Pain Descending Pathways
Original Editor - Adrian Mallows.
Top Contributors - Adrian Mallows, Lucinda hampton, Jo Etherton, Admin and Lauren Lopez
Introduction[edit | edit source]
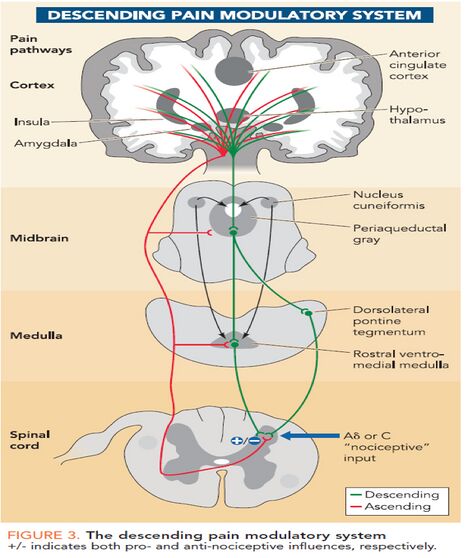
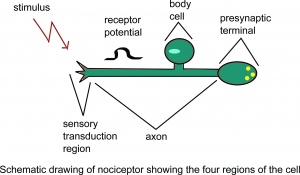
Once a pain signal from the ascending pathway reaches the somatosensory cortex, it triggers the descending pain modulatory system. The goal of this pathway is to allow the organism to function enough to respond to the pain source by reducing the pain signal through neuronal inhibition ie the "top down" modulation of pain. It begins in the periaqueductal gray (PAG), an area of grey matter in the midbrain that is involved in a descending pain control pathway. The periaqueductal gray, or PAG, receives pain information via the spinomescencephalic tract and processes the nociceptive information and relays it to the rostral ventral medulla (RVM). These neurons in the RVM then send a signal down the spinal cord and activate the endogenous opiate system to suppress pain.[1][2]
Endogenous Opioid System[edit | edit source]
Within the central nervous system, there are three types of opioid receptors which regulate the neurotransmission of pain signals. These receptors are called mu, delta, and kappa opioid receptors.
They are G protein-coupled receptors and their activation leads to a reduction in neurotransmitter release and cell hyperpolarisation, reducing cell excitability. Our body contains endogenous opioids which can modulate pain physiologically. There are three types of endogenous opioids:
- Β-endorphins – which predominately binds to mu opioid receptors
- Dynorphins – which predominately bind to kappa opioid receptors
- Enkephalins – which predominately bind to delta opioid receptors
Opioids can regulate pain on a number of levels, both within the spinal cord, brain stem, and cortex. Within the spinal cord, both dynorphins and enkephalins can act to reduce the transmission of pain signals in the:
- Dorsal horn, as the post-synaptic ends of second-order neurones have opioid receptors within the membrane.
- Pre-synaptic ends where first-order neurones contain opioid receptors. [3][4].
When endogenous opioids act on these receptors it reduces neurotransmitter release from the first-order neurone, and causes hyperpolarisation of the second-order neurone. Together, this reduces the firing of action potentials in the second-order neurone, blocking the transmission of pain signals.
Opioid receptors are located in the: brain (highest densities found in the thalamus, the PAG, and the RVM); spinal cord (high density in the dorsal horn region) and; peripheral organs, eg heart, blood vessels, kidneys, sympathetic ganglia, and the adrenal medulla)[5] The analgesic effects are mediated via mu receptors. Opioids reduce pain transmission at the dorsal horn by inhibiting excitatory neurotransmitter release. They act centrally in the PAG by enhancing descending inhibition.[6]
Why is the System Useful?[edit | edit source]
Evidence for pain modulatory mechanisms were first recorded by Beecher[7]. Beecher, a physician serving the US Army during World War II, observed as many as three quarters of badly wounded soldiers reported none to only moderate pain and did not require pain relief medication. According to his report the men were alert and responsive and the injuries were not trivial, including compound fractures and penetrating wounds. This led him to the conclusion that "strong emotions" block pain. This clearly opposes the classical Cartesian view where pain was considered to be a hard-wired system that passively transmitted noxious inputs to the brain.
