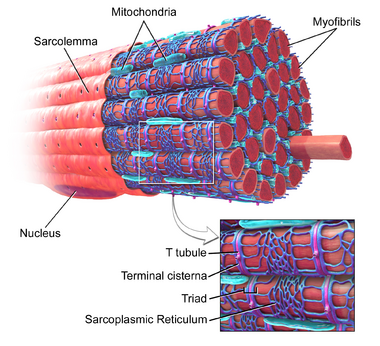
Muscle Proteins
Original Editor - lucinda hampton
Top Contributors - Lucinda hampton
Introduction[edit | edit source]
Proteins are the basic material of tissue structure. They are the most important component of striated skeletal muscle.
The total amount of muscle proteins in humans exceeds that of any other protein. About 40 percent of the body weight of a healthy human adult weighing about 70 kilograms (150 pounds) is muscle, which is composed of about 20 percent muscle protein. Thus, the human body contains about 5 to 6 kilograms (11 to 13 pounds) of muscle protein[1].
Classification[edit | edit source]
The muscle proteins can be divided in to myofibrillar, regulatory, sarcoplasmic and stromal proteins
- Myofibrillar Proteins: Muscle fibers are composed of myofibrils. The proteins that comprise the myofibril, including actin and myosin and several more. The myofibrillar protein components most important for muscle fiber structure are actin and myosin. They are the most abundant proteins in muscle and are directly involved in the ability of muscle to contract and to relax[2]. Myosin constitutes as much as 35% of the total protein volume of skeletal muscles whilst actin is the most abundant protein in most eukaryotic cells.[3].
- Regulatory proteins: Include troponin, tropomyosin, M-protein, beta-actin, gamma-actin and C-protein. The are all important eg troponin-tropomyosin is responsible, in part, not only for transducing (ie converting to an electrical form) the effect of calcium on contractile protein activation, and also for inhibiting actin and myosin interaction when calcium is absent.[4]
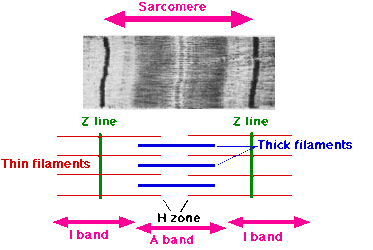
- Image 3: Sarcomere.The sarcoplasmic proteins include hemoglobin and myoglobin pigments and a wide variety of enzymes. eg myogen, myoalbumin and x-globulin. Pigments from hemoglobin and myoglobin help to contribute the red color to muscle. Hemoglobin carries oxygen from the lungs to the tissues— including muscle. Myoglobin is present in muscle and it stores the oxygen transported to the muscle via the blood by hemoglobin until it is utilized in metabolism[2]. It has a dominant effect together with the mitochondria on the aerobic potential of muscles.[5]
- Stromal Proteins: Connective tissue is composed of a watery substance into which is dispersed in a matrix of stromal- protein fibrils. These stromal proteins are collagen, elastin (a key protein of the extracellular matrix) and reticulin (protein substance similar to collagen)[2].
- The remaining of proteins includes myofibrillar proteins of the Z-disc as well as small quantities of other proteins[5].
Muscle Protein Synthesis[edit | edit source]
Muscle protein synthesis (MPS) is the driving force behind adaptive responses to exercise and represents a widely adopted proxy for gauging chronic efficacy of acute interventions, (i.e. exercise/nutrition).
- In healthy, recreationally active individuals, skeletal muscle proteins display turnover rates of ∼1.2% day-1 and exist in dynamic equilibrium.[6]
- An acute exercise stimulus, especially resistance exercise, and protein ingestion both stimulate MPS and work in synergy when protein consumption occurs before or after resistance exercise.
- For building muscle mass and for maintaining muscle mass an overall daily protein intake in the range of 1.4–2.0 g protein/kg body weight/day (g/kg/d) is sufficient for people exercising.
