Huntington's Disease Case Study
Abstract[edit | edit source]
This simulated case study involves a 45-year-old male (Johnny Miller) with a recent diagnosis of early-stage Huntington’s disease. This fictional case study goes into depth on the patient’s characteristics, examination findings, selected interventions of the interdisciplinary team, and outcome reassessments 6 months post-referral. Mr. Miller attended an appointment with his physician due to an increased frequency of errors in work-related fine motor tasks and overall difficulty with coordination. During this visit, the physician also noted cognitive abnormalities including a short temper and unexplained outbursts and decided to refer Mr. Miller to a neurologist for further assessment. Following his diagnosis of Huntington’s Disease and referral to an outpatient rehabilitation physiotherapy clinic, Mr. Miller received an extensive assessment to appropriately guide his treatment plan. Outcome measures were utilized to determine Mr. Miller’s functional capacity in various tasks at the time of admission and were used as comparison data 6 months later to track the progression of the disease as well the effectiveness of interventions utilized. Patient centered goals were utilized to ensure the rehabilitation was meaningful to Mr. Miller. A variety of treatment interventions included aerobic exercise, balance and coordination training, fine motor skill training, and education on relevant functional tasks. Social workers, psychologists, and occupational therapists provided complimentary treatment as members of the therapeutic team. Mr. Miller’s 6-month re-evaluation showed sustained performance levels from his initial assessment during the selected functional outcome measures.
Introduction[edit | edit source]
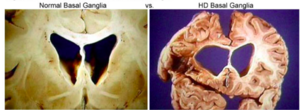
Huntington disease (HD) is an incurable, inherited disorder that occurs from gene mutation and results in the progressive degeneration of nerve cells in the brain[2].The basal ganglia is the primary location of degeneration, specifically the striatum located within it. The primary role of the basal ganglia is to coordinate movement so that it is smooth[3]. When the striatum degenerates, there is a decreased ability to inhibit unwanted movement[4]. This leads to an excessive amount of involuntary movement, known as chorea. HD affects the ability of individuals to move, think, and behave. HD typically first appears at the age of 30 to 40, however symptoms may present at any point in life[2]. HD that occurs before the age of 20 is called juvenile Huntington’s disease[2].
Pathophysiology[5][edit | edit source]
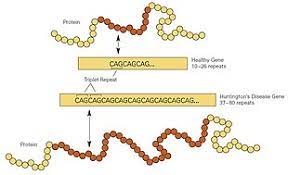
HD has been found to occur from the mutation of the Huntington gene. The Huntington gene contains a repetition of the CAG trinucleotide protein that everyone is born with and is typically repeated anywhere between 10-35 times. The exact function of this gene is unknown, however it is believed to play a part in neural development. In HD, the mutated Huntington gene causes an increase in the number of repeats of the CAG trinucleotide, with more repetitions leading to a greater risk for the disease. It has been found that individuals with HD have the CAG trinucleotide repeat over 36 times, and in severe cases can be seen repeated over 120 times. This elongation of the protein is then fragmented into smaller sections, which accumulate on neurons within the brain and disrupt function. These neurons eventually die and the resulting neurodegeneration is associated with many of the symptoms commonly seen in Huntington’s.
Presentation[edit | edit source]
HD has been found to be heavily influenced by genetics. Humans have two copies of the Huntington gene, however those who have a mutation to at least one of these copies have a greater chance of experiencing the HD symptoms[6]. The inheritance of HD is autosomal dominant, which means that each child of a parent with HD has a 50% chance of inheriting the mutated copy, and further implies that they have a 50% chance of inheriting the disease[7].
Signs & Symptoms[5][edit | edit source]
- Movement - chorea, dystonia, impaired gait/posture/balance, speech/swallowing, eye movement
- Cognitive - difficulty organizing tasks, lack of impulse control, lack of awareness of their behaviours, trouble finding words, learning new information
- Behavioural - depressive symptoms such as sadness, apathy, social withdrawal, insomnia, fatigue
Purpose of Discussing HD in a Case Study Format[edit | edit source]
The physiotherapy management of Huntington’s Disease is highly variable and highly dependent on the symptoms. This fictional case gives an opportunity for both physiotherapists and physiotherapy students to discuss and reflect about the route of assessment taken in order to prepare for clinical cases that may be observed. The discussions surrounding a simulated case allow for a significant learning opportunity for all involved, and overall an improvement in the approach to assessing and managing a case such as Huntington’s Disease.
