Spasticity Management in Spinal Cord Injury
Original Editor - Ewa Jaraczewska based on the course by Melanie Harding
Top Contributors - Ewa Jaraczewska, Jess Bell and Kim Jackson
Introduction[edit | edit source]
Spasticity is a common but often underdiagnosed symptom in people with a spinal cord injury (SCI).[1] According to Holtz et al.,[2] 35% of patients with problematic spasticity require some kind of management within one year of their spinal cord injury.[2] Problematic spasticity can be painful and interfere with an individual's mobility and function, including personal hygiene or toileting. It is most common in patients diagnosed with ASIA grades B through D at the cervical level. However, some individuals can benefit from spasticity as it can help facilitate trunk stability for postural control and enhance activities such as standing, transfers, and other activities of daily living.[3] This article discusses the various definitions of spasticity, assessment methods and options for managing spasticity in spinal cord injury.
Definitions of Spasticity[edit | edit source]
According to the European consensus on the concepts and measurement of the pathophysiological neuromuscular responses to passive muscle stretch,[4] the term spasticity or hypertonia due to an impaired neuromuscular response during passive stretch should be replaced by the term hyper-resistance. They further distinguish between non-neutral (or tissue-related) and neural (or central nervous system-related) contributions to hyper-resistance:
- "Tissue contributions are elasticity, viscosity and muscle shortening."[4]
- "Neural contributions are velocity-dependent stretch hyperreflexia and non-velocity dependent involuntary background activation".[4]
Definitions of spasticity include:
“A motor disorder characterised by a velocity-dependent increase in tonic stretch reflexes (muscle tone) with exaggerated tendon jerks, resulting from hyperexcitability of the stretch reflex, as one component of the upper motor neuron syndrome” -- JW Lance, 1980[5]
"Disordered sensorimotor control, resulting from an upper motor neurone lesion, presenting as intermittent or sustained involuntary activation of muscles." -- 2005 SPASM (Support Programme for Assembly of a Database for Spasticity Measurement) consortium[6]
“Spasticity refers to velocity-dependent stretch hyperreflexia as part of hyper-resistance.” -- 2016 European consensus[4]
Clinicians must be aware that the term spasticity can only be used when it is clearly defined by the type of changes that have occurred. The term stiffness is reserved for the tissue-related contributions to hyper-resistance.[4]
Neurological Components of Spasticity[edit | edit source]
- Upper motor neuron lesion in the spinal cord[7]
- Disinhibition occurs at the spinal reflex loop as a direct result of the neurological insult[4]
Spasticity vs Muscle Spasms[edit | edit source]
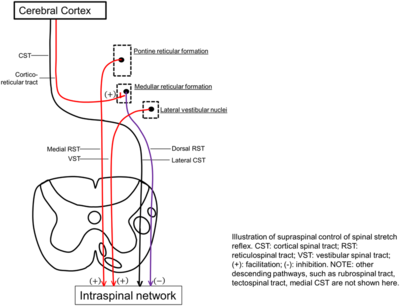
Spasticity is caused by a loss of supraspinal inhibitory modulation of muscle stretch reflexes. Supraspinal inhibitory modulation of these reflexes is normally provided by the corticospinal, corticoreticular, and dorsal reticulospinal tracts.[8]
Muscle spasms are muscular contractions that are involuntary, uncontrolled, periodic, and non-sustained.
Epidemiology of Spasticity[edit | edit source]
- Spasticity occurs more in patients with tetraplegia[7]
- 48% of patients with SCI present with spasticity upon admission to acute inpatient rehabilitation units[9]
- 46-65% of persons with traumatic SCI reported spasticity at discharge[3][9]
- Problematic spasticity has been reported by 79% of patients with cervical SCI, 69% of patients with thoracic SCI and 22% of patients with high lumbar SCI[3]
- Spasticity is less common in lesions between T12-L2[3]
- Spasticity develops in:
- 87% of patients with a cervical injury
- 85% of patients with a thoracic injury
- 57% of patients with lumbar injury
- Spasticity develops in:
- No spasticity develops with lesions below L2[7]
- Medications are commonly used to manage spasticity in a spinal cord injury[9]
Read about facts and figures on spinal cord injury here.
