Proximal Humerus Fractures
Definition/Description[edit | edit source]
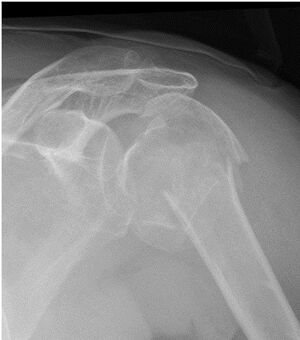
Fracture of the proximal humerus is a common injury often seen in older patients and one of the true osteoporotic fractures.[1][2] Proximal humerus fracture (PHF) includes all fractures to the anatomical neck, surgical neck, greater tuberosity (GT), or lesser tuberosity, either isolated or in combination.[3]
Clinically Relevant Anatomy[edit | edit source]
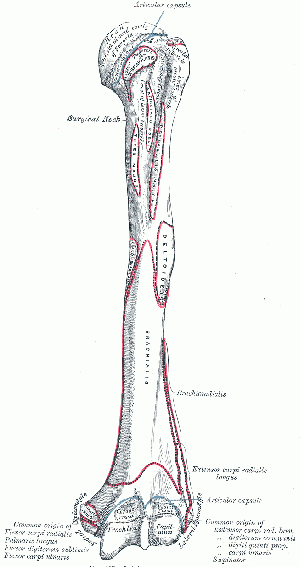
Osteology: Humerus
Classification: long bone
Features:
- Humeral head: The proximal articular surface of the upper extremity. It articulates with the glenoid fossa of the scapula.
- Greater tuberosity: Located lateral to the head at the proximal end.
- Lesser tuberosity: Located inferior to the head, on the anterior part of the humerus.
- Anatomic neck: Located between the head and the tuberosities.
- Surgical neck: Located between the tuberosities and the shaft.
- Intertubercular groove: a narrow groove between the greater tuberosity and the lesser tuberosity.
- Humeral shaft.
Function: serves as an attachment to 13 muscles which contribute to the movement of the hand and elbow, and therefore the function of the upper limb.
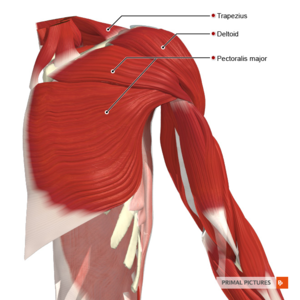
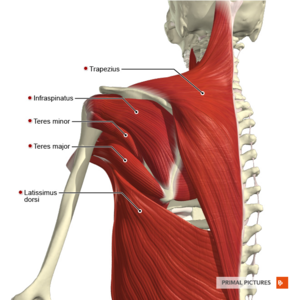
Muscles:[4] The intrinsic muscles of the shoulder which connect the scapula and/or clavicle to the humerus include:
- Deltoid
- Teres major
- Supraspinatus
- Infraspinatus
- Teres minor
- Subscapularis
- Latissimus dorsi
- Pectoralis major
- Coracobrachialis
- Biceps brachii
- Triceps brachii
Mechanism of Injury / Pathological Process[edit | edit source]
Epidemiology[edit | edit source]
PHFs are the third most common fracture in the elderly, after proximal femur and distal radius fractures, and represent 10% of all fractures.[3] PHFs account for approximately 5.7% of fractures in adults[5], and increase in incidence with ageing and are one of the most common fractures among people aged over 65 years old.[6][7] The mean age was 70 years old (16-97).[8]
In the US, about 370,000 ED visits due to humerus fractures, and PHFs were the most common, accounting for 50% of humerus fractures in 2008.[9] A study showed that overall age-standardised incidence rates for PHFs in men and women were 40.6 (95% CI 32.7, 48.5) and 73.2 (95% CI 62.2, 84.1) per 100,000 person-years, respectively, in South-Eastern Australia.[10]
The risk of PHF increases with age and is most prevalent in osteoporotic elderly women. Apart from bone fragility, caused by osteopenia or osteoporosis, commonly reported risk factors include those related to increased risk of falls, such as low level of physical activity, impaired balance, or lower limb pain or injury.[3]
Aetiology[edit | edit source]
The 2 most common mechanisms of PHF:
- High-level energy trauma (e.g., sports injuries and motor vehicle accidents)
- Low-energy trauma (e.g., a fall from a standing position or a direct blow to the shoulder)
Associated conditions[edit | edit source]
•Nerve injury: Common injuries are direct injuries to the brachial plexus or traction injuries to the axillary nerve. More likely in the presence of a fracture-dislocation.[11]
•Vascular injury: Rare, but more likely in the presence of a fracture-dislocation. Higher likelihood in the elderly. Signs of distal ischaemia may be absent, but a large expanding haematoma, pulsatile external bleeding, unexplained hypotension, delayed anaemia and associated nerve trunk or plexus injury should increase the level of suspicion.[11]