Client Characteristics[edit | edit source]
The client in this case is a 45 year old, caucasian male, named Johnny Miller. Mr. Miller has been diagnosed with Huntington’s Disease following a consultation with a neurologist. The consultation with the neurologist occurred as a result of numerous concerning signs, including accidents within the workplace and during leisure activities. Mr. Miller is currently in the early stages of Huntington’s Disease, only showing relatively minor signs and symptoms. Mr. Miller has been referred to this outpatient rehabilitation physiotherapy clinic in order to address his current impairments, as well as prevent the progressive onset of future impairments associated with Huntington’s Disease. Overall, Mr. Miller is a pleasant man, who loves to talk about his involvement as the owner, manager, and head sushi chef at 241 sushi, as well as his dog Kenny, and his 9-year-old daughter's hockey team.
Examination Findings[edit | edit source]
Subjective[edit | edit source]
History of Present Illness (HPI)[edit | edit source]
Mr. Miller has been experiencing an increase in accidents at work over the last couple of months, such as accidentally cutting his fingers with his knife and dropping plates. Two weeks ago on April 2nd, 2021, Mr. Miller got a deep cut on his left thumb. He started bleeding aggressively, and decided to walk into the emergency department within the next hour. He was later seen by a physician who gave him 6 stitches. The physician had noticed that Mr. Miller was stumbling often and had difficulty with his upper limb coordination. He told the physician that he has had a difficult time at work using his knife recently, and mentioned that “it must be due to being tired”. The physician also noted that Mr. Miller had a short temper and got angry with some of the other patients in the waiting room. The physician referred Mr. Miller to a neurologist for an assessment later that month. After the neurologist performed a physical examination to look at his motor and sensory functioning, an MRI and genetic test was ordered based on symptoms that suggested Huntington’s disease. The results of the MRI and genetic testing led to Mr. Miller’s diagnosis of Huntington’s disease on May 4th, 2021.
Past Medical History (PMHx)[edit | edit source]
Mr. Miller was diagnosed with hypertension 5 years ago, likely due to the stress of running his business. Otherwise, relatively healthy.
Family History[edit | edit source]
Mr. Miller's mother passed away at the age of 35 due to a motor vehicle accident. His father is a relatively healthy 68 year old and lives independently in Montreal. Mr. Miller has 1 older sister who is a chemical engineer living independently in Smith Falls; she had breast cancer 3 years ago which was resolved after a mastectomy. No other relevant family history.
Medications[edit | edit source]
Atorvastastin (10 mg per day) to lower cholesterol and reduce the risk of heart disease
Lisinopril (15 mg per day) to lower his blood pressure
Social History (SHx)[edit | edit source]
Mr. Miller currently lives in Kingston, Ontario in a two-story detached home. He lives with his wife and 4 kids who range from 4-12 years old. Mr. Miller’s house consists of 3 stairs to get inside, and 15 stairs to get to the second floor where his bedroom is located. Additionally, Mr. Miller spends much of his day at 241 Sushi where he is the Owner, Manager, and Head Chef. 241 Sushi has 10 stairs to the basement where the refrigerator and freezer are located. When not working, Mr. Miller enjoys watching the Kingston Frontenacs, with whom he is a season ticket holder, as well as coaching his 9-year-old daughter's hockey team.
Health Habits (HH)[edit | edit source]
Mr. Miller does not smoke or use recreational drugs. He drinks alcohol occasionally (1-2 drinks/week).
