Von Willebrand Disease
Introduction[edit | edit source]
Von Willebrand Disease (VWD) is considered to be the most common bleeding disorder in humans and in some animals such as dogs[1]. VWD is named after the Finnish physician who described the disease in the 1920s. 1 in 100 to 10 000 individuals is estimated to have VWD. Patients with mild VWD symptoms are rarely diagnosed resulting in the gap “100 to 10 000“[2]. It is estimated that it affects 1% of all US population[3].
History[edit | edit source]
In 1926 the Finnish physician Erik Von Willebrand described a new bleeding disorder which he called “hereditary pseudo haemophilia.” He recognized that this new disease is different from haemophilia, but he couldn’t identify the responsible plasma factor. Later by many years; this factor was identified and named after him too and now it’s known as Von Willebrand Factor (VWF)[4].
Von Willebrand Factor[edit | edit source]
Von Willebrand Factor (VWF) is a multifunctional multimeric glycoprotein[5] which is synthesized in endothelial cells[6]. The VWF consists of similar subunits that contain binding sites for glycoprotein receptors. The adhesive activity of VWF depends mainly on the size of its multimers[7]. Von Willebrand Factor plays an important role in primary and secondary hemostasis, acts as a mediator for adhesion and a carrier for coagulation FVIII.[6] It is also involved in angiogenesis and inflammatory processes.[8][9]
Definition[edit | edit source]
Von Willebrand Disease can either be inherited or acquired.
- As an autosomal inherited disease it results from a defect in the protein factor called VWF. Type 1 and 2 are autosomal dominant and type 3 is transmitted as autosomal recessive. It affects men and women equally as it is an autosomal linked disease[4][10][11].
- Acquired VWD is different from inherited VWD as the disease develops later in life and is not a result of genetic inheritance. Acquired VWD may be due to autoimmune reactions in people with cardiac defects; certain forms of cancers; diabetes mellitus; autoimmune disease or after usage of certain drugs such as valpronic acid.[12]
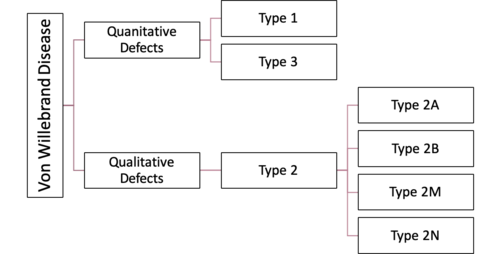
Classification of Inherited Von Willebrand Disease[edit | edit source]
There are several classifications of VWD (Figure below). Inherited VWD is further categorized into types 1, 2 and 3 and acquired VWD. The international society of thrombosis and homeostasis further classifies VWD according to qualitative and quantitative VWF defects[13].
Type 1 and 2 are both autosomal dominant (image): with this pattern of inheritance, the affected individual has 1 copy of a mutant gene and 1 normal gene on a pair of autosomal chromosomes. This means that people with autosomal dominant diseases have a 50/50 chance of passing the mutant gene and the disorder to each of their children. [14]
Type 1 Von Willebrand Disease[edit | edit source]
Type 1 VWD is the most common. About 75% of people with VWD are type 1 [15][16]. It is considered to be the mildest type and it has a partial quantitative VWF defect[15]. Due to its mild presentation, it is the hardest to diagnose. Type 1 VWD is autosomal dominant. The most common manifestation of Type 1 VWD is tooth bleeding and bleeding post-operative or post-injury. [15][17]
Type 2 Von Willebrand Disease[edit | edit source]
20-25% of people with VWD have type 2, making it the second most common[16]. It is known for its qualitative VWF defect. With Type 2 there is enough VWF factor in the blood. But, the VWF factor does not work properly due to a mutation in the VWF multimer[6][18]. It is subdivided into four subtypes which are Type 2A, Type 2B, Type 2M and Type 2N[5].
- Type 2A
Type 2A is the most common subtype in Type 2. The amount of VWF is normal but due to a mutational defect in VWF protein, the platelets can’t bind to each other properly. This causes a problem in the coagulation process[4]. Type 2A manifests by postoperative haemorrhage, menorrhagia and post-dental extraction haemorrhage.[5]
- Type 2B
Type 2B is the second most common subtype in Type 2. It has a different mutation in the VWF protein from Type 2A. This defect leads to the binding of the VWF to the platelets in the bloodstream instead of binding at the injured site. The body then removes the abnormally bound platelets causing a decrease in the platelets amount[16]. Type 2B manifests by bruising easily, prolonged bleeding from minor wounds and nasal bleeding (epistaxis)[6].
