Prion Diseases (or Transmissible Spongiform Encephalopathies)
Original Editor - Lucinda hampton
Top Contributors - Lucinda hampton and Nupur Smit Shah
Introduction[edit | edit source]
Prion diseases also known as transmissible spongiform encephalopathies (TSE's) are a group of progressive neurodegenerative conditions. They are progressive, fatal, and untreatable degenerative brain disorders and they exist in both animals and humans[1].
- Prion diseases result from misfolding of a normal cell-surface brain protein called cellular prion protein (PrPC), whose exact function is unknown. Misfolded prion proteins are called prions or scrapie PrP (PrPSc, from the name of the prototypic prion disease of sheep)[1].
- Scrapie, a disease affecting sheep and goats, was the first prion disease to be identified in the 1730s[2].
Prominent types include
- Creutzfeldt-Jakob disease (CJD), the prototypic example (usually sporadic). A rapidly progressive, rare, transmissible, universally fatal, neurodegenerative condition caused by prion proteins. The most common form, sporadic Creutzfeldt-Jakob disease (CJD), has a worldwide death rate of about 1 case per million people each year and is responsible for about 85% of all CJD cases worldwide. The cause of sporadic CJD remains unknown. Familial Creutzfeldt-Jakob disease (fCJD) is inherited as a result of genetic mutations and counts for 10-15% of the cases worldwide. The remaining cases are iatrogenic and variant Creutzfeldt-Jakob disease[3].
- Variant CJD (vCJD; acquired by eating prion-contaminated beef)
- Variably protease-sensitive prionopathy (VPSPr; sporadic)
- Gerstmann-Sträussler-Scheinker disease (GSS; inherited)
- Fatal insomnia (FI; includes a sporadic and an inherited form)
- Kuru (acquired by ritual cannibalism)[1]
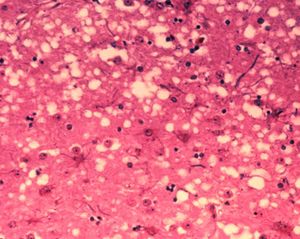
Image: This micrograph of brain tissue reveals the cytoarchitectural histopathologic changes found in bovine spongiform encephalopathy. The presence of vacuoles, i.e. microscopic “holes” in the gray matter, gives the brain of BSE-affected cows a sponge-like appearance when tissue sections are examined in the lab.
Mode of Action and Structure of Prions[edit | edit source]
A prion is a type of protein that can cause disease in animals and humans by triggering normally healthy proteins in the brain to fold abnormally.
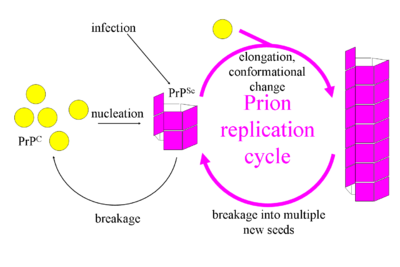
The prion mode of action is very different to bacteria and viruses as they are simply proteins, devoid of any genetic material. Once a misfolded prion enters a healthy person (potentially by eating infected food) it converts correctly-folded proteins into the disease-associated form. To date, nobody knows quite how this happens[4].
The 2 isoforms of Prions are:
1.PrP: These prion proteins are found on the cell membrane and play an important role in cell signalling and cell adhesion. More research is being carried out to discover its functions.
2. PrPsc: This is the disease-causing prion and is resistant to proteases. It affects the confirmation of PrPc and changes it. They are believed to have more beta sheets than the alpha helices. It also forms highly structured amyloid fibres. The other free proteins also attach to the end of these fibres. Similar prions with similar amino acids can only bind.[5]
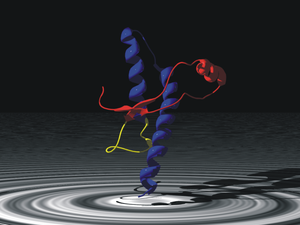
Image: Human prion protein. The coloring illustrates the subdomains, that are proposed to initiate the conversion from the normal cellular to the scrapie form by slow motions. Red: beta1-helix1-beta2. Blue: helix2-helix3
Etiology[edit | edit source]
Prion diseases are associated with the build up in the brain (and some other organs) of an abnormal or ‘rogue’ form of a naturally occurring cellular protein, known as the prion protein.