Current Functional Status (FnSt)[edit | edit source]
| Basic Activities of Daily Living (BADL’s) | Instrumental Activities of Daily Living (IADL’s)[8] | Physical Activity (PA) |
|---|---|---|
| Dressing: Independent
Eating: Independent Ambulating: Independent, but reduced sense of balance recently when walking. Toileting: Independent Hygiene: Independent |
Shopping: Independent
Housework: Shared responsibilities with wife; lack of motivation and confidence due to symptoms Accounting: Shared responsibilities with wife; recently having difficulties with mathematics Food Preparation: Independent but more difficult due to “random jerky movements”; several incidents at work have occurred in last few months Transportation: Independent driver but becoming more anxious that one of these “jerky movements” will cause a dangerous situation. He has reduced his driving because of this unless it is necessary. |
Able to skate on ice and coach hockey, but has experienced a few falls more recently from losing balance. Mr Miller reports feeling less confident running drills and puck handling recently.
Work out at home in his garage 2x/week, primarily with body weight exercises and resistance bands. |
Functional History (FnHx)[edit | edit source]
**Prior to December 7th, 2020**
- Independent in all ADL’s and IADL’s.
- Able to skate on ice rink to help coach hockey for ~2 hour practice twice per week
Objective[edit | edit source]
Observation[edit | edit source]
Upon observation, Mr. Miller was aware and oriented. In sitting, he presents with occasional repeated involuntary movements of both the upper and lower extremities. This was most present at the distal joints of the fingers, wrists, and ankles. Noted was also facial grimacing/twitching. There was no apparent increased tone or atrophy. Sitting posture was normal.
Vital Signs[edit | edit source]
Heart Rate (HR): 98 bpm
Respiratory Rate (RR): 16 breaths per minute
Blood Pressure (BP): 127/78 (while on medication)
Pulse: Strong radial pulse, regular-regular.
Active and Passive Range of Motion (AROM & PROM)[edit | edit source]
All of the following movements were assessed bilaterally:
| Cervical Spine | Flexion, Extension, Side-Flexion, Rotation, Protraction, Retraction |
| Shoulder (Glenohumeral) | Flexion, Extension, Abduction, Adduction, Internal Rotation, External Rotation |
| Elbow | Flexion, Extension, Pronation, Supination |
| Wrist | Flexion, Extension, Pronation, Supination |
| Hip | Flexion, Extension, Abduction, Adduction, Internal Rotation, External Rotation |
| Knee | Flexion, Extension |
| Ankle | Dorsiflexion, Plantarflexion, Inversion, Eversion |
All joints had full active ROM, however chorea (involuntary contractions) was noted globally, with distal joints being the most affected.
All movements assessed for AROM were also assessed for PROM. All measurements were within normal limits, bilaterally. Following full and pain free PROM, therapist overpressure was applied to assess end feel. No abnormal end feels were reported at any of the assessed joints.
Muscle Strength[9][edit | edit source]
Manual Muscle Testing for both the upper and lower limbs was assessed. The upper limbs were assessed in order to better understand potential deficits that may be contributing to the hand dysfunction that Mr. Miller is experiencing. Lower limb assessment provided guidance as to potential causes with regards to ambulatory issues that have recently arisen for Mr. Miller.
Upon assessment, Mr. Miller presented with the following concerning outcomes:
- Grip Strength significantly reduced bilaterally (3+/5)
- Knee Extensors significantly reduce bilaterally (4-/5)
- Knee Flexors significantly reduced bilaterally (4-/5)
All other muscle groups assessed scored at least a 4+/5 at minimum. These findings provide an objective finding that can be addressed through a strengthening program to target and prevent degenerative changes associated with Huntington’s Disease.
Neurological Scan[edit | edit source]
- Cranial Nerves → Normal
- UMN Testing → Positive Babinski Test and Clonus Test, Negative Hoffman's Test
- LMN Testing → Normal biceps, triceps, patellar and Achilles tendon reflexes
Balance[10][edit | edit source]
Mr. Miller presented with a history of balance related issues upon subjective interview. Additionally, observation indicated notable signs of impairment and/or concern with both balance itself, and Mr. Miller’s confidence in his balance.
In order to assess Mr. Miller’s balance, the Berg Balance Scale was performed. The Berg Balance Test has been found to be both reliable and valid in assessing balance and coordination in those with Huntington’s Disease. See Outcome Measures for results and interpretation.
Ambulation[edit | edit source]
Upon entry, Mr. Miller presented with what appeared to be a slightly hesitant gait, being very aware of his environment as well as the speed at which he walked. Observation gave indication to some initial identifiable factors of a choreiform gait pattern. As a result, a TUG assessment was given in addition to the Berg Balance Scale to provide an understanding to Mr. Miller’s risk of fall within the community. The results and interpretations can be found in the outcome measures.