Phases in Spasticity Development[edit | edit source]
- Initial phase of areflexia[10]
- Spinal shock: acute SCI with flaccid tone below the level of injury[11]
- Return of reflexes[10]
- Hyperreflexia, spasms, and clonus emerge[10]
Spasticity Assessment[edit | edit source]
Considerations prior to the assessment:[7]
- spasticity may vary throughout the day, week and even during a treatment session.
- it can be present in all four limbs and the trunk. It may be in the flexors, extensors, rotators, adductors or abductors. All assessments should be performed with the muscle at rest (relaxed) for accurate results.
- spasticity can increase or decrease due to specific movements, activities, stressors or positions.
- it is necessary to assess the effect of spasticity on function and activities of daily living (e.g. gait, bed mobility, transfers, sleep, bladder and bowel function).
The goals of a spasticity assessment are to:[7]
- understand the patient and the caregiver's perception of spasticity
- identify where the problem may be
- validate how realistic the desired outcome is
- evaluate treatment interventions[12]
Outcome Measures[edit | edit source]
While there are still challenges associated with measuring spasticity, the following methods can be used:[13]
1. Clinical scales
- Modified Ashworth Scale (MAS)[14]
- six-point ordinal scale for grading resistance encountered during passive movement stretching at increasing velocity
- 0 = no increase in muscle tone
- 1 = slight increase in muscle tone, manifested by a catch and release or by minimal resistance at the end of the range of motion (ROM) when the affected part(s) is moved in flexion or extension
- 1+ = slight increase in muscle tone, manifested by a catch, followed by minimal resistance throughout the remainder (less than half) of the ROM
- 2 = more marked increase in muscle tone through most of the ROM, but the affected part(s) is easily moved
- 3 = considerable increase in muscle tone; passive movement is difficult
- 4 = affected part(s) rigid in flexion or extension
- has adequate reliability for assessing lower-extremity spasticity in patients with SCI
- the MAS is available here
- six-point ordinal scale for grading resistance encountered during passive movement stretching at increasing velocity
- Modified Tardieu Scale (MTS)[15]
- validated in SCI
- assesses three key components:
- velocity of stretch
- quality of the muscle reaction
- angle of the muscle reaction
- the MTS is considered superior to the MAS
- it can distinguish between neural and peripheral contributions to spasticity
- quantifies "muscle tone at specified velocities"
- quantitative joint angle measurements are measured at three speeds of passive movements:[15]
- very slow
- "passive fall of the limb under the influence of gravity"[15]
- as fast as possible.
- acts as a complementary tool for planning the treatment for people with SCI
- the MTS is available here
- Penn Spasm Frequency Scale (PSFS)
- a self-report measure of the frequency of muscle spasms
- commonly used to quantify spasticity
- contains two parts: spasm frequency and spasm severity
- spasm frequency:
- 0 = no spasms
- 1 = one or a few spasms per day
- 2 = between 1 and 5 spasms per day
- 3 = between 5 and <10 spasms per day
- 4 = 10 or more spasms per day or continuous contraction
- spasm severity:
- mild (1)
- moderate (2)
- severe (3)
- poorly correlated with MAS
- more information about the PSFS can be found here.