- The rogue protein results from a change in shape of the normal prion protein. They go on to recruit and convert more of the normal prion protein into the misfolded form.
- This misfolding sets off a cascade of events: the proteins oligomerise (a number of identical molecules join together); accumulate; nucleate (form a nucleus or centre); polymerise (combine to build a structure with its components); self-replicate; and eventually, propagate and spread throughout the brain[6].
- Normally once prion proteins have served their purpose, they are broken down by enzymes in the body.
- The abnormal prions however are more resistant to this process; so they accumulate and cause damage in the brain, which interferes with normal brain functioning.
- All forms of the disease are thought to be associated with an incubation period.
- This is a clinically 'silent phase' during which replication of the rogue protein is thought to be taking place[2].
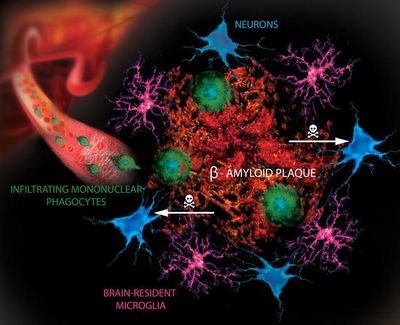
Scientists have always kept (and still do) an open mind about whether Alzheimer’s disease and other neurodegenerative conditions are transmissible. We’ve known since the early 1960s that amyloid fibrils (the accumulations of Aβ-amyloid in the brain) are self-propagating entities[6].
Causes[edit | edit source]
The normal form of the prion protein PrP (shown here) is found on the surface of nerve cells, but when it changes into its misfolded form, it aggregates into long fibrils that clog up the normal functioning of the brain.
Prion diseases can be of three types: acquired; sporadic; genetic.
- Acquired Prion Disease: Infection occurs when a little bit of the misfolded protein is eaten or accidentally gets into the blood through an injury. These prions are rare eg. A devastating example occurred in a native population in Papua New Guinea, where ritual cannibalism was part of funeral ceremonies. The epidemic probably started when one person developed the disease spontaneously (PrP occasionally adopts the misfolded state all by itself, causing very rare sporadic cases of the disease). Then the misfolded prions spread through the community when the infected person was eaten. More recently, there has been concern that the prions that cause mad cow disease could spread to humans by eating infected meat. The cow PrP protein is very similar to human PrP, and several cases of this type of infection have been seen.[3][7].
- Genetic Prion Disease: The familial prion diseases are caused as a result of genetic transmissions. However, it is not necessarily inherited from the ancestors. It may be caused due to the mutation in some DNA.
- Sporadic Prion Disease: Prion diseases are also believed to be sporadic. This means that its cause is not confirmed. This form of prion disease is most common to date[5].
Symptoms of Prion Disease[edit | edit source]
Following are the symptoms of Prion diseases:
- Developing dementia
- Hallucinations
- Fatigue
- Stiffening of muscles
- Confusion
- Difficulty in speaking[5]
Diagnosis[edit | edit source]
Prion diseases can present similar symptoms to other neurodegenerative disorders and are difficult to diagnose.
- To confirm a diagnosis of prion disease definitively a brain biopsy performed after death.
- A healthcare provider can use symptoms, medical history, and several tests to help diagnose prion disease.
- The tests they may use include: Magnetic resonance imaging (MRI): Cerebrospinal fluid testing (collected and tested for markers associated with neurodegeneration). In 2015, a test was developed to specifically detect markers of human prion disease; Electroencephalography (EEG)[8].
Treatment[edit | edit source]
There’s currently no cure for prion disease. However, treatment focuses on providing supportive care.
Examples of this type of care include:
- Medications. eg reducing psychological symptoms with antidepressants or sedatives; providing pain relief using opiate medication; easing muscle spasms with drugs like sodium valproate and clonazepam
- Assistance. As the disease advances, many people need help taking care of themselves and performing daily activities.
- Providing hydration and nutrients. In advanced stages of the disease, IV fluids or a feeding tube may be required.
Scientists continue to work to find an effective treatment for prion diseases[8].
