Scale (NPRS)
- In cervicogenic headache, NPRS showed moderate reliability (ICC = 0.72; [95 % CI: 0.08–0.90])[27]
Both NDI and NRS Demonstrates good construct validity at 1 week, 4 weeks and 3 months (p=0.0001) in cervicogenic headache [27].
Management: A Multi-disciplinary Approach [edit | edit source]
The evidence suggests a multi-disciplinary biopsychosocial model in order to effectively treat and manage post-traumatic cervicogenic headaches [28] Post-traumatic cervicogenic headaches are multi-dimensional and often part of the larger persistent post-traumatic symptom picture.
Mental Health[edit | edit source]
Clinical psychologists and neuropsychologists are key members of the multidisciplinary team when treating post-traumatic cervicogenic headaches. Symptoms of anxiety and depression are tragically common post-traumatic injuries. [28] This is vital to consider when planning a treatment model for a patient with post-traumatic injury to the head and/or neck. Depression and anxiety have been proven to continue to increase negative symptoms and worsened the prognosis in headache disorders. Mental health challenges paired with ongoing pain are linked with poor sleep quality, deficient nutrients, decreased exercise, pain catastrophising post trauma, emotional distress, and post-traumatic stress disorder are linked with persistent post-traumatic cervicogenic symptoms. [29]
In alignment with a holistic management approach, cognitive behavioural therapy has been suggested to aid with post-traumatic cervicogenic symptoms and the burden it carries. A journal from Brain Injury, by Gurr & Coetzer explore the effectiveness of CBT for post-traumatic headaches. This study hones into the importance of psychological factors presenting as highly influential in increasing or decreasing pain. [30] In this study, Forty-five participants attended the Brain Injury Service for the initial three weekly relaxation group sessions. Following this, participants received 6 fortnightly 30-minute individual therapy sessions with one follow-up. Interventions included: progressive muscle relaxation combined with imager, psycho-education, cognitive-behavioral strategies, life management, maintenance, and relapse prevention. The findings present with over half of participants of the brain therapy programme reporting a significant decrease in headache symptoms and disability of function, as well as intensity, frequency per month, and pain levels. Although the inclusion criteria do not specify cervicogenic headaches within the population’s diagnosis, the mechanism of injury of a cervicogenic headache discussed is synonymous with post-traumatic headache: a new onset of head and neck pain following a traumatic injury (road traffic accident, sporting injury, fall). [30]
Graded Exposure to Exercise[edit | edit source]
The sport-related concussion is defined as a mild traumatic brain injury caused by direct force to the head causing a degree of functional impairments. Cervicogenic headaches are a secondary type of headache and can occur in response to a sporting injury to the head. Prevalence of post-concussion symptoms has been reported in previous studies with 90-92.2% of athletes experiencing headaches, 90% experiencing neck pain, and 68.9% experiencing dizziness. [31] In the 2021 journal from Sports Health clinically reviews the effect of exercise for persistent post-concussive symptoms. [32] The majority of studies show that spontaneous physical activity is safe after SRC and that sub-symptom threshold aerobic exercise safely speeds up recovery after SRC and reduces symptoms in those with PPCS. [32]Exercise tolerance can safely be assessed using graded exertion test protocols within days of injury, and the degree of early exercise tolerance has diagnostic and prognostic value. It is key to note clinicians must provide patients with specific instructions regarding acceptable activity levels post head and neck injury as intense physical and cognitive activity can exacerbate symptoms. A method to monitor exercise tolerance is for concussed patients to calculate their age-appropriate maximum heart rate using the Karvonen equation (maximum heart rate HRmax = 220 – age in years) and begin exercising at 50% of their maximum using a consumer-approved heart rate monitoring device.(13) Patients should aim to exercise without symptomatic experiences (sweating, heavy breathing, panting, etc.) Graded exposure to walking, stationary cycling, and swimming is recommended for the optimal graded return to aerobic exercise. [33]
Moreover, further evidence supports aerobic exercise to aid in diminishing the burden of a cervicogenic headache with the proposed mechanisms of upregulation of brain-derived neurotropic factor BDNF, improved neurovascular regulation, and modulation of pain. [33]