Cognitive Functioning[edit | edit source]
When taking a subjective history from Mr. Miller, we noticed that he had difficulty answering some questions about his home and job. He was continuously slow to answer and understand our questions while also requesting us to repeat some questions[11]. We communicated our concerns on his possible cognitive symptoms with our psychologist, and she decided to carry out a MoCA[12]. Although the Mini Mental State Examination (MMSE) has previously been the most commonly used method for assessing cognitive impairment in individuals with HD, the MoCA has recently been identified as the best screening tool for identifying cognitive impairment in HD[13].
Outcome Measures[edit | edit source]
Berg Balance Scale (BBS)[edit | edit source]
The BBS involves 14 tasks that are graded from 0-4, with higher scores indicating better function[10]. The maximum score that can be achieved is 56. The tasks in the BBS include static sitting, sit-to-stand transitions, static standing, decreased base of support while standing, and dynamic standing tasks with internal perturbations. A score below 36 is associated with an increased fall risk close to 100%.
Mr. Miller scored 49 on the BBS. This score indicates that Mr. Miller has mild to moderate balance deficits and has a 6-8% increased chance of fall risk[10]. The short to medium term prognosis of HD indicates that Mr. Miller can prevent further deficits and improve upon his balance and coordination with appropriate training[10].
Timed Up and Go (TUG) Test[10][edit | edit source]
The TUG test involves the patient doing a sit-to-stand transfer from a chair, ambulating 3m, turning around at the 3m mark and returning to a seated position in the chair they began the test in. If the patient takes >13.5s to complete the TUG, they have a greater associated risk of a fall.
Mr. Miller took 9.7 seconds to complete the test. Scoring <10s indicates that Mr. Miller is independent in balance and motor skills, and is not at a point where he is at a risk for falling.
Unified Huntington's Disease Rating Scale (UHDRS)[14][15][edit | edit source]
This test was created to assess four primary characteristics of Huntington’s Disease: motor functioning, cognitive functioning, behavioural abnormalities and functional capacity. These four categories are further broken down in multiple sections that utilize both self-report measures and performance based assessments. Because the UHDRS tests many different aspects of HD, it has been identified as a tool which can help track progression of the disease, as well as help guide interventions which would be most useful for the patient. It is recommended that the UHDRS be performed once every 2 years in order to provide an accurate gauge of how the disease is progressing.
During our baseline assessment of the UHDRS, Mr. Miller performed quite well with most of the tasks. However, we were able to identify some specific aspects of his condition which related to scoring a bit lower in the Motor functioning section and the cognitive functioning sections; Mr. Miller scored within normal limits for the functional capacity and behavioral abnormalities sections.
Montreal Cognitive Assessment (MoCA)[edit | edit source]
The MoCA is a tool that assesses an individual’s cognitive functioning in areas which include: memory (STM and Delayed Recall), attention, visuospatial and executive functioning, abstraction, and orientation[16]. This test has a range from 0-30 points, with a score less than 26 suggesting a reduced level of cognitive functioning[16].
Mr. Miller scored a 25/30 on the MoCA. This identifies that Mr. Miller is experiencing mild cognitive impairment[13].
Clinical Impression[edit | edit source]
Problem List[edit | edit source]
Body structure/function:[edit | edit source]
Mr. Miller is currently experiencing difficulty with involuntary movements, specifically when utilizing fine motor skills of the hands. Finger control at work has become particularly problematic, as a result of the chorea movements.
Activity Limitations:[edit | edit source]
Mr. Miller has experienced difficulty maintaining balance while walking, particularly under increasingly demanding cognitive tasks. As well, Mr. Miller has found it increasingly difficult to skate on the ice due to balance difficulties.
Participation Restrictions:[edit | edit source]
Mr. Miller’s balance impairments have affected his ability to coach his daughter’s hockey team. Additionally, Mr. Miller’s ability to play the role of head sushi chef at 241 Sushi has become limited. He is having difficulty keeping pace with the amount of orders that come in as a result of the hand and finger dysfunction.