Clinical Presentation[edit | edit source]
Symptoms[edit | edit source]
- Pain and swelling
- Decreased motion
Classification[edit | edit source]
There are 2 main classifications used for PHF (Neer and AO/OTA classifications) and one specific to GT fractures.
The Neer Classification[edit | edit source]
The four-segment classification system describes PHFs by the number of displaced segments or parts, and additional categories for articular fracture and dislocation. The GT, lesser tuberosity, articular surface, and humeral diaphysis are the potential segments involved. It is defined that the segment is displaced when there is greater than 1 cm separation or 45° angulation.[12]
•One-Part fractures: No fragment satisfies the displacement criteria. Fractures without fragments considered displaced are defined as partial fractures, regardless of the actual number of fracture lines or their locations.
•Two-Part fractures: One segment is displaced, which is the GT, lesser tuberosity, or articular segment at the level of the anatomical neck or surgical neck.
•Three-Part fractures: One tuberosity is displaced, and surgical neck fracture is displaced. The remaining tuberosity is attached, and rotational deformation occurs.
•Four-Part fractures: All four segments (both tuberosities, articular surfaces, and shaft) satisfy the criteria for displacement. The articular segment is normally laterally displaced and out of contact with the glenoid. This is a serious injury and a high risk of avascular necrosis.
•Valgus-Impacted Four-Part Fractures: This pattern was added as a separate category in 2002. The head is rotated into a valgus position and pushed down between the tuberosities to accommodate the head. The articular surface remains in contact with the glenoid and is not laterally displaced.
•Fracture Dislocations and Articular Surface Injuries: Separate categories were added for dislocations, as they represent more severe injuries and are more likely to develop avascular necrosis and heterotopic ossification. Likewise, articular surface fractures fell into a separate category due to their unique management considerations. There are two types, which are head-splitting fractures and impaction fractures.
The AO/OTA Classification[edit | edit source]
The AO/OTA 2007 classification is based on the severity and articular/extraarticular and unifocal/bifocal pattern of the fracture, defining three main types (A, B, and C).[14]
•Type A: extraarticular and unifocal
•Type B: extraarticular and bifocal
•Type C: articular
Diagnostic Procedures[edit | edit source]
Radiological assessment[15][edit | edit source]
•Plain x-rays: Plain radiographic imaging is the primary baseline investigation for diagnosis, classification, and management planning of PHFs. X-rays should include:
- A true anteroposterior view
- A Trans-scapular Y view
- An axillary lateral view
•Doppler ultrasound: May be used to assess the associated vascular injury and concomitant rotator cuff tears.
•Computerised tomography (CT): May be used to assess complex fracture patterns, while it also allows quantification of available bone stock and evaluation of the extent and location of fracture union.
•CT angiography: Can be used for accurate diagnosis and guiding management of co-existing arterial injury.
•Magnetic resonance arthrography: Additional imaging tool for the assessment of periarticular soft tissue.
