Sarcomere
Original Editor - Lucinda hampton
Top Contributors - Lucinda hampton and Vidya Acharya
Introduction[edit | edit source]
A sarcomere is the basic contractile unit of a myocyte (muscle fibre). A sarcomere is composed of two main protein filaments (thin actin and thick myosin filaments) which are the active structures responsible for muscular contraction. The widely accepted theory describing muscular contraction is called the sliding filament theory, which proposes that the active force is generated as actin filaments slide past the myosin filaments, resulting in contraction of an individual sarcomere[1].
Sliding Filament Theory[edit | edit source]
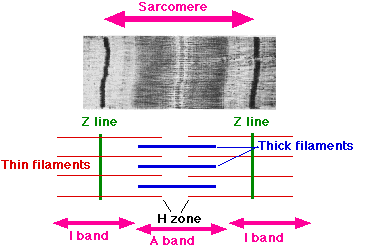
The sliding theory, proposed by scientists, who through the use of high-resolution microscopes visualised the actin and myosin filaments within a sarcomere. They could see length of the sarcomere when relaxed and its shortening as it contracted, and were able to give names to particular zones.
- A bands (or anisotropic bands): It is also called the dark band and contains the whole thick filament (myosin).
- I bands (or isotropic bands): it is called the light band that contains only the thin filament (actin). The thin filament lies between the two thick filaments.
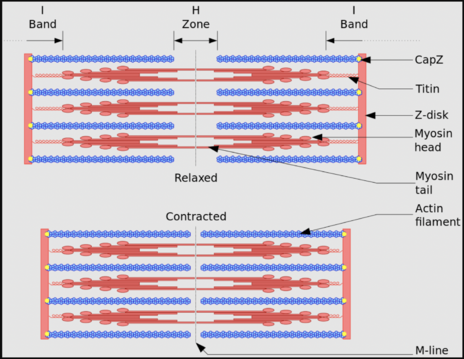
- Z disc: it is the area where two actin filaments connect and transverse the I bands. Similarly, sarcomere can also be described as the structure between the two z discs.
- M line: M line contains the protein called myomesin and it marks the centre of the sarcomere.
- H zone: it is the area between the M line and Z disc and this zone contains only the myosin[2].
This video explains the structure of a Sarcomere-sarcomere bands and actin and myosin arrangement.
Sliding Filaments[edit | edit source]
In essence, during contraction a sarcomere shortens like a collapsing telescope, as the actin filaments at each end of a central myosin filament slide toward the myosin's centre, as described below.
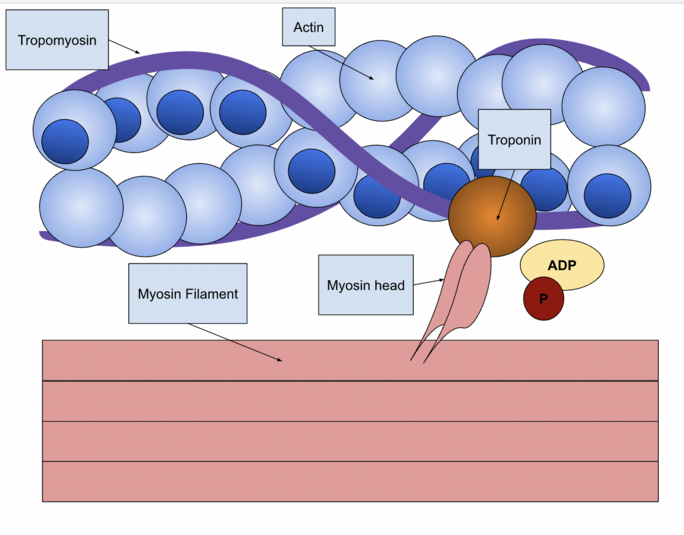
The movements of myosin appears like a molecular dance, with the myosin reaching forward to binds to the actin, contracting, then releasing actin, before it reaches forward again to bind actin in a new cycle. This is the so called myosin-actin cycling. As the myosin S1 segment binds and releases actin, it forms what are called cross bridges, which extend from the thick myosin filaments to the thin actin filaments. The contraction of myosin's S1 region is called the power stroke, which requires the hydrolysis of ATP, which breaks a high-energy phosphate bond to release energy, resulting in force generation and shortening of an individual sarcomere. Because sarcomeres are joined end to end throughout an entire muscle fiber, their simultaneous contraction shortens the entire muscle[4].[1]
The mitochondria that are present in large numbers in skeletal muscle produce the much needed ATP.
In sarcomeres, the contractile actin and myosin filaments are integrated in paracrystalline order by the action of accessory cytoskeleton proteins, forming what is often termed the sarcomeric cytoskeleton[5].
Myosin and Actin[edit | edit source]
Myosin is one of three major classes of molecular motor proteins: myosin, dynein, and kinesin.
Actin is the most abundant protein in most eukaryotic cells and has a pivotal role in muscle contraction as well as in cell movements. It is the essential building block of the microfilament system.[6]
References[edit | edit source]
- ↑ 1.0 1.1 Mansfield PJ, Neumann DA. Essentials of kinesiology for the physical therapist assistant e-book. Elsevier Health Sciences; 2018 Oct 23.Available:https://www.sciencedirect.com/book/9780323544986/essentials-of-kinesiology-for-the-physical-therapist-assistant (accessed 10.7.2022)
- ↑ The biology notes Sarcomere Available:https://thebiologynotes.com/sarcomere/ (accessed 10.7.2022)
- ↑ Larry Keeley Sarcomere Structure. Available from https://www.youtube.com/watch?v=P1zD_MpTo0MAccessed 20/7/22
- ↑ Krans JL. The sliding filament theory of muscle contraction. Nature Education. 2010;3(9):66. Available:https://www.nature.com/scitable/topicpage/the-sliding-filament-theory-of-muscle-contraction-14567666/ (accessed 11.7.2022)
- ↑ Gautel M, Djinović-Carugo K. The sarcomeric cytoskeleton: from molecules to motion. Journal of Experimental Biology. 2016 Jan;219(2):135-45. Available:https://journals.biologists.com/jeb/article/219/2/135/33486/The-sarcomeric-cytoskeleton-from-molecules-to (accessed 11.7.2022)
- ↑ Roger Bouillon, in Vitamin D (Third Edition), 2011 Available:https://www.sciencedirect.com/book/9780123819789/vitamin-d (accessed 11.7.22)