Personal Factors:[edit | edit source]
Mr. Miller has become increasingly frustrated with these choreatic movements and is worried that they will not go away. His reduced ability to function at work properly and coach his daughter’s hockey team has left him feeling depressed from time to time. As well, Mr. Miller has found it increasingly difficult to maintain balance while ambulating and
Environmental Factors:[edit | edit source]
The demands of work are causing stress for Mr. Miller. He feels as though customer satisfaction is falling as a result of prolonged timing to serve the food people order. Additionally, Mr. Miller’s wife has been concerned about his wellbeing as a result of his balance impairments. She is making him be increasingly cautious with his everyday activities despite his abilities. An overall heightened state of concern surrounds Mr. Miller from his family.
Diagnosis (Dx)[edit | edit source]
Medical Dx: Early-Stage of Huntington’s Disease due to a lesion to the basal ganglia.
Physiotherapy Dx: Mr. Miller is a 45 year old male currently demonstrating signs of chorea, decreased coordination and difficulty with fine motor skills of both upper and lower extremities due to his recent diagnosis of Huntington’s Disease.
Interventions[edit | edit source]
Patient Centered Goals[edit | edit source]
| Short Term Goals | Long Term Goals |
| 1. Mr. Miller will perform 3 balance related training activities at least 5 days per week for the next 3 weeks. | 1. Mr. Miller will participate in an organized skating session at least once per week to prevent an increase in confidence-related skating falls. |
| 2. Mr. Miller will learn effective techniques to be able to safely cut 10 bowls of vegetables each day within 3 weeks. | 2. Mr. Miller will continue to ambulate 500m without a gait aid, while maintaining a walking speed of at least 1 metre per second in every 50m interval. |
| 3. Mr. Miller will perform 30 minutes of aerobic exercise training at least 4 days per week for the next 3 weeks. | 3. By 2 months, Mr. Miller will be able to take his dog for a 1 kilometer walk 2x a week. |
Intervention Approaches and Techniques[edit | edit source]
| Intervention | Frequency | Intensity | Time/Volume | Type |
Fine Motor Skill Training
|
3 Days per week | Low to Moderate, avoiding fatigue | 15 minutes each day, no more than 5 minutes per task. | -Marble organization, dice games |
Education Regarding Kitchen Safety
|
Initially and follow up as required. | N/A | N/A | N/A |
| Balance and Coordination Training[17] | 3 Days per week | Low to Moderate | 15 minutes of increasingly demanding training | Begin with double stance, remove sensory inputs as we progress such as closing eyes, standing on foam, reducing BOS, single leg stance. |
Cardiorespiratory Training
|
2 Days per Week | Moderate to Vigorous | 20 Minutes each day. | Fast paced (2m/s) walk with varying inclines throughout the intended route. |
Wheelchair education
|
1 Day every 2 weeks until adequate then as needed. | N/A | N/A | N/A |
Cognitive Functioning Training[18]
|
1 day per week | Easy Mode | 3 songs per session | Dance Dance Revolution at arcade with youngest son. |
Interdisciplinary Care Team Management[edit | edit source]
Physiotherapy[19][edit | edit source]
The physiotherapist will focus on improving balance and retraining gait, with fall prevention being a priority. Reliable and valid outcome measures will be used to track the patient’s progress during functional tasks. During later stages of the disease, the focus will shift to improving respiratory function and education on wheelchair usage when independent gait is no longer feasible.
Occupational Therapy[19][edit | edit source]
Occupational therapists aid in the management of basic skills such as eating, dressing and bathing. OTs can also help in achieving many of Mr. Miller’s goals, specifically with the learning of new techniques for cutting vegetables.
Social Worker[19][edit | edit source]
Social workers would offer support for both Mr. Miller and his family, as well as provide advice for responsibilities such as finances and social networking. The utilization of support groups and counseling may also be helpful during this time for Mr. Miller’s mental health. Additionally, social workers can aid in the goal setting.
Psychologist[19][edit | edit source]
Psychologists would offer in home visits to perform cognitive behavioral strategies to keep Mr. Miller’s outlook positive. They will help Mr. Miller stay motivated to adhere to his established programmes which will help him reach his outlined goals.
