Spasticity: Difference between revisions
No edit summary |
No edit summary |
||
| Line 71: | Line 71: | ||
| | | | ||
|} | |} | ||
<br>Permanent loss of joint range has been known to occur 3-6 weeks after both stroke and brain injury and therefore it is important that spasticity is identified early on in the assessment in order for it to be monitored and managed as required. In a person with hemiplegia the lower limb pattern is plantar flexion and inversion of the ankle with hamstring tightness limiting knee range of motion as well as adductor spasticity. Upper limb presentation is usually shoulder adduction, internal rotation, elbow flexion, forearm pronation with wrist and elbow flexion. | <br>Permanent loss of joint range has been known to occur 3-6 weeks after both stroke and brain injury and therefore it is important that spasticity is identified early on in the assessment in order for it to be monitored and managed as required. In a person with hemiplegia the lower limb pattern is plantar flexion and inversion of the ankle with hamstring tightness limiting knee range of motion as well as adductor spasticity. Upper limb presentation is usually shoulder adduction, internal rotation, elbow flexion, forearm pronation with wrist and elbow flexion. | ||
[[File:UL Tone. | {| width="100%" cellspacing="1" cellpadding="1" | ||
|- | |||
|[[File:UL + LL Tone.png|center|thumb]] | |||
|[[File:UL Tone.png|center|thumb]] | |||
====== IS IT SPASTICITY? ====== | ====== IS IT SPASTICITY? ====== | ||
| Line 199: | Line 203: | ||
# People with stroke should not be routinely offered splinting for the arm and hand. | # People with stroke should not be routinely offered splinting for the arm and hand. | ||
{| width="100%" cellspacing="1" cellpadding="1" | |||
|- | |||
|{{#ev:youtube|mvs4QOzM7wg|300}} | |||
|{{#ev:youtube|sHV3c4NvZP0|300}} | |||
|} | |||
== Recent Related Research (from [http://www.ncbi.nlm.nih.gov/pubmed/ Pubmed]) == | == Recent Related Research (from [http://www.ncbi.nlm.nih.gov/pubmed/ Pubmed]) == | ||
Revision as of 22:44, 19 June 2018
Original Editor - Timothy Assi
Top Contributors - Naomi O'Reilly, Sheik Abdul Khadir, Matt Ross, Rhiannon Clement, Kim Jackson, Lucinda hampton, Evan Thomas, Admin, Timothy Assi, Scott Buxton, Vidya Acharya, Garima Gedamkar, Lauren Heydenrych, Tony Lowe, Shreya Pavaskar, Wendy Walker, Ewa Jaraczewska, Oyemi Sillo, George Prudden, WikiSysop, Rachael Lowe and Rania Nasr
Definition[edit | edit source]
The most well-known and referenced description of spasticity is the physiological definition proposed by Lance in 1980:[1]
'Spasticity is a motor disorder characterised by a velocity-dependent increase in tonic stretch reflexes (muscle tone) with exaggerated tendon jerks, resulting from hyperexcitability of the stretch reflex, as one component of the upper motor neurone syndrome.'
More recently, a definition from Pandyan et al (2005)[2] states that spasticity is:
'Disordered sensorimotor control, resulting, resulting from an upper motor neuron lesion (UMN), presenting as an intermittent or sustained involuntary activations of muscles'
- Spasticity is a velocity-dependent disorder of the stretch reflex that results in increased muscle tone. [3]
Anatomy and Pathology[edit | edit source]
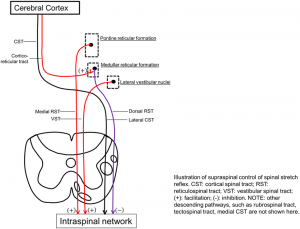
Primary impairments that result from an UMNL is usually due to the disruption of supraspinal control of descending pathways that normally control the excitatory and inhibitory influences on proprioceptive, cutaneous and nociceptive spinal reflexes.
The inhibitory system[edit | edit source]
Corticorecticular fibres form the corticoreticular spinal tract. These tracts travel with but are separate from, the corticospinal tract, and is responsible for the facilitation of inhibitory area within the medulla called the ventromedial reticular formation. Here, the dorsal reticulospinal tract originates, which is responsible for an inhibitory action on both the stretch and flexor reflexes.
the excitatory system[edit | edit source]
The bulbopontine tegmentum gives rise to the medial reticulospinal tract and, acting weakly with the vestibulospinal tract, is excitatory to both stretch and extensor reflexes and like the dorsal reticulospinal tract, is also inhibitory to the flexor reflexes.