- Spastic Paraplegia Rating Scale (SPRS)
- takes 15 minutes to administer
- does not require any special equipment[16]
- has a high interrater correlation
- contains 13 variables to measure the severity of spastic paraplegia[17]
- rates functional impairment by assessing walking ability, muscle power, spasticity, pain and urinary function[18]
- a score is given for each variable, ranging from 0 (normal) to 4 (severe impairment)[17]
- the maximum total score is 52
2. Biomechanical methods
- Pendulum Test
- a simple method to quantify the severity of spasticity
- the patient is seated or lying, and their leg is hanging over the end of a bed / couch
- the tester extends the leg and then releases it, so that it swings freely
- the amount of swing is generally reduced in individuals with spasticity
- this test can be quantified by using goniometers to measure the ratio between the initial flexion angle and the final position of the knee[19]
- results depend on the patient's sitting position and their ability to fully relax fully[19]
- this test can only evaluate spasticity in the knee muscles
- this test cannot "dissociate increased resistance in the muscle due to viscoelastic changes from the velocity-dependent resistance due to spasticity"
3. Neurophysiological-Electrophysiological methods
- Spinal Cord Assessment Tool for Spastic Reflexes (SCATS)
Characteristics of Spasticity in SCI[edit | edit source]
Spasticity can negatively influence a person's quality of life by limiting their ability to perform activities of daily living, inhibiting self-care, causing pain and affecting safety. However, spasticity can have some benefits, such as improving trunk stability in sitting and standing, facilitating transfers and walking, and increasing venous return, thus reducing the incidence of deep vein thrombosis. The presence and severity of spasticity may vary throughout the day, and well-known factors can aggravate or alleviate the presence and symptoms of spasticity.
Aggravating Factors[edit | edit source]
- Immobility or remaining too long in one position
- Pain (fracture, muscle overuse injury, pressure sore, kidney stone, bladder infection, appendicitis, labour)
- Overfull bladder or bowel
- Constipation
- Various infections: urinary tract infection, ingrown toenail, and decubitus ulcer
- Anxiety, stress / emotional disorders
- Sudden heat and moisture changes in the environment (e.g. cold weather, coming rain)
- Pressure on the ball of the foot (stimulates the plantar reflex)
- Bed or wheelchair positioning with not enough hip and knee flexion
- Heterotrophic ossification (HO)
- Contractures
- Syringomyelia
- Deep-vein thrombosis
- Fever
- Tight-fitting clothes or urinary leg bag straps
- Uncomfortable orthotics
Alleviating Factors[edit | edit source]
- Slow passive movements and stretches when performed regularly[7][21]
- Standing and weight bearing[22]
- Rhythmical trunk side flexion and extension combined with trunk torsion (e.g. during horse riding)[23]
- Physical activity and treatment, including stretching and bending[24]
- Proper positioning in bed and chair
- Pain management
- Bladder and bowel management
- Stress management
- Deep breathing and relaxation
- Cold / ice[21]
General Principles in Spasticity Management[edit | edit source]
Spasticity should be addressed when it is detrimental to the patient, causing issues such as pain, functional or positional limitations or muscle contracture.[3]
Early interventions for preventing and treating post-spinal cord injury spasticity are recommended.[10]
When planning treatment for spasticity, clinicians should consider the following factors:[3]
- quality of the spasticity
- severity of the spasticity
- distribution of spasticity
- alleviating factors
- aggravating factors
- a patient's perception of / explanation for their spasticity
Therapeutic Management of Spasticity[edit | edit source]
Self-Management[edit | edit source]
Self-management is an essential part of the therapeutic management of spasticity. The patient and caregivers must understand the patient's medical conditions and potential therapeutic interventions in order to select an effective treatment and achieve the best outcome.[25] The patient should be instructed on self-management techniques for spasticity control. Patient education should cover the following:
- how to recognise and prevent factors that may aggravate spasticity and spasms[25]
- performing regular stretching, including a prone position stretch, to maintain muscle length
- pushing down on the knee to flatten the foot on the floor or the footplate to stop clonus
- how to use spasticity for function
Weight-Bearing[edit | edit source]
The long-term effectiveness of weight-bearing activities has not been well-researched and is frequently questioned.[26] A 1993 case study by Bohannon[27] found an immediate reduction in lower extremity spasticity after weight-bearing interventions (i.e. using a tilt table), which lasted a few hours after treatment. There was also a reduction in spasms, which lasted until the morning after treatment.