•Angiography: Additional imaging tool for the assessment of vascular injuries.
Differential Diagnosis[edit | edit source]
Evaluation should begin with an examination of the neurological and vascular status of the affected extremity. Evaluate the entire limb for potential fractures. Concurrent fractures are especially seen in high-energy injury mechanisms. Injuries most commonly associated with PHFs include axillary, suprascapular, or brachial plexus nerve injuries, axillary artery injuries, pneumothorax, haemothorax, and rotator cuff injuries.
Outcome Measures[edit | edit source]
•Disabilities of the Arm, Shoulder, and Hand score[16]
•American Shoulder and Elbow Surgeons score[16]
Management / Interventions[edit | edit source]
PHF can be treated conservatively with the anticipation that the fracture will heal, and the patient will regain function in that shoulder.[2] The treatment is usually a sling and early range-of-motion exercises, however, sometimes can be open reduction with internal fixation (ORIF) or prosthetic joint replacement. Choosing conservative treatment or surgical fracture management depends on the type and severity of the fracture, concomitant soft-tissue injuries, and other individual factors such as age, comorbidities, activity level, quality of bone, and motivation of the patient.[3]
Conservative medical management[edit | edit source]
The vast majority of nondisplaced PHFs are successfully treated conservatively and some systematic reviews and meta-analysis have reported that non-operative treatment resulted in less complications as well as that surgical intervention did not show significant improvement of functional and clinical outcomes.[17][18][19]
Patients receiving a conservative treatment are generally immobilised 4 to 6 weeks in a simple sling, with the glenohumeral joint in the resting position, in internal rotation, and 0° of adduction.[3]
Rehabilitation after initial conservative treatment[edit | edit source]
Aim: Restoring range of motion and normal shoulder strength and ultimately function[3]
< Within a week after PHF >
- Early passive mobilisation of the shoulder in forward elevation, abduction, and external rotation with respect to patients’ tolerance (External rotation should be limited to 30°-40° for the first 3 weeks to restrict rotational stress to the fracture)
- Education to prevent the complications caused by the immobilisation)
- Pain control
- Active mobilisation of the neck, elbow, wrist, and hand
< 3 to 5 weeks >
- Active-assisted mobilisation
< 4 weeks >
- Submaximal isometric exercises in flexion, abduction, and internal rotation
< 4 to 6 weeks >
- Active mobilisations (when the sling is no longer needed and when adequate fracture consolidation is achieved; start in the supine position and gradually progress to the sitting position)
< 8 to 12 weeks >
- Strengthening exercises of the rotator cuff and scapulathoracic muscles
< 12 weeks >
- Functional exercises
- Return to leisure or sport activities (depends on the patients’ tolerance and personal demands)
< 8 to 16 weeks >
- Return to work (depends on the patients’ physical work demands)
Surgical fracture management[edit | edit source]
Recent evidence has indicated that surgery has no significant advantage over conservative treatment, even for complex fractures, in terms of functional outcomes and health-related quality of life in elderly patients.[18] However, surgical treatments are recommended for active patients with displaced PHF on initial x-rays or CT imaging or following secondary displacement in the first 3 weeks of follow-up.[3]
List of common surgical complications[edit | edit source]
Most common surgical complications in surgical management of PHFs [3]
| Open reduction internal fixation | Shoulder replacement |
| Avascular necrosis | Joint instability/dislocation |
| Screw cut out | Implant malpositioning |
| Nonunion | Implant loosening |
