Bone Healing
Original Editor - Jess Bell
Top Contributors - Jess Bell, Khloud Shreif, Kim Jackson, Tarina van der Stockt and Olajumoke Ogunleye
Introduction[edit | edit source]
Bone fractures are a common injury and the healing process is complex.[1] Bone is one of a few tissues that is able to heal without forming a fibrous scar.
"Bone healing is an intricate regenerative process which can be classified into primary (direct) and secondary (indirect) bone healing."[2]
Direct/ primary healing occurs when the bony fragments are fixed together with compression. There is no callus formation. The bony ends are joined and healed by osteoclast and osteoblast activity.[1]
Indirect healing is more common than direct healing and involves both endochondral and intramembranous bone healing.[3] Anatomical reduction and stable conditions are not required for indirect healing to occur.[3] Rather, there is a small amount of motion and weight-bearing at the fracture, which causes a soft callus to form, leading on to secondary bone formation.[1] It should be noted though that too much load/movement can result in delayed healing or non-union,[3] which occurs in 5-10% of all fractures.[4]
Indirect healing usually occurs with:
- Non-operative fracture treatment
- Operative treatments where some motion occurs at the fracture site, such as:
- Intramedullary nailing
- External fixation
- Internal fixation of comminuted fractures.[3]
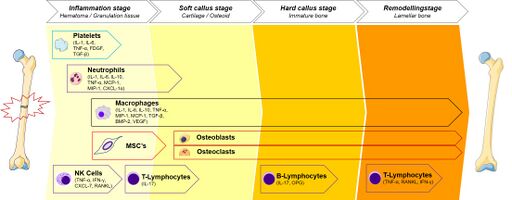
Stages of Indirect Healing[edit | edit source]
Acute Inflammatory Response[edit | edit source]
The acute inflammatory response peaks within 24 hours and ends after 7 days and is essential for healing to occur.[3] A haematoma forms immediately after trauma. This consists of cells from the peripheral and intramedullary blood and bone marrow cells. The inflammatory response causes the haematoma to coagulate around the fracture ends and within the medulla, which creates a model for callus formation.[3]
Tumor necrosis factors TNF-α, interleukin-1,IL-6, IL-11, and IL-18 are releases to maintain and promote blood vessels growth[5].
Recruitment of Mesenchymal Stem Cells[edit | edit source]
Bone is unable to regenerate unless specific mesenchymal stem cells are recruited, proliferated, and differentiated into osteogenic cells. It is not currently understood exactly where these cells come from.[3]
Generation of Cartilaginous and Periosteal Bony Callus[edit | edit source]
After the hematoma has formed, a fibrin-rich granulation tissue forms. Endochondral formation occurs between the fracture ends and beyond the periosteal sites in this tissue. These areas are less stable, so the cartilaginous tissue forms a soft callus, giving the fracture more stability.[3]
In animal studies, soft callus formation peaks at 7 to 9 days when type II procollagen and proteoglycan core protein extracellular markers are at their highest levels.[3] Concurrently, an intramembranous ossification response occurs subperiosteal immediately by the fracture ends. This creates a hard callus. The bridging of this central hard callus provides the fracture with a semi-rigid structure which enables weight-bearing.[3]
Revascularization and Neoangiogenesis[edit | edit source]
Adequate blood supply is necessary for bone repair to occur. Angiogenic pathways, chondrocyte apoptosis, and cartilaginous degradation are essential to this process because cells and extracellular matrices must be removed in order to ensure that blood vessels can move into the repair site.[3]
Mineralisation and Resorption of the Cartilaginous Callus[edit | edit source]
The primary soft cartilaginous callus must be resorbed and replaced by a hard bony callus for bone regeneration to continue.[3] In some ways, this stage repeats embryological bone development and involves cellular proliferation and differentiation, as well as an increase in cellular volume and matrix deposition.[3]
Bone Remodelling[edit | edit source]
While the hard callus is rigid and provides stability, it does not mean that the fracture site has all the properties of normal bone. A second restorative stage is necessary. This stage results in the remodeling of the hard callus into a lamellar bone structure with a central medullary cavity.[3]
Remodeling occurs when the hard callus is resorbed by osteoclasts and lamellar bone is deposited by osteoblasts. This starts at 3–4 weeks, but the whole process may take years. Remodeling may be faster in younger patients (and other animals).[3]
Bone remodeling results from the production of electrical polarity. This occurs when pressure is applied in a crystalline environment.[3]
- When axial loading of long bones occurs, an electropositive convex surface and an electronegative concave surface are created
- This activates osteoclastic and osteoblastic activity.