Tilt table or standing frame:
- The benefits of using a tilt table or standing frame exceed manual stretching alone
- Stretching while standing influences cutaneous and joint receptor input to the spinal motor neurons, which results in decreased excitability
- A prolonged stretch of the ankle plantar flexor muscles in a standing position causes a reduction in spasticity in the lower extremities
Positioning[edit | edit source]
Appropriate positioning in bed or in a wheelchair is vital to maintain muscle length. While there are clinical reports on the effectiveness of positioning on spasticity reduction, scientific proof is lacking.[26] There are also specific factors that can cause an increase in spasticity for individuals with SCI sitting in a wheelchair:
- poor trunk stability and the effort required to compensate for it
- pain as a result of poor positioning
- muscle shortening due to poor positioning can become a trigger if stretched
To decrease the impact of abnormal tone, we must consider the following:
- standard solutions for wheelchair positioning cannot usually be applied[28]
- a team approach between occupational therapists and physiotherapists is essential
- knowledge of the patient's condition and physical impairments, wheelchair adaptation, seating systems and cushions is a must
- understanding a patient's wishes / requirements should influence the team's decision
- a golden rule of positioning is to change positions throughout the day[25]
This section in the Positioning and General Management of Upper Limbs in Spinal Cord Injury course discusses positioning in supine and in the wheelchair in more detail.
Upper Limb Spasticity Management[edit | edit source]
You can learn more about upper limb spasticity management here.
Modalities[edit | edit source]
This article discusses the use of modalities in upper limb management in tetraplegia.
Splinting[edit | edit source]
- A palmar-dorsal splint can be recommended when there is increased tone in the fingers and/or wrist.
- Resting splint: a night splint can be used to keep the hand in a functional position with the wrist and fingers slightly flexed.
You can find more information on splinting for individuals with spinal cord injury in this guideline.
Medication for Pain, Spasticity and Infection Depending on Cause[edit | edit source]
Baclofen tablets:[24]
- only a small portion of the active substance penetrates the blood-brain barrier, so it has a limited effect
- has strong side effects, including sedation, nausea, dizziness and difficulty in breathing
- a sudden interruption in taking this medication may cause epileptic seizures, psychosis and hyperthermia
Tizanidine:
- offers a pronounced muscle relaxant effect
- suppresses polysynaptic reflexes in a complete SCI
- can be combined with baclofen[29]
- has the same side effects as baclofen
Cannabis products:
- conflicting results[24]
- according to Karst et al., cannabis "is not recommended for treating spasticity because of the narrow therapeutic range and risk of side effects and dependence"[30]
Botulinum toxin injections:
- used when spasticity is present in a few muscles or a limited muscle group
- temporarily blocks the connection between the nerve terminal and muscle fibre by stopping the presynaptic release of acetylcholine from nerve terminals
- thorough assessment with a detailed analysis and description of function is essential for a good outcome
- medication is delivered into the subarachnoid space by a programmable pump via a catheter system
- the pump must be refilled, and the dose of the medicine can be adjusted
Surgical Management[edit | edit source]
- Z-plasty or tenotomies to lengthen the muscle:[31][32]
- The most common indications for tendon surgery in the lower extremity include:[32]
- hip flexion, hip adduction
- knee flexion contracture, knee extension contracture
- equinovarus at the ankle
- flexion contracture of the toe and, sometimes, hallux extension
- In the upper extremity, tendons commonly released include flexor digitorum profundus and superficialis, as well as flexor carpi radialis and flexor pollicis longus. A Z-lengthening of flexor carpi ulnaris may be used if necessary.[31]
- The most common indications for tendon surgery in the lower extremity include:[32]
Positive Effects of Spasticity on Function[edit | edit source]
Gait training:
- Extensor spasticity can assist with gait, sit-to-stand and other transfers. However, severe spasticity increases energy usage, limits walking speed and affects safety. These factors may outweigh the advantages of walking over the speed and ease of using a wheelchair.[7]
- More information on gait after spinal cord injury can be found here.
Bed mobility:
- Trunk spasticity can facilitate rolling over or sitting up[7]
Transfers:
- Spasticity can assist with lifting legs up onto a bed[7]
Coughing:
- Spasticity can be used to stimulate a more effective cough[7]
Hand Grip:
- Spasticity in the finger flexors can be stimulated to assist in strengthening the tenodesis grip[7]
Resources[edit | edit source]
- Biering-Sørensen F, Nielsen JB, Klinge K. Spasticity-assessment: a review. Spinal Cord. 2006 Dec;44(12):708-22.