| Malunion | Infection (Propionibacterium acnes) |
| Persistent joint dislocation | Biceps pathology |
| Infection (Propionibacterium acnes) | Axllary nerve palsy |
| Capsulitis | Brachial plexus palsy |
| Rotator cuff tear | Hemiarthroplasty: rotator cuff failure |
| Biceps pathology | Reverse arthroplasty: glenoid notching |
| Axillary nerve palsy | Periprosthetic fracture |
| Brachial plexus palsy | Heterotopic ossification |
| Heterotopic ossification |
Types of surgeries[edit | edit source]
- Closed reduction and percutaneous stabilisation using pins or wires
- External fixation
- Open reduction and plating: for example, buttress plates, angle blade plates and proximal humeral locking plates
- Open reduction and fixation using a tension-band principle
- Intramedullary nailing either antegrade or retrograde insertion; nowadays, intramedullary nails are 'locked' into place, generally using screws
- Hemiarthroplasty (replacement of the humeral head)
- Total shoulder replacement (replacement of the entire joint; thus both the 'ball' (humeral head) and 'socket' (glenoid)). This includes reverse polarity arthroplasty, where the joint polarity is reversed such that the ball is on glenoid side and the socket on the humeral side.[19]
Rehabilitation after surgical treatment[edit | edit source]
Rehabilitation protocols are often variable because restrictions will depend on the concomitant injuries and surgical protocols, such as associate shoulder dislocation or concomitant rotator cuff repair. [3]
After open reduction internal fixation (ORIF)[edit | edit source]
This protocol[1][20] is criteria-based as well as time-based (depending on tissue healing). Specific interventions should be based on individual needs and therapists should consider laboratory results and clinical decision-making. The expected outcome timeframes included in this protocol may vary depending on surgeon preference, additional procedures performed, and/or complications.
< Phase I: Immediate post-operation (1-4 weeks after surgery) >
Goals
- Minimise pain and inflammatory response
- Protect fracture and optimise bony healing
- Restore shoulder passive range of motion (PROM)
- Maintain elbow, wrist and hand function
Precautions
- No abduction past 90°
- Shoulder external rotation 0°-40°
- No lifting greater than 1lb
- No driving until adequate ROM, sling is discharged, and no narcotic pain medication is being used
- No motions into painful ranges
Interventions
- Sling: For at least 3 weeks
- Pain/Swelling management: Cryotherapy and modalities as indicated
- Range of motion/mobility: Shoulder PROM / shoulder pendulums / elbow, wrist and hand AROM
- Strengthening: Ball squeezes / scapular retraction and mobility exercises
Criteria to progress
- Wean from sling at 4 weeks
- Adequate pain control
- Full elbow AROM
- Shoulder PROM flexion to 140°, external rotation to 40°, abduction to 90°
< Phase II: Intermediate post-operation (4-8 weeks after surgery) >
Goals
- Full shoulder PROM
- Initiate shoulder active assisted range and active range of motion (AAROM/AROM)
- Start active range of motion at 6 weeks
- Initiate gentle elbow isotonic strengthening
- Initiate shoulder isometrics
- Minimise compensatory motions of involved upper extremity
- Encourage return to normal ADLs within lifting precautions
Precautions
- No lifting greater than 2lbs before 6 weeks
- Start shoulder AROM at 6 weeks post-op
- No forceful end range over pressure to involved shoulder
- No isotonic strengthening of the shoulder
Interventions
- Continue with Phase I interventions
- AAROM: Lawn chair progression / table slides, rail slides, wall slides / pully
- AROM: Supine shoulder AROM flexion / side-lying shoulder ER with towel roll under arm / side-lying shoulder abduction to 90° / side-lying shoulder flexion / low punch
- Strengthening: shoulder isometric flexion, extension, internal rotation and external rotation / biceps curls / triceps extension / prone rows
