Primary Lateral Sclerosis (PLS): A Case Study
ABSTRACT[edit | edit source]
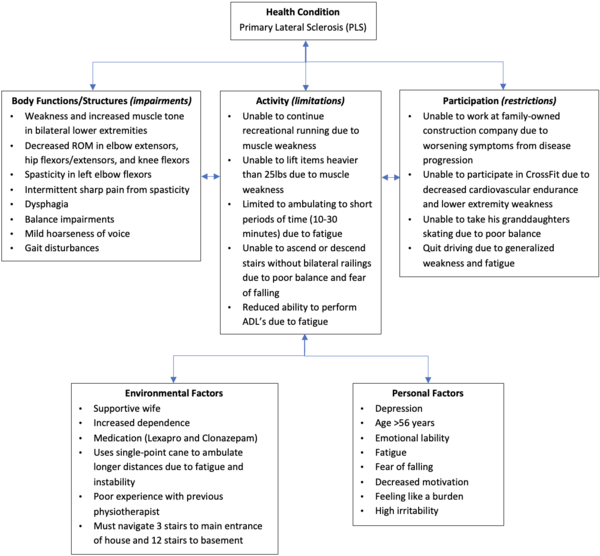
This fictional case study involves Mr. Parker, a 56-year-old male, who was recently diagnosed with Primary Lateral Sclerosis (PLS). PLS is a rare neurodegenerative disease of the upper motor neurons (UMN) and is characterized by progressive muscle weakness and stiffness in voluntary muscles. The purpose of the case study is to explore possible management strategies and barriers encountered by an individual with early stage PLS. Mr. Parker first presented with progressive muscle weakness in his lower limbs three years ago and was initially misdiagnosed with Amyotrophic Lateral Sclerosis (ALS). Upon recent assessment, Mr. Parker’s neurologist confirmed a PLS diagnosis through physical assessment and diagnostic tools - including Magnetic Resonance Imaging (MRI), electromyography (EMG), and nerve conduction studies. Mr. Parker was referred to physiotherapy (PT) by his neurologist. Mr. Parker was prescribed an intervention program by his physiotherapist that focused on patient education, stretching, strengthening, balance training, and fall prevention strategies to maintain or improve his symptoms. Upon reassessment four months later, Mr. Parker showed either maintenance or improvement in muscle strength, spasticity, gait measures, and walking endurance. However, his balance scores decreased post-intervention, putting him at increased risk of falls. Throughout Mr. Parker's care, referrals were made to other interdisciplinary professionals, as appropriate.
INTRODUCTION[edit | edit source]
Primary Lateral Sclerosis (PLS) is a rare, idiopathic progressive disease that affects the upper motor neurons (UMN) [1][2]. Degeneration of the motor cortex and corticospinal tracts will ensue with no loss of lower motor neurons (LMN) or disruption to the anterior horn cells [3][4][5][6]. While the clinical presentation of PLS is very similar to that of Amyotrophic Lateral Sclerosis (ALS), PLS only affects the UMNs while ALS affects both the UMNs and LMNs [1][2][6]. As a result, this often leads to PLS being misdiagnosed as ALS [1][2][3][4][5][6][7]. In comparison to ALS, the prognosis of PLS is considered to cause less harm as it is not fatal in most cases [1] [2]. A true diagnosis of PLS can only occur many years later when it is confirmed that there is no LMN disruption [3][4][5][6][7]. There is little research surrounding PLS as it is not only similar to ALS but to a lot of other neurological conditions as well [2].
Previous case studies found that the clinical signs and symptoms of PLS had to be present, on average, for a minimum of three to five years to conclude a definite diagnosis [3][4][5][6][7]. Additionally, those with PLS rarely have a family history of it [1][2][7]. PLS is a slowly progressive disease that presents more commonly in males than females [3][4][5][6][7]. The onset of the disease usually occurs between 40 to 60 years of age, with 50 to 55 years of age being the average age in most samples studied [1][3][4][5][6][7].
PLS is characterized by progressive weakness and stiffness in voluntary muscles, typically starting in the lower extremity [1][2][3][4][5][6][7]. The disease may then progress to the trunk, followed by the upper extremity, and lastly to the corticobulbar tract, typically causing a pseudobulbar affect (emotional lability) [1][2][3][4][5][6][7]. In addition, patients may also experience dysarthria (difficulty speaking) and dysphagia (difficulty swallowing) [1][2][3][4][5][6][7]. In a case study by Wais et al. (2016), they found that very few patients displayed a symmetrical distribution of symptoms [7]. Some patients may undergo latent periods where symptoms seem to subside slightly to allow for more normal functioning [3][4][6]. Typical signs and symptoms that patients reported included: weakness, clumsiness, hyperreflexia, increased muscle tone (spasticity), and increased urinary urgency [3][4][6]. However, the first sign that patients typically described was a disruption in the smoothness of their gait cycle [1][2][3][4][5][6][7]. Patients with comorbidities may present with some signs and symptoms that are not typical to the clinical presentation of PLS, including: sensory symptoms and LMN degeneration such as muscle atrophy, fasciculations, and decreased reflexes or tone [2][3]. Most patients will experience a slow spread of symptoms over many years before plateauing, indicating a better prognosis and more independent life than ALS [1][2]. Death is typically as a result of bulbar dysfunction, malnutrition, or breathing decline [4][6].