- Balci BP. Spasticity Measurement. Noro Psikiyatr Ars. 2018;55(Suppl 1):S49-S53.
- Postural Assessment And Seating Systems for People with Spinal Cord Injury.
- Positioning and General Management of Upper Limbs in Spinal Cord Injury
- SPINAL CORD INJURY GUIDELINES 2019
- Paralyzed Veterans of America Consortium for Spinal Cord Medicine. Preservation of upper limb function following spinal cord injury: a clinical practice guideline for health-care professionals. J Spinal Cord Med. 2005;28(5):434-70.
References[edit | edit source]
- ↑ Baunsgaard CB, Nissen UV, Christensen KB, Biering-Sørensen F. Modified Ashworth scale and spasm frequency score in spinal cord injury: reliability and correlation. Spinal Cord. 2016 Sep;54(9):702-8.
- ↑ 2.0 2.1 Holtz KA, Lipson R, Noonan VK, Kwon BK, Mills PB. Prevalence and Effect of Problematic Spasticity After Traumatic Spinal Cord Injury. Arch Phys Med Rehabil. 2017 Jun;98(6):1132-1138.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Billington ZJ, Henke AM, Gater DR Jr. Spasticity Management after Spinal Cord Injury: The Here and Now. J Pers Med. 2022 May 17;12(5):808.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 Noort JCVD, Bar-On L, Aertbeliën E, Bonikowski M, Braendvik SM, Broström EW, et al. European consensus on the concepts and measurement of the pathophysiological neuromuscular responses to passive muscle stretch. Eur J Neurol. 2017.
- ↑ Lance JW. Symposium synopsis. In Feldman RG, Young RR, Koella WP, editors. Spasticity: disordered motor control. Yearbook Medical, Chicago; 1980, pp. 485–494.
- ↑ Pandyan AD, Gregoric M, Barnes MP, Wood D, Van Wijck F, Burridge J, Hermens H, Johnson GR. Spasticity: clinical perceptions, neurological realities and meaningful measurement. Disabil Rehabil. 2005 Jan 7-21;27(1-2):2-6.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 Harding M. General Principles of Spasticity Management in Spinal Cord Injury. Plus 2023
- ↑ Billington ZJ, Henke AM, Gater DR Jr. Spasticity management after spinal cord injury: the here and now. J Pers Med. 2022 May 17;12(5):808.
- ↑ 9.0 9.1 9.2 Dragojlovic N, Romanoski NL, Verduzco-Gutierrez M, Francisco GE. Prevalence and Treatment Characteristics of Spastic Hypertonia on First-Time Admission to Acute Inpatient Rehabilitation. Am J Phys Med Rehabil. 2022 Apr 1;101(4):348-352.
- ↑ 10.0 10.1 10.2 10.3 Stampas A, Hook M, Korupolu R, Jethani L, Kaner MT, Pemberton E, Li S, Francisco GE. Evidence of treating spasticity before it develops: a systematic review of spasticity outcomes in acute spinal cord injury interventional trials. Ther Adv Neurol Disord. 2022 Feb 17;15:17562864211070657.
- ↑ Ko HY. Revisit Spinal Shock: Pattern of Reflex Evolution during Spinal Shock. Korean J Neurotrauma. 2018 Oct;14(2):47-54.
- ↑ Rahimi F, Eyvazpour R, Salahshour N, Azghani MR. Objective assessment of spasticity by pendulum test: a systematic review on methods of implementation and outcome measures. Biomed Eng Online. 2020 Nov 9;19(1):82.
- ↑ Balci BP. Spasticity Measurement. Noro Psikiyatr Ars. 2018;55(Suppl 1):S49-S53.
- ↑ Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther. 1987 Feb;67(2):206-7.
- ↑ 15.0 15.1 15.2 Akpinar P, Atici A, Ozkan FU, Aktas I, Kulcu DG, Sarı A, Durmus B. Reliability of the Modified Ashworth Scale and Modified Tardieu Scale in patients with spinal cord injuries. Spinal Cord. 2017 Oct;55(10):944-949.