- New evidence that proposes that higher protein intakes (>3.0 g/kg/d) may have positive effects on body composition in resistance-trained individuals. That is it promotes loss of fat mass.
- The ideal protein intake per serving for athletes to maximise MPS vary but common recommendations are 0.25 g of a high-quality protein per kg of body weight, or an absolute dose of 20–40 g. These protein doses should ideally be evenly distributed, every 3–4 h, across the day.
- Physically active individuals may obtain their daily protein requirements through the consumption of whole foods, supplementation is an option to ensure intake of adequate protein quality and quantity, without too many calories.
- Athletes should consider focusing on whole food sources of protein that contain all of the EAAs as they stimulate MPS.
- Pre-sleep casein protein intake (30–40 g) provides increases in overnight MPS and metabolic rate without influencing lipolysis.
- The relationship between diet and protein balance is not straightforward. Even with increased protein intake, MPS is triggered for only a finite period of time. This is because the body can only utilize so much of the essential amino acids (EAAs) it receives; anything more will be broken down and excreted by the liver.[7]. However the anabolic effect of exercise is long-lasting (at least 24 h), but likely diminishes with increasing time post-exercise.[8]
See more: Muscle Function and Protein and Exercise and Protein
Physiotherapy[edit | edit source]
Educate clients about the importance of muscle mass.
It is recommend a two-phase approach to limit inactivity-mediated losses of muscle mass and function in older adults.
- Lifestyle: Educate re diet, they should eat a moderate amount (25-30 g) of high-quality protein with each meal and incorporate habitual exercise in close temporal proximity to protein-containing meals.
- Illness times eg hospital stay. To combat the accelerated loss of muscle mass and function during acute catabolic crises and periods of reduced physical activity educate clients that they should include nutritional support such as targeted protein or amino acid supplementation and integrated physical therapy.
References[edit | edit source]
- ↑ Britannica Muscle Proteins Available:https://www.britannica.com/science/protein/The-muscle-proteins (accessed 24.11.2021)
- ↑ 2.0 2.1 2.2 Belk, K., 2015. Does Muscle Tissue Contain Different Types of Protein. Champaign, IL: American Meat Science Association). Available:https://meatscience.org/TheMeatWeEat/topics/fresh-meat/article/2015/07/31/does-muscle-tissue-contain-different-types-of-protein (accessed 24.11.2021)
- ↑ Dominguez R, Holmes KC. Actin structure and function. Annual review of biophysics. 2011 Jun 9;40:169-86.Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3130349/ (accessed 24.11.2021)
- ↑ Malhotra A. Role of regulatory proteins (troponin-tropomyosin) in pathologic states. Molecular and cellular biochemistry. 1994 Jun;135(1):43-50.Available:https://pubmed.ncbi.nlm.nih.gov/7816055/ (accessed 24.11.2021)
- ↑ 5.0 5.1 Makovický P, Jílek F. Short review of some properties of muscular proteins. Ceskoslovenska fysiologie. 2008 Jan 1;57(1):10-4.Available:https://pubmed.ncbi.nlm.nih.gov/18630139/ (accessed 24.11.2021)
- ↑ Atherton PJ, Smith K. Muscle protein synthesis in response to nutrition and exercise. The Journal of physiology. 2012 Mar 1;590(5):1049-57.Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3381813/(accessed 24.11.2021)
- ↑ Vey well fit Is Muscle Protein Synthesis the Same as Growth? Available: https://www.verywellfit.com/muscle-protein-synthesis-and-muscle-growth-4148337(accessed 24.11.2021)
- ↑ Jäger R, Kerksick CM, Campbell BI, Cribb PJ, Wells SD, Skwiat TM, Purpura M, Ziegenfuss TN, Ferrando AA, Arent SM, Smith-Ryan AE. International society of sports nutrition position stand: protein and exercise. Journal of the International Society of Sports Nutrition. 2017 Jun 20;14(1):20. Available:https://jissn.biomedcentral.com/articles/10.1186/s12970-017-0177-8#Sec33 (accessed 23.11.2022)