With respect to the fictional case study of Mr. Parker, the main challenge experienced during the physiotherapy (PT) sessions was managing his sporadic emotional outbursts. This presentation aligns with the emotional lability characteristic of PLS. While these outbursts did not happen often, it did take some time to refocus the session back to the activities that were being performed prior.
The purpose of this fictional case study is to provide a brief overview of what PT care may involve in an out-patient setting with respect to the assessment and management of an individual in the early stage of PLS. Further, we will discuss the role of PT in the management and maintenance or adaptation of function associated with PLS. There is currently limited research on PLS which is why this fictional case study aims to provide a framework for this gap in literature.
CLIENT CHARACTERISTICS[edit | edit source]
Mr. Parker is a retired 56-year-old male, right-hand dominant, who presents with early stage PLS. Until about three years ago (2019), he was an avid Cross Fitter, participated in leisure activities with his two granddaughters, and worked part-time at his family-owned construction company. Previously, he would train at the gym 2-3 days per week and would go on 3-5 km runs twice a week. Mr. Parker first presented to his family physician three years ago with reports of weakness in his legs that had progressed over time to cause clumsiness and stiffness during his runs. He reported that he “had to stop running because of a fear of falling”. He was then diagnosed with ALS in 2019 by a neurologist through subjective history, physical examination, and diagnostic tools (MRI, EMG, and nerve conduction studies). However, upon a recent reassessment in 2022, the neurologist concluded that Mr. Parker has PLS, as his symptoms did not progress to the LMNs - which was concluded by a secondary physical examination and diagnostic tools. Mr. Parker’s neurologist initially referred him to PT in 2019 following his ALS diagnosis. However, he still recommends that Mr. Parker continues with his rehabilitation following his PLS diagnosis for maintenance purposes.
Since the initial diagnosis of ALS, Mr. Parker's health status has slowly deteriorated. Mr. Parker was prescribed a single-point cane with a 2-point step through pattern by his previous PT in 2019, which he uses on occasion.
In 2020, he was forced into an earlier-than-expected retirement as he was unable to keep up with part-time hours and work duties at the construction company. Furthermore, he is now unable to participate in any recreational running or CrossFit training due to the severity and worsening of his symptoms. He has now adjusted his fitness regime, using resistance bands to work out from time to time at home as well as trying to go for daily walks with his wife totaling 10-30 minutes. His home activities have also been reduced due to decreased activity tolerance, decreased endurance, and fatigue. Therefore, his wife has taken on more responsibilities at home.
Upon reassessment in 2022, Mr. Parker reports that he is starting to experience swallowing difficulties and painful spastic posturing in his left arm. Overall, his condition continues to remain relatively steady since the initial onset, although he has since started to consistently take medication (Lexapro) for his concurrent depression. Additionally, his wife describes that he has been having intermittent, uncharacteristic behavioral outbursts as of two months ago.
EXAMINATION FINDINGS[edit | edit source]
Subjective Assessment[edit | edit source]
Patient Profile[edit | edit source]
- Date of Birth (DOB): May 9, 1966 (56 years old)
- Gender: Male
- Hand Dominance: Right-hand dominant
- Significant Presentation: PLS (misdiagnosed as ALS three years ago)
History of Present Illness[edit | edit source]
Insidious onset of bilateral leg weakness and stiffness which Mr. Parker noticed during his daily runs three years ago. Symptoms developed to include painful spastic posturing in the left arm, difficulty balancing, clumsiness, intermittent dysphagia, and generalized fatigue. He experienced several falls over the last few years due to poor balance control during ambulation. He developed a fear of falling that has greatly limited his participation in work, leisure activities, and general ambulation. He discussed his onset of symptoms with his family physician, who referred him to a neurologist in Toronto. He was initially diagnosed with ALS about three years ago (2019) by his neurologist, who then updated the diagnosis to PLS about five weeks ago after reassessment.
Pain: Describes intermittent, sharp pain isolated to the left arm, which refers down the shoulder to the elbow. The onset of pain occurs during active elbow extension. Reports pain 6/10 with movement and 0/10 at rest. Takes 30 minutes for the pain to subside (moderate irritability).
Past Interventions:
- Physiotherapy (PT): 2.5 years ago with no effect.
- Previous therapist only used passive treatment (i.e., stretching, PROM, and modalities like NMES to treat the lower extremity), which Mr. Parker and his family were not fond of.