- ↑ Schüle R, Holland-Letz T, Klimpe S, Kassubek J, Klopstock T, Mall V, Otto S, Winner B, Schöls L. The Spastic Paraplegia Rating Scale (SPRS): a reliable and valid measure of disease severity. Neurology. 2006 Aug 8;67(3):430-4.
- ↑ 17.0 17.1 Galvão CRC, Cavalcante PMA, Olinda R, Graciani Z, Zatz M, Kok F, Santos S, Lancman S. Motor impairment in a rare form of spastic paraplegia (Spoan syndrome): a 10-year follow-up. BMC Neurol. 2019 Oct 27;19(1):256.
- ↑ Chou CT, Soong BW, Lin KP, Tsai YS, Jih KY, Liao YC, Lee YC. Clinical characteristics of Taiwanese patients with Hereditary spastic paraplegia type 5. Ann Clin Transl Neurol. 2020 Apr;7(4):486-496.
- ↑ 19.0 19.1 Biering-Sørensen F, Nielsen JB, Klinge K. Spasticity-assessment: a review. Spinal Cord. 2006 Dec;44(12):708-22.
- ↑ Akpinar P, Atici A, Ozkan FU, Aktas I, Kulcu DG, Kurt KN. Reliability of the Spinal Cord Assessment Tool for Spastic Reflexes. Arch Phys Med Rehabil. 2017 Jun;98(6):1113-1118.
- ↑ 21.0 21.1 Adams MM, Hicks AL. Spasticity after spinal cord injury. Spinal cord. 2005 Oct;43(10):577-86.
- ↑ Adams MM, Hicks AL. Comparison of the effects of body-weight-supported treadmill training and tilt-table standing on spasticity in individuals with chronic spinal cord injury. The journal of spinal cord medicine. 2011 Sep 1;34(5):488-94.
- ↑ Lechner HE, Feldhaus S, Gudmundsen L, Hegemann D, Michel D, Zäch GA, Knecht H. The short-term effect of hippotherapy on spasticity in patients with spinal cord injury. Spinal cord. 2003 Sep;41(9):502-5.
- ↑ 24.0 24.1 24.2 Rekand T, Hagen EM, Grønning M. Spasticity following spinal cord injury. Tidsskrift for Den norske legeforening. 2012 Apr 30.
- ↑ 25.0 25.1 25.2 Mansoor Rayegani S, Babaee M, Ahmad Raeissadat S. Rehabilitation Medicine Management of Spasticity [Internet]. Neurostimulation and Neuromodulation in Contemporary Therapeutic Practice. IntechOpen; 2020. Available from https://www.intechopen.com/chapters/72741 [last access 24.07.2023]
- ↑ 26.0 26.1 Jozefczyk PB. The management of focal spasticity. Clin Neuropharmacol. 2002 May-Jun;25(3):158-73.
- ↑ Bohannon RW. Tilt table standing for reducing spasticity after spinal cord injury. Arch Phys Med Rehabil. 1993 Oct;74(10):1121-2.
- ↑ Bolin I, Bodin P, Kreuter M. Sitting position - posture and performance in C5 - C6 tetraplegia. Spinal Cord. 2000 Jul;38(7):425-34.
- ↑ Taricco M, Pagliacci MC, Telaro E, Adone R. Pharmacological interventions for spasticity following spinal cord injury: results of a Cochrane systematic review. Eura Medicophys. 2006 Mar;42(1):5-15.
- ↑ Karst M, Wippermann S, Ahrens J. Role of cannabinoids in the treatment of pain and (painful) spasticity. Drugs. 2010 Dec 24;70(18):2409-38.
- ↑ 31.0 31.1 Hashemi M, Sturbois-Nachef N, Keenan MA, Winston P. Surgical Approaches to Upper Limb Spasticity in Adult Patients: A Literature Review. Front Rehabil Sci. 2021 Aug 31;2:709969.
- ↑ 32.0 32.1 Eltorai I, Montroy R. Muscle release in managing spasticity in spinal cord injury. Paraplegia. 1990 Sep;28(7):433-40.