- Type 2M
M stands for Multimer. Type 2M is characterized by a mutation in the multimer of the VWF protein leading to decreased activity of VWF and its failure to bind with the platelets. Type 2 M manifests by prolonged bleeding from minor wounds similar to Type 2B[12].
- Type 2N[19]
“N “ refers to Normandy or in French Normaundie, a town in France where this subtype was first identified.[16] It is characterized by a failure of Factor VIII transporting on VMF despite normal platelet binding with VWF. This results in low factor VIII levels. Type 2N is commonly mistaken for haemophilia A due to the low factor VIII levels.[16]
Type 3 Von Willebrand Disease[edit | edit source]
The most severe and rarest type of VMD is type 3 VWD. It is characterized by the complete absence of VWF in the blood plasma and blood platelets. In contrast to type 1 and type 2, type 3 is autosomal recessive. Type 3 VWD manifests as severe bleeding in soft tissues, joints, muscle, nose and gut[15].
Clinical Manifestations[edit | edit source]
The clinical manifestation of VWD is different according to the type of VWD[15].
The most common manifestations include:[20][21]
- Nasal haemorrhage
- Dental and oral cavity haemorrhage
- Prolonged wound healing
- Menorrhagia which is excessive menstrual haemorrhage
- Gastrointestinal haemorrhage (with severe type)
Some common manifestations in the pediatric population include :[22]
- Umbilical stump haemorrhage
- Cephalic haematoma
- Cheek haematoma
- Conjunctival haemorrhage
- After circumcision haemorrhage
- After venipuncture haemorrhage
Diagnostic Tools[edit | edit source]
Von Willebrand disease is the most common bleeding disorder, but it is also the hardest to diagnose.[23] Unfortunately, the usual blood clotting screening laboratory tests such as CBC, Activated Partial Thromboplastin Time (APTT) Test, Prothrombin Time (PT) Test and Fibrinogen test are all normal in patients with VWD especially those with mild and moderate types.[24]
To diagnose VWD there are screening and diagnostic laboratory tests that can be used.[25]
- VWD Screening Tests
- VWF antigen (VWF:Ag)
VWF antigen is a quantitative reliable assessment tool of the plasma VWF protein level24. This method is efficient in detecting VWD quantitative defect types. The normal range of VWF: Ag is 50 to 200IU/dl. Anything lower than 50 may indicate the presence of VWD.[26]
- VWF ristocetin cofactor activity (VWF:RCo)
The VWF: RCo is the most commonly used test to assess the ability of binding ability of VMF24. The normal ranges of VWF:RCo are between 50 and 200 IU/dL.[26]
- Factor VIII activity (FVIII: C)
The measurement of FVIII: C is included in the screening laboratory tests of VWD. VWF is a carrier protein for FVIII. Normal ranges of FVIII:C/VWF: Ag ratio is approximately 1. In type 2N, this ratio is low, and in type 3 VWD, the FVIII:C is less than10 IU/dL[26].
- VWF:RCo/VWF:Ag Ratio
The VWF: RCo/VWF: Ag ratio is used to diagnose the type of VWD. In Type 1 VWD the levels of both VWF: RCo and VWF: Ag decreases and as such the ratio between them remains around one. While in type 2 VWD the VWF: RCo decreases compared to the VWD: Ag level so the VWF: RCo/VWF: Ag is approximately 0.6.[26]
- VWD Confirmatory Tests
Once VWD is diagnosed some confirmatory tests are run to indicate the type of VWD such as[26]:
- VWF multimer distribution which is found to be abnormal in type 2A and type 2B
- VWF: CB is abnormal in type 2A and types 2B, some type 2M.
- VWF: PB increases in type 2
- LD-RIPA increases in type 2B.