- As a result, the external callus is slowly replaced by a lamellar bone structure. As well as this, the internal callus remodels which re-creates a medullar cavity, similar to diaphyseal bone.[3]
Bone remodeling will only be successful if there is adequate blood supply and a gradual increase in mechanical stability. If not, complications such as non-union may occur.[3]
Direct Fracture Healing[edit | edit source]
Direct healing requires reduction of the fracture ends, without any gap formation, as well as stable fixation. Thus, it does not usually occur naturally but rather following open reduction and internal fixation surgery.[3]
Direct bone healing can occur by direct remodeling of lamellar bone, the Haversian canals, and blood vessels. The process usually takes from months to years.[3]
Primary healing of fractures occurs through:
- Contact healing
- Gap healing.
Both processes consist of an attempt to re-create lamellar bone structure. Direct bone healing is possible only when the fracture ends are compressed together and rigid fixation is used to decrease interfragmentary strain.[3][1]
Contact Healing[edit | edit source]
A fracture can unite by contact healing when the gap between each bone end is less than 0.01 mm and interfragmentary strain is less than 2%.[3] In such instances cutting cones form at the ends of the osteons by the fracture site. The tips of the cutting cones consist of osteoclasts. These tips cross the fracture line and generate longitudinal cavities.[3]
The cavities are eventually filled by bone that is produced by osteoblasts. This causes the bony union to generate while also restoring the Haversian systems, which are formed in an axial direction. The Haversian systems enable blood vessels carrying osteoblasts to enter the area. Bridging osteons eventually mature into lamellar bone, which results in fracture healing without a periosteal callus forming.[3]
Gap Healing[edit | edit source]
Gap healing is unique in that bony union and Haversian remodeling does not happen at the same time.[3] For gap healing to occur, the gap must be less than 800 μm to 1 mm.[3]
During this process, the fracture site is filled largely by lamellar bone which runs perpendicular to the long axis and needs secondary osteonal reconstruction. The primary bone structure is eventually replaced by longitudinal revascularized osteons, which carry osteoprogenitor cells that differentiate into osteoblasts. These osteoblasts then produce lamellar bone on each surface of the gap.[3] The lamellar bone is laid down perpendicular to the long axis, which means that it is not strong. This process takes between 3 and 8 weeks. After this, a secondary remodeling phase occurs, which is similar to the cascade with cutting cones in contact healing.[3]
Factors Affect Bone Healing[edit | edit source]
- Patient with nutritional deficits, smoking, and diabetic patient will experience delayed bone healing.
- Parathyroid hormones have a vital role in bone healing by stimulating the differentiation and proliferation of osteoblasts and osteoclasts.
- Aging process in which the patient will have:
- Chronic inflammation or the delay in resolving the inflammation to prepare for the anabolic process affect healing and cause a small callus formation, in addition to higher levels of circulating pro-inflammatory cytokines.
- Patient at this age will show a decrease in the quantity of the muscle stem cells and negatively affect affect the healing process.
- Decrease in the bone marrow blood vessels compared to young age and decrease in vascularization and angiogenesis[6].
- Factors related to the fracture site:
- Infection of the fracture site at healing process.
- Insufficient formation of cartilage within fracture gap and marrow space.
- Open, comminuted fracture, and the extent of soft tissue injury also show delayed union healing[7].
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 Ghiasi MS, Chen J, Vaziri A, Rodriguez EK, Nazarian A. Bone fracture healing in mechanobiological modeling: A review of principles and methods. Bone Rep. 2017; 6: 87-100.
- ↑ Maruyama M, Rhee C, Utsunomiya T, Zhang N, Ueno M, Yao Z, Goodman SB. Modulation of the Inflammatory Response and Bone Healing. Front Endocrinol (Lausanne). 2020 Jun 11;11:386.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 3.19 3.20 3.21 3.22 3.23 3.24 3.25 3.26 3.27 Marsell R, Einhorn TA. The biology of fracture healing. Injury. 2011; 42(6): 551-555.
- ↑ Buza JA, Einhorn T, Bone healing in 2016. Clin Cases Miner Bone Metab. 2016; 13(2): 101-105.
- ↑ Beckmann R, Tohidnezhad M, Lichte P, Wruck CJ, Year H, Pape HC, Pufe T. From old to new.relevant factors for fracture healing in aging bone. The orthopedist. 2014 Apr; 43 (4): 298-305.
- ↑ Clark D, Nakamura M, Miclau T, Marcucio R. Effects of aging on fracture healing. Current osteoporosis reports. 2017 Dec 1;15(6):601-8.
- ↑ Kostenuik P, Mirza FM. Fracture healing physiology and the quest for therapies for delayed healing and nonunion. Journal of Orthopaedic Research®. 2017 Feb;35(2):213-23.
- ↑ Osmosis. Bone remodeling and repair. Available from: http://www.youtube.com/watch?v=Ei4seya3dOg[last accessed 8/8/2021]