The signs and symptoms between cortical UMN lesions and spinal cord UMN vary due to the location of where the disruption has taken place.
Different Lesions and Their Presentations[edit | edit source]
(Use the picture above to assist with understanding)
According to Strokes and Stack (2012) (reference)
Normal[edit | edit source]
Both the inhibitory system (corticospinal tract and dorsoreticulospinal tract) and excitatory systems (medial reticulospinal and vestibulospinal tract) are in dynamic balance and therefore the inhibition to the spinal cord is easily adjusted according to demand.
CORTICOSPINAL TRACT LESION[edit | edit source]
Although the corticospinal tract has an inhibitory influence on stretch and flexor reflex, the main inhibitory system produced by the dorsal reticulospinal tract remains intact and therefore the balance of excitatory and inhibitory influences are maintained.
internal capsule lesion[edit | edit source]
Leads to interruption of both the corticopinal and corticoreticular tract pathways that are responsible for the inhibitory response and some loss of inhibition to stretch and flexor stretches. The excitatory systems from both the medial reticulospinal and vestibulospinal tract are more dominant which leads to the facilitation of extensor and stretch reflexes but inhibition of flexors.
incomplete spinal cord lesion[edit | edit source]
Signs and symptoms will vary dependant on site and extent. If inhibitory system is affected then there will be an unopposed excitatory drive to stretch and extensor reflexes with partial inhibition of flexor reflexes.
COMPLETE SPINAL CORD LESION[edit | edit source]
Spinal reflexes are unopposed due to the complete loss of supraspinal control. Both flexor and extensor reflexes are disinhibited and therefore people may experience both flexor and extensor spasms.
Clinical Presentation[edit | edit source]
Spasticity is usually accompanied by one of more components of an upper motor neurone lesion seen in the table below, and is seldom exists in isolation. The impact of spasticity varies from person to person, ranging from being a clinical sign with no impact on function or a large increase in tone that affects transfers, mobility and personal care. It can also lead to contractures due to the shortening of muscles and tendons.
Positive Components
Negative Components
| Positive Component | Negative Component |
|---|---|
| Exaggerated tendon reflexes | Spastic Co-Contractions |
| Released reflexes | Motor Weakness |
| Babinski Sign | Slowed Movements |
| Increased Tone | Loss of Dexterity |
| Clonus | Loss of Selective Motor Control |
| Spastic Dystonia |
Permanent loss of joint range has been known to occur 3-6 weeks after both stroke and brain injury and therefore it is important that spasticity is identified early on in the assessment in order for it to be monitored and managed as required. In a person with hemiplegia the lower limb pattern is plantar flexion and inversion of the ankle with hamstring tightness limiting knee range of motion as well as adductor spasticity. Upper limb presentation is usually shoulder adduction, internal rotation, elbow flexion, forearm pronation with wrist and elbow flexion.
|
IS IT SPASTICITY?[edit | edit source]Spasticity has a number of characteristics that differentiates it from rigidity:
Diagnostic Procedures[edit | edit source]Ashworth Scale:[edit | edit source]The Ashworth scale is the most widely used assessment tool to measure resistance to limb movement in a clinic setting, although it is unable to distinguish between the neural and non-neural components of increased tone.[4] The scale is as follows: Tardieu Scale:[5][edit | edit source]This scale quantifies muscle spasticity by assessing the response of the muscle to stretch applied at specified velocities. Velocity to Stretch V1 As slow as possible V2 Speed of the limb segment falling V3 As fast as possible (> natural drop) Quality of Muscle Reaction 0 No resistance throughout passive movement 1 Slight resistance throughout,with no clear catch at a precise angle. 2 Clear catch at a precise angle followed by release 3 Fatigable clonus (<10secs) occurring at a precise angle 4 Unfatigable clonus (>10secs) occurring at a precise angle 5 Joint Immobile Spasticity Angle R1- Angle of catch seen at Velocity V2 or V3 Interpretation[6]
Outcome Measures
|
Recent Related Research (from Pubmed)[edit | edit source]
Failed to load RSS feed from http://www.ncbi.nlm.nih.gov/entrez/eutils/erss.cgi?rss_guid=1ry3PQ2nXoqvB6LNmV_aJDdgGeKvTQZ7Sa3cGngKJKIH7xMILQ: Error parsing XML for RSS
References[edit | edit source]
- ↑ Lance JW. Symposium synopsis. In: Feldman RG,fckLRYoung RR, Koella WP (eds). Spasticity: Disordered Motor Control. Chicago, IL: Year Book 1980:485–94.