It is now generally accepted that the experience of pain does not solely rely on noxious inputs, but many variables interplay with the experience, including memory, mood, environment, attention and expectation. This means the resultant pain experienced to the same sensory input can vary considerably[8]. It is the brain's job to weigh all the information and decide whether creating pain is the most appropriate response. This provides a necessary survival function since it allows the pain experience to be altered according to the situation rather than having pain always dominate[8]. See Also Sociological Basis of Pain
Implications for Physiotherapists[edit | edit source]
Knowledge of the descending pain modulatory system and its components can help physiotherapists in several ways.
- It helps physiotherapists explain why the amount of pain a patient is experiencing does not always relate to the amount of tissue damage they have sustained[7]. Physiotherapists can educate their patients about the role of the descending pain modulatory system and how the central nervous system weighs all the information before deciding if a pain experience is the most appropriate action for survival. See Pain neuroscience education[9][10][11][12] [13][14].
- Knowledge of the anatomy involved in the descending pain modulatory system can help physiotherapists utilise management strategies to that access and activate the system. These could include adding distractions to exercises and performing exercises in different emotional states and or in different environments.
- Manual techniques eg joint mobilisations, manipulations have been proposed to activate the system and significantly contribute to their therapeutic effects[15]. It is important to understand the extent to which manual therapy influences these underlying mechanisms. New studies of manual therapy should aim to link the many immediate changes in neurophysiological function (e.g., changes in sympathetic nervous system function and the endogenous pain inhibitory systems, among others) more closely to the clinical complaints of our patients[16].
References[edit | edit source]
- ↑ Neuroscientifically Challenged PAG Available:https://neuroscientificallychallenged.com/glossary/periaqueductal-gray (accessed 18.4.2022)
- ↑ Tufts Available: Opioid peptideshttps://sites.tufts.edu/opioidpeptides/pathways-and-receptors/classical-opioid-signaling/ (accessed 18.4.2022)
- ↑ teach me physiology Pain pathways Available:https://teachmephysiology.com/nervous-system/sensory-system/pain-pathways/ (accessed18.4.2022)
- ↑ NPS medicine Opioids Available: https://www.nps.org.au/australian-prescriber/articles/opioids-mechanisms-of-action(accessed 18.4.2022)
- ↑ Decaillot F, Abul-Husn NS, Devi L. Opioid and Opioid-like Receptors.Available: https://www.sciencedirect.com/science/article/pii/B9780080552323601035 (accessed 8.4.2022)
- ↑ Reddi D, Curran N, Stephens R. An introduction to pain pathways and mechanisms. British journal of hospital medicine. 2013 Dec;74(Sup12):C188-91.Available:https://www.ucl.ac.uk/anaesthesia/sites/anaesthesia/files/IntrotoPainPathwaysandMechanisms.pdf (accessed 18.4.2022)
- ↑ 7.0 7.1 Beecher HK. Pain in men wounded in battle. Ann Surg. 1946;123(1):96-105
- ↑ 8.0 8.1 Bingel U, Tracey I. Imaging CNS modulation of pain in humans. Physiolohy 2008;23:371-380
- ↑ Moseley G Combined physiotherapy and education is efficacious for chronic low back pain. Australian Journal of Physiotherapy 2002; 48:297–302
- ↑ Louw A, Louw Q, Crous LCC. Preoperative Education for Lumbar Surgery for Radiculopathy. South African Journal of Physiotherapy. 2009;65(2):3-8.
- ↑ Moseley, GL. A randonmised controlled trial of intensive neurophysiology education in chronic low back pain. Clin J Pain 2002;20:324-330
- ↑ Meeus MJ. Pain physiology education improves pain beliefs in patients with chronic fatigue syndrome compared with pacing and self-management education:a double-blind randomised controlled trial. Arch Phys Med Rehabil 2010;91:1153-1159
- ↑ Clarke CL. Pain Neurophysiology education for the management for the management of individuals with chronic low back pain: systematic review and meta-analysis. Manual Therapy 2011;16:544-549
- ↑ Louw A. The effect of neuroscience education on pain and disability, anxiety, and stress in chronic musculoskeletal pain. Archives of Physical Medicine and Rehabilitation 2011;92:2041-2056
- ↑ Wright A. Hypoalgesia post manipulative therapy: a review of a potential neurophysiological mechanism. Man Ther 1995;1(1), 11-16
- ↑ Bishop MD, Torres-Cueco R, Gay CW, Lluch-Girbés E, Beneciuk JM, Bialosky JE. What effect can manual therapy have on a patient's pain experience?. Pain management. 2015 Nov;5(6):455-64. Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4976880/ (accessed 18.4.2022)