Transmission[edit | edit source]
- Prion disease is not contagious: no evidence suggests it can be spread from person to person by close contact. Once a person has developed prion disease, central nervous system tissues (brain, spinal cord and eye tissue) are thought to be extremely infectious. However this is only relevant for those handling infected tissue directly, which does not include carers looking after a person with the disease.
- Infectivity in the rest of the body varies in different types of prion disease but is generally much less than in brain tissue. People with any form of prion disease are requested not to be blood or organ donors, and are requested to inform their doctor and dentist prior to any invasive medical procedures or dentistry.
- As prions cannot be completely destroyed by conventional sterilisation procedures, transmission has also occurred inadvertently through the use of surgical instruments previously used during neurosurgery on a person with sporadic prion disease. Instruments used on patients with known prion disease are not reused[2].
- Prions are not alive, so they can not be killed but they can be deactivated. Prions are highly resistant to disinfectants, heat, ultraviolet radiation, ionizing radiation and formalin. Prions can be destroyed through incineration providing the incinerator can maintain a temperature of 900 F for four hours. In an autoclave, prions can be deactivated by using a temperature of 270 F at 21 psi for 90 minutes. If the infectious material is in a solution of sodium hydroxide, deactivation will occur after one hour at 250 F and 21 psi[9].
The Future[edit | edit source]
Discovering the mechanism of replication of prions has been a very difficult task. To find out how can a protein multiply without any cellular machinery was next to impossible. But this is what prions do. There are many other concepts related to prions that are unclear. Research is being carried out to figure out more about prions and the diseases they cause.[5] eg.
- Antibodies targeting the normal PrP version of the prion protein have been found in humans selected at random with no history of any associated transmissible spongiform encephalopathies[10]. Antibodies against such proteins may be beneficial and offer potential for therapies against such diseases by targeting the pathological aggregates for degradation by phagocytic cells[10].
- Multiple system atrophy (MSA) is a prion disorder, according to data published online ahead of print August 31 in Proceedings of the National Academy of Sciences of the United States of America. A unique strain of α-synuclein prions causes MSA, and these prions are different from the prions believed to cause Parkinson’s disease.[11]
References[edit | edit source]
- ↑ 1.0 1.1 1.2 MSD manual Prion Diseases Available from:https://www.msdmanuals.com/professional/neurologic-disorders/prion-diseases/overview-of-prion-diseases (last accessed 9.12.2020)
- ↑ 2.0 2.1 2.2 UCL Prions Available from: http://www.prion.ucl.ac.uk/clinic-services/information/prion-disease/ (last accessed 9.12.2020)
- ↑ 3.0 3.1 WHO Prion Diseases Available from:https://www.who.int/zoonoses/diseases/prion_diseases/en/ (last accessed 9.12.2020)
- ↑ Microbiology society prions Available from;https://microbiologysociety.org/why-microbiology-matters/what-is-microbiology/prions.html (last accessed 10.12.20200
- ↑ 5.0 5.1 5.2 5.3 Byjus Prions Available from;https://byjus.com/biology/prions/ (last accessed 9.12.2020)
- ↑ 6.0 6.1 The Conversation New prion disease raises questions about whether Alzheimer’s and Parkinson’s could be infectious Available from;https://theconversation.com/new-prion-disease-raises-questions-about-whether-alzheimers-and-parkinsons-could-be-infectious-46936 (last accessed 9.12.2020)
- ↑ Corner E, Geis I, Goodsell D. Molecule of the Month.Available from: https://pdb101.rcsb.org/motm/101(accessed 10.12.2020)
- ↑ 8.0 8.1 Healthline Prion Disease Available from:https://www.healthline.com/health/prion-disease#diagnosis (last accessed 10.12.2020)
- ↑ Soctaric society Prions Available from: https://socratic.org/questions/how-can-prions-be-killed (accessed 10.12.2020)
- ↑ 10.0 10.1 Medical Press Naturally occurring antibodies against prion proteins found in humans Available from:https://medicalxpress.com/news/2020-08-naturally-antibodies-prion-proteins-humans.html (last accessed 9.12.2020)
- ↑ Prusiner SB, Woerman AL, Mordes DA. MSA May Be a Prion Disorder. Available from: https://www.mdedge.com/neurology/article/103118/rare-diseases/msa-may-be-prion-disorder Accessed 1012.2020