- ↑ Pandyan AD, Gregoric M, Barnes MP et al. Spasticity: clinical perceptions, neurological realities and meaningful measurement. Disabil Rehabil 2005;27:2–6.
- ↑ Lance JW. The control of muscle tone, reflexes, and movement: Robert Wartenberg Lecture. Neurology. 1980;30(12): 1303-13.
- ↑ Ashworth B. Preliminary trial of carisoprodal in multiple sclerosis. Practitioner 1964;192:540–2.
- ↑ Tardieu G, Rondont 0, Mensch J, Dalloz J-C, Monfraix C, Tabary J-C. Responses electromyographiques a l'etirement musculaire chez l'homme normal. Rev Neurol 1957; 97: 60-61.
- ↑ Boyd R, Graham K. Objective Measurement of clinical findings in the use of Botox type A for the management of children fckLR with Cerebral Palsy. European Journal of Neurology 6(Supp 4) S23-35
- ↑ Hobart J, Riazi A, Thompson A et al. Getting the measure of spasticity in MS: The Multiple Sclerosis Society Spasticity Scale (MSSS-88). Brain 2006;129(Pt1):224–34.
- ↑ Valerie L Stevenson, Louise J Lockley and Louise Jarrett, In: Assessment of the individual with spasticity, chapter 2, SPASTICITY MANAGEMENT:A PRACTICAL MULTIDISCIPLINARY GUIDE,Pg-20-21.
- ↑ Snow BJ, Tsui JKC, Bhatt MH et al. Treatment of spasticity with botulinum toxin: a double blind study.Ann Neurol 1990;28:512–15.
- ↑ Penn RD, Savoy SM, Corcos D et al. Intrathecal baclofen for severe spinal spasticity. N Engl J Med 1989;320:1517–21.
- ↑ Smith C, Birnbaum G, Carlter JL et al. Tizanidine treatment of spasticity caused by multiple sclerosis:results of a double-blind, placebo-controlled trial. US Tizanidine Study Group. Neurology 1994;44:S34–42.
- ↑ Wade DT, Wood VA, Heller A et al. Walking after stroke: measurement and recovery over the first three months. Scand J Rehabil Med 1987;19:25–30.
- ↑ Morris SL, Dodd KJ, Morris ME. Outcomes of progressive resistance strength training following stroke: a systematic review. Clin Rehabil. 2004 Feb;18(1):27-39.
- ↑ Lourenção MI, Battistella LR, de Brito CM, Tsukimoto GR, Miyazaki MH. Effect of biofeedback accompanying occupational therapy and functional electrical stimulation in hemiplegic patients. Int J Rehabil Res. 2008 Mar;31(1):33-41
- ↑ Manganotti P, Amelio E. Long-term effect of shock wave therapy on upper limb hypertonia in patients affected by stroke. Stroke. 2005 Sep;36(9):1967-71. Epub 2005 Aug 18.
- ↑ Amelio E, Manganotti P.Effect of shock wave stimulation on hypertonic plantar flexor muscles in patients with cerebral palsy: a placebo-controlled study. J Rehabil Med. 2010 Apr;42(4):339-43.(B)
- ↑ Ansari NN, Adelmanesh F, Naghdi S, Tabtabaei A.The effect of physiotherapeutic ultrasound on muscle spasticity in patients with hemiplegia: a pilot study. Electromyogr Clin Neurophysiol. 2006 Jul-Aug;46(4):247-52.(B)
- ↑ Ansari NN, Naghdi S, Bagheri H, Ghassabi H. Therapeutic ultrasound in the treatment of ankle plantarflexor spasticity in a unilateral stroke population: a randomized, single-blind, placebo-controlled trial. Electromyogr Clin Neurophysiol. 2007 May-Jun;47(3):137-43. (B)
- ↑ Harlaar J, Ten Kate JJ, Prevo AJ, Vogelaar TW, Lankhorst GJ. The effect of cooling on muscle co-ordination in spasticity: assessment with the repetitive movement test. Disabil Rehabil. 2001 Jul 20;23(11):453-61.
- ↑ Van der Salm A, Veltink PH, Ijzerman MJ, Groothuis-Oudshoorn KC, Nene AV, Hermens HJ. Comparison of electric stimulation methods for reduction of triceps surae spasticity in spinal cord injury. Arch Phys Med Rehabil. 2006 Feb;87(2):222-8.
- ↑ RCP (2016). National Clinical Guidelines for Stroke. Available at: https://www.strokeaudit.org/Guideline/Full-Guideline.aspx [Accessed 19th June 2018].