Speech Language Pathologist[19][edit | edit source]
Speech Language Pathologists (SLPs) can help identify Mr. Miller’s areas of speech limitations and address them appropriately. They can teach breathing exercises to ensure speech patterns stay consistent. If speech becomes a greater issue, a SLP can prescribe an Augmented and Alternative Communication (AAC) to help Mr. Miller continue communicating in everyday life. Furthermore, a SLP can teach Mr. Miller exercises to promote muscular control which will improve swallowing and chewing effectiveness and decrease risk associated with these activities.
Physician[19][edit | edit source]
The physician’s role will involve the initial assessment and diagnosis of HD and prescribing medication throughout the condition’s progression.
Outcome Reassessment - 6 Months Post-Referral[edit | edit source]
Berg Balance Scale (BBS)[10][edit | edit source]
After 6 months of undergoing physiotherapy interventions, Mr. Miller once again did the Berg Balance Scale and scored a 51. Although this score indicates that Mr. Miller improved slightly, his improvement of 2 points does not meet the MCID value to make a notable change in his functioning. However, due to the nature of HD and an expectation of worsening symptoms with time, seeing no reduced capacity in his balance is seen as a positive sign.
Timed Up and Go (TUG) Test[10][edit | edit source]
After 6 months of undergoing physiotherapy interventions, Mr. Miller did the TUG test and scored a similar time to his initial trial. He was able to complete the test in 10.2 seconds, which is 0.5 seconds slower than 6 months ago. A difference of 0.5 seconds is not a clinically significant difference.
Referrals[19][edit | edit source]
- A social worker would provide ongoing support for the patient and family and as they navigate compensatory strategies to continue managing daily responsibilities.
- Psychologist to provide supportive strategies to maintain cognition and manage frustration, stress, and depression. Behavioural and cognitive symptoms are a primary aspect of HD and a psychologist would play a critical part in helping manage these symptoms.
- Occupational Therapist to provide ongoing techniques to maintain safety at work, during activities of daily living, and monitor use of assistive devices.
Discharge Planning[edit | edit source]
Currently, there is no plan for patient discharge from physiotherapy due to ongoing management of HD. Owing to the degenerative nature of the disease, continued support is essential to preserving function and maintaining quality of life for Mr. Miller.
Discussion[edit | edit source]
Implications to Clinical Practice[edit | edit source]
Currently, there is no known cure for HD. Physiotherapy, along with support from the interdisciplinary care team, can help patients manage the progressive course of the disease to help maintain daily function and quality of life[20]. Studying HD, a hyperkinetic disorder, may assist health care professionals in managing other hyperkinetic movement disorders like Ataxia, Restless Leg Syndrome, and Myoclonus[20]. Since the initial onset of symptoms for individuals with HD is commonly in adulthood, this can be extremely challenging for both patients and family members to adapt. By understanding the physiology of the disease and introducing the necessary health care supports, the burden of HD may be reduced to help these individuals cope while still participating in a fulfilling life.
References[edit | edit source]
- ↑ 1.0 1.1 Liou S. The basic neurobiology of Huntington's disease [Internet]. HOPES Huntington's Disease Information. 2016 [cited 2022, May10]. Available from: https://hopes.stanford.edu/the-basic-neurobiology-of-huntingtons-disease-text-and-audio/
- ↑ 2.0 2.1 2.2 Folstein SE. Huntington's Disease: A Disorder of Families. The Johns Hopkins University Press. 1989
- ↑ Reiner A, Dragatsis I, Dietrich P. Genetics and neuropathology of Huntington's disease. Int Rev Neurobiol. 2011;98:325-72. Doi 10.1016/B978-0-12-381328-2.00014-6.
- ↑ Waldvogel HJ, Kim EH, Tippett LJ, Vonsattel JP, Faull RL. The Neuropathology of Huntington's Disease. Curr Top Behav Neurosci. 2015;22:33-80. doi: 10.1007/7854_2014_354.