Criteria to progress
- Full shoulder PROM
- Full elbow AROM
- Adequate pain control
- Good tolerance to shoulder isometrics and elbow strengthening
< Phase III: Late post-operation (8-12 weeks after surgery) >
Goals
- Full shoulder AROM
- Initiate shoulder strengthening
- Progress elbow and wrist strengthening
- Adequate pain control
Precautions
- No lifting greater than 10bls
- No painful or forceful stretching
- No excessive weight bearing on involved extremity
Interventions
- Continue with Phase I-II interventions
- AAROM: Standing shoulder flexion/abduction with dowel
- AROM: Standing shoulder elevation / standing shoulder PNF diagonals / Prone I, Prone Y, Prone T
- Stretching: Doorway stretch / Pec and biceps stretch / cross body stretch
- Strengthening: Rows / straight arm pull-down / resisted shoulder external and internal rotation (neutral shoulder position) / low punch with resistance / supine shoulder protraction
Criteria to progress
- Full shoulder AROM with appropriate mechanics
- No pain or compensatory strategies with strengthening exercises
< Phase IV: Advanced strengthening (12 weeks after surgery) >
Goals
- Progress shoulder strength with heavier resistance and compound movements
- Return to normal functional activities
- Continue to improve shoulder ROM in needed
Interventions
- Continue with Phase II-III interventions
- Strengthening: Rhythmic stabilisations / push up progression (wall, counter top, knees, high plank) / high plank stability progression / scaption raises / resisted shoulder diagonals / resisted shoulder external and internal rotation at 90 ° / quadruped stability progression / shoulder plyometrics / interval return to sports training if appropriate
Criteria to progress
- 80% or > strength of involved upper extremity compared to uninvolved arm with dynamometry testing
- No pain with progressive strengthening exercises
- Low level to no disability score on patient reported outcome measure
Resources[edit | edit source]
Interventions for treating proximal humerus fractures in adults
Proximal humeral fractures: current concepts in classification, treatment and outcomes
Management of proximal humerus fractures in adults
References[edit | edit source]
- ↑ 1.0 1.1 Handoll HHG, Brorson S. Interventions for treating proximal humerus fractures in adults. Cochrane Datebase of Systematic Reviews 2015, issue 11.
- ↑ 2.0 2.1 Jo MJ, Gardner MJ. Proximal humerus fractures. Curr Rev Musculoskelet Med. 2012;5(3):192-198. doi:10.1007/s12178-012-9130-2
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Lowry V, Bureau NJ, Desmeules F, Roy JS, Rouleau DM. Acute proximal humeral fractures in adults. Journal of Hand Therapy. 2017 Apr 1;30(2):158-66.
- ↑ Miniato MA, Anand P, Varacallo M. Anatomy, Shoulder and Upper Limb, Shoulder. [Updated 2021 Jul 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK536933/
- ↑ Court-Brown CM, Caesar B. Epidemiology of adult fractures: a review. Injury. 2006 Aug 1;37(8):691-7.
- ↑ Tarantino U, Capone A, Planta M, D'Arienzo M, Letizia Mauro G, Impagliazzo A, Formica A, Pallotta F, Patella V, Spinarelli A, Pazzaglia U. The incidence of hip, forearm, humeral, ankle, and vertebral fragility fractures in Italy: results from a 3-year multicenter study. Arthritis research & therapy. 2010 Dec;12(6):1-9.
- ↑ Passaretti D, Candela V, Sessa P, Gumina S. Epidemiology of proximal humeral fractures: a detailed survey of 711 patients in a metropolitan area. Journal of shoulder and elbow surgery. 2017 Dec 1;26(12):2117-24.
- ↑ Roux A, Decroocq L, El Batti S, Bonnevialle N, Moineau G, Trojani C, Boileau P, De Peretti F. Epidemiology of proximal humerus fractures managed in a trauma center. Orthopaedics & Traumatology: Surgery & Research. 2012 Oct 1;98(6):715-9.
- ↑ Kim SH, Szabo RM, Marder RA. Epidemiology of humerus fractures in the United States: nationwide emergency department sample, 2008. Arthritis Care Res (Hoboken). 2012;64(3):407-414.