- VWF: FVIIIB decreases in type 2
- VWFpp/VWF: Ag ratio increases in type 1
- VWF gene sequencing is most helpful in differentiating type 2 variants
Medical Management[edit | edit source]
Treatment of VWD is based on the severity of symptoms and the amount of haemorrhage. As most patients with VWD are type 1 (mild type); they don’t need regular treatment. The goals of treatment are to increase the circulating VWF activity and reduce haemorrhage.[27]
Some medications are used in treating and decreasing the symptoms of VWD such as:
- Desmopressin
Desmopressin can be administered by nasal spray, intravenous or subcutaneous injection. It that can be administered easily at home and is used for type 1 VWD. The recommended dosage is 0.3 mcg/kg intravenous or subcutaneous or 2 sprays intranasally (for patients above 50 kg) or 1 spray intranasal (for patients less than 50 kg).[28]
- Plasma-derived VWF and FVIII Concentrates
Plasma-derived VWF and FVIII concentrate such as Humate P (VWF:RCo: FVIII ratio= 2.4:1), Wilate (VWF:RCo: FVIII ratio = 1:1) and Alphanate Plasma-derived VWF and FVIII concentrate (VWF:RCo: FVIII ratio 1:3) are intravenous medications that are used as an acute treatment in severe types of VWD. They can also act as prophylaxis against haemorrhage. The recommended dosage is 50-60 ristocetin cofactor activity units/kg for major surgery, depending on baseline VWF level and desired goal level.[28]
- Antifibrinolytics
Antifibrinolytics such as Aminocaproic acid and Tranexamic acid inhibits fibrinolysis. They are used as active and prophylactic treatments, especially for mucosal surfaces. They are introduced orally or intravenously. The recommended dosage for Aminocaproic acid is 100 mg/kg then 50 mg/kg every 6 hours. The dosage for Tranexamic acid is 1500 mg 3 times daily for 5 days for menorrhagia cases.[27][28]
- Hormonal therapy
Hormonal therapy is also an option in treating menorrhagia.[29]
Physical Therapy Management[edit | edit source]
Physical therapy has an important role in promoting functional skills in paediatrics and adults with VWD; that is why the National Haemophilia Foundation formed a physical therapy working group to create the best physical therapy practice for bleeding disorders such as VWD.[30]
The Medical and Scientific Advisory Council (MASAC) developed guidelines and a framework for physical therapy management in bleeding disorders. MASAC stated that physical therapy is crucial in joint and muscles rehabilitation post soft tissue injuries and hemarthroses These clinical presentations mostly occur with the more severe types of VWD.
Physical Therapy Evaluation[edit | edit source]
According to the MASAC; the physical therapy evaluation is an important element of VWD management. The evaluation aims to detect musculoskeletal and other limitations caused by the bleeding disorder that affect functional activities and daily life activities(ADL).[31]
History and Interview[31][edit | edit source]
It includes interviewing the patient or the caregiver and taking the notes about:
- Personal history
- Family history
- Bleeding history
- Medical and surgical history
- Pain history
- ADL concerns
- Occupational concerns
Joints that have recurrent bleeding disorders are known as “Target Joints”. The most common target joints are knee, elbow, ankle, hip and shoulders.[30]
Subjective Assessment[edit | edit source]
- Palpation of joints at rest and during active range of motion to detect crepitus, synovitis, oedema or temperature.
- Girth measurement (measuring circumference with a tape measure) to assess oedema/ muscle atrophy.
- Atypical Joint End feel detection via a passive range of motion.
- Manual Muscle Testing to assess muscular strength.
- Muscle Flexibility test.
- Sensation and proprioception.
Objective Assessment[edit | edit source]
- Balance and fall assessment
- Posture and alignment assessment
- Assessment of functional activities
- Gait analysis
- Neuromotor assessment
- Musculoskeletal Ultrasound
Physical Therapy Treatment[edit | edit source]
There is a recommended physical therapy program by the MASAC for muscles and joints bleeding in different recovery phases (Acute, Subacute and Chronic). All muscles follow the same guidelines, except for iliopsoas muscle, and all physical therapy suggested protocols are performed post factor replacement medication as follows :[32]
| Acute Phase | Subacute Phase | Chronic | Other Treatment Considerations | Precautions | |
|---|---|---|---|---|---|
| Muscles Bleeding except iliopsoas | Main Problem:
Present pain at rest and with movement |
Main Problem :
Limitations in ADL without increasing pain from baseline |
Main Problem :
ADL limitation but without pain |
Electric stimulation
- Work closely with a haematologist - Teach patient to avoid overstretching - Education on activity modification - Kinesiotape® - Treatment duration will vary based on individual needs - Ultrasound for blood absorption can be used with precautions. |
Monitor for neurovascular compromise while Splinting
- Caution with the use of compression on the affected muscle. - Use of heat modalities including ultrasound with precautions |
| Physical Therapy Program:
- No compression in case of neuromuscular symptoms. - No active movement or weight-bearing till bleeding stoppage. - Splinting. - RICE: Rest, Ice, Compression & Elevation - TENS |
Physical Therapy Program:
- Splinting and assistive device to limit activity - Toe touch Weight-bearing - Isometric contractions - Active range of motion - Positioning - AAROM without pain - TENS |
Physical Therapy Program:
- Full Weight Bearing without assistive devices - Positional stretching - Active Range of Motion exercises - unrestricted lifting - Re-evaluation. | |||
| Iliopsoas Muscle Bleeding | Main Problem:
pain presented at rest and with movement |
Main Problem :
ADL limitation without increase pain from baseline |
Main Problem :
ADL limitation but without pain |
Electrical-stimulation to prevent femoral nerve palsy/atrophy.