- ↑ 5.0 5.1 Roos RA. Huntington's disease: a clinical review. Orphanet J Rare Dis. 2010 Dec 20;5:40. doi: 10.1186/1750-1172-5-40
- ↑ Myers RH. Huntington's disease genetics. NeuroRx. 2004 Apr;1(2):255-62. doi: 10.1602/neurorx.1.2.255
- ↑ Conneally PM. Huntington disease: genetics and epidemiology. Am J Hum Genet. 1984 May; 36(3): 506-26. Doi: https://doi.org/10.1016/0888-7543(89)90062-1
- ↑ Beglinger LJ, O'Rourke JJ, Wang C, Langbehn DR, Duff K, Paulsen JS; Huntington Study Group Investigators. Earliest functional declines in Huntington disease. Psychiatry Res. 2010 Jul 30;178(2):414-8. doi: 10.1016/j.psychres.2010.04.030
- ↑ Cruickshank, TM, Reyes, AP, Penailillo, LE, et al. Effects of multidisciplinary therapy on physical function in Huntington's disease. Acta Neurol Scand. 2018 Dec; 138(6): 500-507. https://doi-org.proxy.queensu.ca/10.1111/ane.13002
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 Quinn L, Khalil H, Dawes H, Fritz NE, Kegelmeyer D, Kloos AD, et al. Reliability and Minimal Detectable Change of Physical Performance Measures in Individuals With Pre-manifest and Manifest Huntington Disease. Physical Therapy. 2013 Jul 1;93(7):942–956. Doi: https://doi.org/10.2522/ptj.20130032
- ↑ Paulsen JS. Cognitive impairment in Huntington's disease: diagnosis and treatment. Curr Neurol Neurosci Rep. 2011 Oct; 11(5) :474-83. Doi: 10.1007/s11910-011-0215-x
- ↑ Gluhm S, Goldstein J, Brown D, Van Liew C, Gilbert PE, Corey-Bloom J. Usefulness of the Montreal Cognitive Assessment (MoCA) in Huntington's disease. Mov Disord. 2013 Oct;28(12):1744-7. Doi: 10.1002/mds.25578
- ↑ 13.0 13.1 Rosca EC, Simu M. Montreal cognitive assessment for evaluating cognitive impairment in Huntington’s disease: a systematic review. CNS Spectrums. 2022 Feb; 27(1): 27-45. Doi: https://doi.org/10.1017/S1092852920001868
- ↑ Seisling S., van Vugt, J.P., Zwinderman, K.A., Roos, R.A. Unified Huntington’s Disease Rating Scale: a follow up. 1998 Nov; 13(6). doi: 10.1002/mds.870130609.
- ↑ Huntington Study Group. Unified Huntington's disease rating scale: reliability and consistency. Movement disorders. 1996;11:136-42. Doi: 10.1002/mds.870110204
- ↑ 16.0 16.1 Gluhm S, Goldstein J, Brown D, Van Liew C, Gilbert PE, Corey-Bloom J. Usefulness of the Montreal Cognitive Assessment (MoCA) in Huntington's disease. Mov Disord. 2013 Oct;28(12):1744-7. Doi: 10.1002/mds.25578
- ↑ 17.0 17.1 17.2 17.3 Wyant KJ, Ridder AJ, Dayalu P. Huntington's Disease-Update on Treatments. Curr Neurol Neurosci Rep. 2017 Apr;17(4):33. doi: 10.1007/s11910-017-0739-9.
- ↑ Kloos AD, Fritz NE, Kostyk SK, Young GS, Kegelmeyer DA. Video game play (Dance Dance Revolution) as a potential exercise therapy in Huntington's disease: a controlled clinical trial. Clin Rehabil. 2013 Nov;27(11):972-82. doi: 10.1177/0269215513487235
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 Veenhuizen RB, Kootstra B, Vink W, Posthumus J, van Bekkum P, Zijlstra M, Dokter J. Coordinated multidisciplinary care for ambulatory Huntington's disease patients. Evaluation of 18 months of implementation. Orphanet J Rare Dis. 2011 Nov 18;6:77. doi: 10.1186/1750-1172-6-77.
- ↑ 20.0 20.1 Cardoso F, Seppi K, Mair KJ, Wenning GK, Poewe W. Seminar on choreas. Lancet Neurol. 2006 Jul;5(7):589-602. doi: 10.1016/S1474-4422(06)70494-X