Pharmacological Management[edit | edit source]
Table 2: Examples of pharmacological Treatments
| Pharmacological Treatment: | Purpose: |
|---|---|
| Over the counter medication (i.e. NSAIDs) | Pain relief; should not be started until after first 3 days due to the risk of delayed bleeding [34] |
| Peripheral nerve blocks | To treat persistent post traumatic CGHs; decrease in afferent feedback to trigeminal nucleus caudalis, decreasing nociceptive transmission [35] |
| Botox injections | Induces muscle relaxant, encouraging reduced tone and decreased response to nerve stimulation |
| Trigger point injections | Alleviate head and neck pain; most commonly injected into trapezius, sternocleidomastoid, temporalis, and levator scapulae [36] |
| Triptan medications | Treat migraines |
Physiotherapy Management[edit | edit source]
Conflicting evidence remains for the efficacy of physiotherapy management post traumatic CGHs. In accordance with the biopsychosocial model of post traumatic CGH (figure 8), patient education with detailed explanation of the condition; support of normal movement; avoiding immobilisation; resumption of work and targeted physiotherapy have been suggested. [37] An update on the management of post traumatic headache concluded intense physiotherapy is not superior to standard therapy with simple patient education, hence cannot be recommended when considering cost-benefit ratios.[37] The Managing Injuries of Neck Trial (MINT) [38] investigated the effects of ‘standard treatment’, including simple treatment of symptoms and brief information about the condition was compared to active treatment and detailed information given in a Whiplash information booklet. Patients were able to report back after 3 weeks if showing unsatisfactory symptom improvement and randomised into a group of 6 physical therapy sessions including manual therapy, soft tissue techniques, endurance training, and behavioral techniques, or one physical therapy session with a refresh of advice. The groups showed no significant difference in the primary outcome and neck disability index (NDI). [38]
The PROMISE study [39]compared extensive physical therapy including tailored exercise programmes over 12 weeks (cervical spine exercises; posture re-education; sensorimotor exercise; manual therapy) to advice given in booklet form, with minimally assisted exercises in a 30-minute session. The exercises programme was not more effective than advice alone for red education of pain. [39]
Contradictory evidence shows patients randomised to additional manual therapy techniques (including temporomandibular region) experienced statistically significant decrease in headache intensity at 3 and 6 months when compared to usual care of just manual therapy for cervical spine region.[40] A further study with patients experiencing CGHs and cervical trigger points underwent manual therapy techniques or simulated manual therapy. The treatment group experienced increased cervical range of motion and a greater reduction in headache intensity.[41]
Overall, more conclusive evidence with larger sample sizes is required to make a better judgment about the efficacy and significance of physical therapy interventions in the management of CGHs post-trauma.
Summary of Management[edit | edit source]
Table 3: Summary of Treatments
| Pharmacological: | Non-Pharmacological: |
|---|---|
| Muscle relaxants (cervical spasms) | Physiotherapy (upper back and neck) |
| Trigger Point Injections | Acupuncture |
| Anti-epileptics (e.g. gabapentin, topiramate) | Greater occipital neurolysis/ neuroectomy |
| Tricyclic Antidepressents (e.g. amitriptyline) | Cognitive Behavioural Therapy |
| Serotonin-norepinephrine reuptake inhibitor (e.g. venlafaxine) | Massage |
References[edit | edit source]
- ↑ Smith, J., 2021. Cervicogenic Headache. [Online] Available at: https://www.jeremysmithmd.com/cervicogenic-headache-orthopaedic-spine-surgeon-orange-irvine-ca.html [Accessed 12 05 2022].
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Al Khalili Y, Ly N, Murphy PB. Cervicogenic Headache. In: StatPearls [Internet] 2022 Mar 9. Treasure Island (FL): StatPearls Publishing
- ↑ 3.0 3.1 3.2 Page P. Cervicogenic headaches: an evidence-led approach to clinical management. Int J Sports Phys Ther. 2011 Sep;6(3):254-66.
- ↑ Packard RC. The relationship of neck injury and post-traumatic headache. Curr Pain Headache Rep. 2002 Aug;6(4):301-7.
- ↑ 5.0 5.1 5.2 Kaiser JT, Reddy V, Lugo-Pico JG. Anatomy, Head and Neck, Cervical Vertebrae. 2021 Aug 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing
- ↑ 6.0 6.1 Fiester P, Rao D, Soule E, Orallo P, Rahmathulla G. Anatomic, functional, and radiographic review of the ligaments of the craniocervical junction. Journal of Craniovertebral Junction & Spine. 2021 Jan;12(1):4.