- Hip flexor stretching with caution. - Teach patient to avoid overstretching - Treatment duration will vary based on individual |
- Monitor for femoral nerve palsy |
| Physical Therapy Program :
- Bedrest - Toe Touch Weight Bearing for household mobility - Rest - Opposite limb ROM Ankle pumps involved lower limb with pain avoidance |
Physical Therapy Program:
- Toes touch weight-bearing without an increase in pain - Isometric contractions - Active Range Of Motion exercises to involve Lower limb without an increase in pain - Positioning with increasing hip extension Range Of Motion in supine and prone over pillows without an increase in pain. - therapeutic exercises for non-involved limb |
Physical Therapy Program:
- Full Weight Bearing without assistive devices - positional stretches from a prone position - Active range of motion exercises - Re-evaluation | |||
| Joint Bleeding | Main Problem:
pain presented at rest and with movement |
Main Problem :
ADL limitation without increase pain from baseline |
Main Problem :
ADL limitation but without pain |
- TENS
- Relaxation Techniques - Kinesiology tape - Cryotherapy - Elastic stockinette - Myofascial release - Work with the haematologist. - Further MRI to determine the presence of chronic synovitis) - Treatment duration will vary based on individual needs. |
- Avoid aggressive exercise too early
- Monitor for nerve compression - Use of heat modalities including ultrasound with precautions - No return to sports or activity until pain-free full ROM and strength |
| Physical Therapy Program:
- RICE (ice for 10-20 minutes every 1-2 hours) - Splinting - Non-Weight Bearing using an assistive device |
Physical Therapy Program :
- Continue RICE for pain and after exertion Splinting - Night resting splint for protection - Begin progressive weight-bearing - Activity modification to avoid pain - Pain-free gentle active Range of motion - Pain-free progressive strengthening |
Physical Therapy Program :
- Dynamic splinting to increase ROM - Active range of motion Progressive - Strengthening as Open chain, closed chain and resistive band - proprioceptive training - Gentle joint mobilizations - Modified functional activities - Orthotics - Reevaluations |
Additional Resources[edit | edit source]
References[edit | edit source]
- ↑ Brooks MB, Catalfamo JL. von Willebrand disease. Schalm's veterinary hematology. 2022 Apr 22:731-8.
- ↑ Genetic Home of Reference. Von Willebrand Disease. U.S. National Library of Medicine. Published 2020. Accessed June 15, 2020.
- ↑ Von Willebrand Disease. National Hemophilia Foundation. Published 2020. Accessed June 15, 2020.
- ↑ 4.0 4.1 4.2 Leebeek FWG, Eikenboom JCJ. Von Willebrand’s disease. N Engl J Med. 2016;375(21):2067-2080.
- ↑ 5.0 5.1 5.2 Rassoulzadegan M, Ala F, Jazebi M, et al. Molecular and clinical profile of type 2 von Willebrand disease in Iran: a thirteen-year experience. Int J Hematol. 2020;111(4):535-543.
- ↑ 6.0 6.1 6.2 6.3 Randi AM, Laffan MA. Von Willebrand factor and angiogenesis: basic and applied issues. J Thromb Haemost. 2017;15(1):13-20.
- ↑ Stockschlaeder M, Schneppenheim R, Budde U. Update on von Willebrand factor multimers: focus on high-molecular-weight multimers and their role in hemostasis. Blood Coagul Fibrinolysis. 2014;25(3):206-216.
- ↑ Lenting PJ, Casari C, Christophe OD, Denis C V. von Willebrand factor: the old, the new and the unknown. J Thromb Haemost. 2012;10(12):2428-2437.
- ↑ Kalot MA, Husainat N, El Alayli A, Abughanimeh O, Diab O, Tayiem S, Madoukh B, Dimassi AB, Qureini A, Ameer B, Eikenboom JC. von Willebrand factor levels in the diagnosis of von Willebrand disease: a systematic review and meta-analysis. Blood advances. 2022 Jan 11;6(1):62-71.
- ↑ Sharma R, Haberichter SL. New advances in the diagnosis of von Willebrand disease. Hematology. 2019;2019(1):596-600.