- ↑ Holloway KL, Bucki-Smith G, Morse AG, et al. Humeral Fractures in South-Eastern Australia: Epidemiology and Risk Factors. Calcif Tissue Int. 2015;97(5):453-465. doi:10.1007/s00223-015-0039-9
- ↑ 11.0 11.1 Murray IR, Amin AK, White TO, Robinson CM. Proximal humeral fractures: current concepts in classification, treatment and outcomes. J Bone Joint Surg Br. 2011;93(1):1-11. doi:10.1302/0301-620X.93B1.25702
- ↑ Carofino BC, Leopold SS. Classifications in brief: the Neer classification for proximal humerus fractures, Clin Orthop Relat Res. 2013;471(1):39-43. doi:10.1007/s11999-012-2454-9
- ↑ nabil ebraheim. Proximal Humerus Fractures classification - Everything You Need To Know - Dr. Nabil Ebraheim. Available from: https://www.youtube.com/watch?v=IXCD_BcbgOw&t=5s [last accessed 6/11/2022]
- ↑ Marongiu G, Leinardi L, Congia S, Frigau L, Mola F, Capone A. Reliability and reproducibility of the new AO/OTA 2018 classification system for proximal humeral fractures: a comparison of three different classification systems. Journal of Orthopaedics and Traumatology. 2020 Dec;21(1):1-9.
- ↑ Vachtsevanos L, Hayden L, Desai AS, Dramis A. Management of proximal humerus fractures in adults. World journal of Orthopedics. 2014 Nov 11;5(5):685.
- ↑ 16.0 16.1 16.2 16.3 Richard GJ, Denard PJ, Kaar SG, Bohsali KI, Horneff JG, Carpenter S, Fedorka CJ, Mamelson K, Garrigues GE, Namdari S, Abboud JA. Outcome measures reported for the management of proximal humeral fractures: a systematic review. Journal of Shoulder and Elbow Surgery. 2020 Oct 1;29(10):2175-84.
- ↑ Song JQ, Deng XF, Wang YM, Wang XB, Xue Lİ, Bin YU. Operative vs. nonoperative treatment for comminuted proximal humeral fractures in elderly patients: a current meta-analysis. Acta Orthopaedica et Traumatologica Turcica. 2015 Jan 1;49(4):345-53.
- ↑ 18.0 18.1 Xie L, Ding F, Zhao Z, Chen Y, Xing D. Operative versus non-operative treatment in complex proximal humeral fractures: a meta-analysis of randomized controlled trials. Springerplus. 2015 Dec;4(1):1-9.
- ↑ 19.0 19.1 Handoll HH, Brorson S. Interventions for treating proximal humeral fractures in adults. Cochrane Database Syst Rev. 2015;(11):CD000434. Published 2015 Nov 11. doi:10.1002/14651858.CD000434.pub4
- ↑ Canbora MK, Kose O, Polat A, Konukoglu L, Gorgec M. Relationship between the functional outcomes and radiological results of conservatively treated displaced proximal humerus fractures in the elderly: A prospective study. Int J Shoulder Surg. 2013;7(3):105-109. doi:10.4103/0973-6042.118911
- ↑ Penn Medicine. Shoulder Surgery Rehab: Exercises for the First Six Weeks | Martin Kelley, DPT of Penn Rehab. Available from: https://www.youtube.com/watch?v=hsauKSU_Qww [last accessed 6/11/2022]
- ↑ Penn Medicine. Shoulder Surgery Rehab: Exercises for 7-12 Weeks After Surgery| Martin Kelley, DPT of Penn Rehab. Available from: https://www.youtube.com/watch?v=0F8VcqYCq-8 [last accessed 6/11/2022]
- ↑ OrthoClips. Proximal Humerus Fractures 1 (OTA lecture series III u03a). Available from: https://www.youtube.com/watch?v=eZO-fWOlFGE [last accessed 6/11/2022]
- ↑ WesternTrust. Physiotherapy Exercises following a Proximal Hummus Fracture. Available from: https://www.youtube.com/watch?v=TREhz8tagS4 [last accessed 6/11/2022]