- ↑ Landén Ludvigsson M, Peterson G, Widh S, Peolsson A. Exercise, headache, and factors associated with headache in chronic whiplash: Analysis of a randomized clinical trial. Medicine (Baltimore). 2019 Nov;98(48):e18130.
- ↑ Becker WJ. Cervicogenic headache: evidence that the neck is a pain generator. Headache. 2010 Apr;50(4):699-705.
- ↑ Blumenfeld A, Siavoshi S. The Challenges of Cervicogenic Headache. Curr Pain Headache Rep. 2018 Jun 13;22(7):47.
- ↑ Physiotutors. The Trigeminal Nerve's Role in Cervicogenic Headache | Referred Pain . Available from: http://www.youtube.com/watch?v=ZAmhCiA0Eio[last accessed 22/8/2022]
- ↑ Tripathi A, Das A, Kar SK. Biopsychosocial Model in Contemporary Psychiatry: Current Validity and Future Prospects. Indian J Psychol Med. 2019 Nov 11;41(6):582-585.
- ↑ Riechers RG, Walker MF, Ruff RL. Post-traumatic headaches. Handbook of Clin Neurol 128; 567–578.
- ↑ Howlett JR, Nelson LD, Stein MB. Mental Health Consequences of Traumatic Brain Injury. Biol Psychiatry. 2022 Mar 1;91(5):413-420.
- ↑ Leeuw M, Goossens ME, Linton SJ, Crombez G, Boersma K, Vlaeyen JW. The fear-avoidance model of musculoskeletal pain: current state of scientific evidence. J Behav Med. 2007 Feb;30(1):77-94.
- ↑ Wijenberg ML, Stapert SZ, Verbunt JA, Ponsford JL, Van Heugten CM. Does the fear avoidance model explain persistent symptoms after traumatic brain injury?. Brain injury. 2017 Oct 15;31(12):1597-604.
- ↑ Mingels S, Dankaerts W, van Etten L, Bruckers L, Granitzer M. Spinal postural variability relates to biopsychosocial variables in patients with cervicogenic headache. Sci Rep. 2021 Jul 2;11(1):13783.
- ↑ Diener I. The impact of cervicogenic headache on patients attending a private physiotherapy practice in Cape Town. South African Journal of Physiotherapy. 2001 Feb 1;57(1):35.
- ↑ 18.0 18.1 18.2 18.3 18.4 18.5 Blumenfeld A, Siavoshi S. The challenges of cervicogenic headache. Current Pain and Headache Reports. 2018 Jul;22(7):1-5.
- ↑ Hall T, Briffa K, Hopper D. Clinical evaluation of cervicogenic headache: a clinical perspective. Journal of Manual & Manipulative Therapy. 2008 Apr 1;16(2):73-80.
- ↑ National Institute for Health and Care Excellence. Scenario: Headache-Diagnosis. Available from: https://cks.nice.org.uk/topics/headache-assessment/diagnosis/headache-diagnosis/ (accessed 07 July, 2022).
- ↑ Aljaaf AJ, Mallucci C, Al-Jumeily D, Hussain A, Alloghani M, Mustafina J. A study of data classification and selection techniques to diagnose headache patients. In Applications of Big Data Analytics 2018 (pp. 121-134). Springer, Cham.
- ↑ Geeky Medics. Cranial Nerve Examination - OSCE Guide (New Version) . Available from: http://www.youtube.com/watch?v=sJBpai74tlU [last accessed 26/8/2022]
- ↑ Rubio-Ochoa J, Benítez-Martínez J, Lluch E, Santacruz-Zaragozá S, Gómez-Contreras P, Cook CE. Physical examination tests for screening and diagnosis of cervicogenic headache: A systematic review. Man Ther. 2016 Feb;21:35-40
- ↑ Hall T, Briffa K, Hopper D, Robinson K. Long-term stability and minimal detectable change of the cervical flexion-rotation test. J Orthop Sports Phys Ther. 2010 Apr;40(4):225-9.
- ↑ Kaae, C., Cadigan, K., Lai, K. and Theis, J. (2022). Vestibulo-ocular dysfunction in mTBI: Utility of the VOMS for evaluation and management – A review. NeuroRehabilitation, [online] 50(3), pp.279–296. doi:10.3233/NRE-228012.