- ↑ Echahdi H, El Hasbaoui B, El Khorassani M, Agadr A, Khattab M. Von Willebrand’s disease: case report and review of the literature. Pan Afr Med J. 2017;27:147.
- ↑ 12.0 12.1 Nazzaro A-M, Philipp CS, Johnson RW, James AH. Von Willebrand Disease. National Organization of Rare Diseases... Published 2020. Accessed June 15, 2020.
- ↑ Bharati KP, Prashanth UR. Von Willebrand disease: an overview. Indian J Pharm Sci. 2011;73(1):7-16.
- ↑ Schiel, WC. Medical Definition of Autosomal Dominant. MedicineNet.
- ↑ 15.0 15.1 15.2 15.3 15.4 Von Willebrand Disease. NHS. https://www.nhs.uk/conditions/von-willebrand-disease/. Published 2017. Accessed June 16, 2020.
- ↑ 16.0 16.1 16.2 16.3 16.4 Types of von Willebrand disease. Canadian Hemophilia Society. https://www.hemophilia.ca/types-of-von-willebrand-disease/. Published 2018. Accessed June 16, 2020.
- ↑ Vanduine S, Ridley K, Bashutski J, Snyder M, Powell C, Taichman S. Gingival bleeding and oral hygiene in women with von Willebrand Disease (VWD): a pilot study. J Haemoph Pract. 2018;4(1):49-57.
- ↑ Hemophilia of Georgia. Types of von Willebrand Disease. The Hemophilia, von Willebrand Disease & Platelet Disorders Handbook. Published 2007. Accessed June 16, 2020.
- ↑ Seidizadeh O, Peyvandi F, Mannucci PM. Von Willebrand disease type 2N: an update. Journal of Thrombosis and Haemostasis. 2021 Apr;19(4):909-16.
- ↑ Pollak ES. Von Willebrand Disease Clinical Presentation. Medscape. https://emedicine.medscape.com/article/206996-clinical. Published 2019. Accessed June 16, 2020
- ↑ Ichinose A, Osaki T, Souri M, Favaloro EJ. A review of autoimmune acquired von Willebrand factor deficiency in Japan. InSeminars in Thrombosis and Hemostasis 2022 Jul 8. Thieme Medical Publishers, Inc..
- ↑ Sanders Y V, Fijnvandraat K, Boender J, et al. Bleeding spectrum in children with moderate or severe von Willebrand disease: Relevance of pediatric-specific bleeding. Am J Hematol. 2015;90(12):1142-1148.
- ↑ Castaman G, Linari S. Diagnosis and Treatment of von Willebrand Disease and Rare Bleeding Disorders. J Clin Med. 2017;6(4):45.
- ↑ National Center on Birth Defects and Developmental Disabilities. von Willebrand Disease (VWD) Diagnosis. Centres for Disease Control and Prevention. https://www.cdc.gov/ncbddd/vwd/diagnosis.html. Published 2019. Accessed June 17, 2020.
- ↑ Roberts JC, Flood VH. Laboratory diagnosis of von Willebrand disease. Int J Lab Hematol. 2015;37(1):139-148.
- ↑ 26.0 26.1 26.2 26.3 26.4 Ng C, Motto DG, Paola J Di. Diagnostic approach to von Willebrand disease. Blood. 2015;125(13):2029-2037. doi:10.1182/blood-2014-08-528398
- ↑ 27.0 27.1 Chapin J. Von Willebrand disease in the elderly: clinical perspectives. Clin Interv Aging. 2018;13:1531-1541.
- ↑ 28.0 28.1 28.2 Sharma R, Flood VH. Advances in the diagnosis and treatment of Von Willebrand disease. Haematology. 2017 Dec 8;2017(1):379-84.
- ↑ Green D. Diagnosis and Treatment of von Willebrand Disease. NEJM Jornal Watch. https://www.jwatch.org/na45666/2017/12/06/diagnosis-and-treatment-von-willebrand-disease. Published 2017. Accessed June 17, 2020.
- ↑ 30.0 30.1 Lowes LP, Alfano L, Orlin MN. Musculoskeletal System: Considerations and Interventions for Specific Pediatric Pathology. In: Effgen SK, ed. Meeting the Physical Therapy Needs of Children. 2nd ed. Philadelphia: F. A. Davis Company; 2013:226-227.
- ↑ 31.0 31.1 Medical and Scientific Advisory Council. Physical Therapy Evaluation Recommendations. Natl Hemoph Found. 2018.
- ↑ National Hemophilia Foundation. Physical Therapy Practice Guidelines for Persons with Bleeding Disorders: Muscle Bleed. 2018;