- ↑ ImPACT Applications. VOMS How-To Video. Available from: http://www.youtube.com/watch?v=yQI2HRXCEs8[last accessed 26/8/2022]
- ↑ 27.0 27.1 27.2 Young IA, Dunning J, Butts R, Cleland JA, Fernández-de-Las-Peñas C. Psychometric properties of the Numeric Pain Rating Scale and Neck Disability Index in patients with cervicogenic headache. Cephalalgia. 2019 Jan;39(1):44-51.
- ↑ 28.0 28.1 Kamins J. Models for treating post-traumatic headache. Current Pain and Headache Reports. 2021 Aug;25(8):1-9.
- ↑ Silverberg ND, Iverson GL, Panenka W. Cogniphobia in mild traumatic brain injury. Journal of neurotrauma. 2017 Jul 1;34(13):2141-6.
- ↑ 30.0 30.1 Gurr B, Coetzer BR. The effectiveness of cognitive-behavioral therapy for post-traumatic headaches. Brain Injury. 2005 Jul 1;19(7):481-91.
- ↑ Steiner TJ, Stovner LJ, Jensen R, Uluduz D, Katsarava Z. Migraine remains second among the world’s causes of disability, and first among young women: findings from GBD2019. The Journal of Headache and Pain. 2020 Dec;21(1):1-4.
- ↑ 32.0 32.1 Haider MN, Bezherano I, Wertheimer A, Siddiqui AH, Horn EC, Willer BS, Leddy JJ. Exercise for sport-related concussion and persistent postconcussive symptoms. Sports health. 2021 Mar;13(2):154-60.
- ↑ 33.0 33.1 Tiwari D, Goldberg A, Yorke A, Marchetti GF, Alsalaheen B. Characterization of cervical spine impairments in children and adolescents post-concussion. International journal of sports physical therapy. 2019 Apr;14(2):282.
- ↑ Conidi, F. (2017). Aches and Trauma: Understanding Post-Traumatic Headache, Part 2: Management and Treatment. [online] Practical Neurology. Available at: https://practicalneurology.com/articles/2017-sept/aches-and-trauma-understanding-post-traumatic-headache-part-2-management-and-treatment.
- ↑ Labastida-Ramírez A, Benemei S, Albanese M, D’Amico A, Grillo G, Grosu O, Ertem DH, Mecklenburg J, Fedorova EP, Řehulka P, Di Cola FS. Persistent post-traumatic headache: a migrainous loop or not? The clinical evidence. The Journal of Headache and Pain. 2020 Dec;21(1):1-5.
- ↑ Robbins MS, Kuruvilla D, Blumenfeld A, Charleston IV L, Sorrell M, Robertson CE, Grosberg BM, Bender SD, Napchan U, Ashkenazi A. Trigger point injections for headache disorders: expert consensus methodology and narrative review. Headache: The Journal of Head and Face Pain. 2014 Oct;54(9):1441-59.
- ↑ 37.0 37.1 Obermann M, Naegel S, Bosche B, Holle D. An update on the management of post-traumatic headache. Therapeutic Advances in Neurological Disorders. 2015 Nov;8(6):311-5.
- ↑ 38.0 38.1 Lamb SE, Gates S, Williams MA, Williamson EM, Mt-Isa S, Withers EJ, Castelnuovo E, Smith J, Ashby D, Cooke MW, Petrou S. Emergency department treatments and physiotherapy for acute whiplash: a pragmatic, two-step, randomised controlled trial. The Lancet. 2013 Feb 16;381(9866):546-56.
- ↑ 39.0 39.1 Michaleff ZA, Maher CG, Lin CW, Rebbeck T, Jull G, Latimer J, Connelly L, Sterling M. Comprehensive physiotherapy exercise programme or advice for chronic whiplash (PROMISE): a pragmatic randomised controlled trial. The Lancet. 2014 Jul 12;384(9938):133-41.
- ↑ von Piekartz H, Lüdtke K. Effect of treatment of temporomandibular disorders (TMD) in patients with cervicogenic headache: a single-blind, randomized controlled study. Cranio®. 2011 Jan 1;29(1):43-56.
- ↑ Bodes-Pardo G, Pecos-Martín D, Gallego-Izquierdo T, Salom-Moreno J, Fernández-de-Las-Peñas C, Ortega-Santiago R. Manual treatment for cervicogenic headache and active trigger point in the sternocleidomastoid muscle: a pilot randomized clinical trial. Journal of manipulative and physiological therapeutics. 2013 Sep 1;36(7):403-11.