- Massage Therapy: Attended weekly for 3 months (2020) with no effect.
- Acupuncture: Attended weekly for 3 months (2020) with no effect.
- Pharmacological Agents: Baclofen (prescribed by a neurologist to treat muscle spasticity) but stopped after a couple of months as it was not helpful and the side effects outweighed the benefits associated with the medication.
Past Medical History[edit | edit source]
- Depression
- Otherwise functionally healthy
Medications[edit | edit source]
- Lexapro (10 mg OD, for 8 weeks) - to treat depression
- Clonazepam (0.5 mg TID) - to treat muscle spasticity
Health Habits[edit | edit source]
- Smoking History: Non-smoker
- Alcohol Consumption: Maximum of 4 alcoholic drinks per week
- Recreational Drug Use: N/A
Family History[edit | edit source]
- Mother: Diabetes and Hypertension
- Father: Colon Cancer
- No family history of PLS or other neurodegenerative diseases
Psychosocial Factors[edit | edit source]
Mr. Parker experiences feelings of depression often such as hopelessness, frustration, and general sadness. He reports a strong fear of falling due to his poor balance and muscle weakness, and uses the walls, railings, and installed support bars throughout his home for support. He reports decreased confidence and motivation with ambulation and exercise. Mr. Parker has found it difficult adjusting to his current, less active lifestyle. He constantly worries about burdening his wife with their household chores, especially because of her lower back pain. The growing conflict between his decreased ability to perform his usual activities and desire to help his wife and family business has made him increasingly irritable and restless.
Social History[edit | edit source]
Mr. Parker lives in a bungalow with his wife and two small dogs. The house has three concrete steps leading up to the front door (rails x2), and 12 stairs inside to get down to the basement (rails x2). There is one bedroom on the main floor with a full bathroom and a half bathroom closer to the living room. There is hardwood flooring throughout the hallways, tile floor in the kitchen, and carpet in the bedroom. He enjoys spending time with his two granddaughters - including taking them to the park by his house during the summer and skating with them during the winter. He used to work part-time at his family-owned construction company which he enjoyed greatly, but has had to retire early in 2020 due to the progression of his disease.
Functional History[edit | edit source]
Mr. Parker reports being very active prior to disease onset as he participated in CrossFit, attended the gym 2-3 times/week, and ran 3-5km twice a week. Until about a year ago (2021), Mr. Parker would be able to drive himself around, but preferred to walk or get his wife to drive him. He was able to complete all activities of daily living, household chores, and property management (i.e. mowing the lawn and gardening) independently. Prior to his diagnosis, he did not use any mobility aid and ambulated independently with no balance issues or worries of falls. He was prescribed a single-point cane by his previous physiotherapist in 2019.
Current Functional Status[edit | edit source]
He is currently able to ambulate for 10-30 minutes independently with no mobility aid. He utilizes a single-point cane with a 2-point step through pattern when feeling fatigued or when he does not feel confident ambulating longer distances. When using the stairs, he either uses rails x2 or rails x1 with a single point cane due to his instability.
At home, he currently performs light upper extremity exercises using a resistance band. He is able to perform light household chores such as dusting, folding laundry, and washing dishes, but needs assistance from his wife for heavy chores like vacuuming, shopping, or lifting anything heavier than 25 lbs. He is currently able to perform self-care activities independently with no reports of difficulty. As mentioned above, Mr. Parker has refrained from driving since 2021 due to his generalized weakness and fatigue. He reports feeling unbalanced in a variety of situations such as ascending/descending stairs with no support, ambulating longer distances, and intermittently when standing.
Imaging and Medical Testing[edit | edit source]
EMG: Found sparse fasciculation in bilateral calves. Conducted on April 3, 2022.
Nerve Conduction Studies: No outstanding results were found. Conducted in both May 2021 and April 2022.
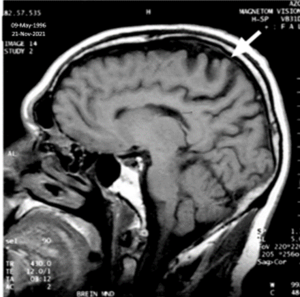
MRI: Sagittal T1 weighted image of the brain showing atrophy of the pre-central area and degeneration of the underlying white matter [10]. Imaged on November 21, 2021.
Precautions/ Contraindications[edit | edit source]
Concurrent depression, emotional lability (particularly regarding heightened irritability and uncontrollable sadness), and intermittent dysphagia.
Objective Assessment[edit | edit source]
General Observations[edit | edit source]
| Standing Posture |
|
| Sitting Posture |
|
Speech and Language[edit | edit source]
Mr. Parker’s wife indicates that the hoarseness in his voice became apparent several months ago and was not present before. His language abilities are intact upon examination and mild notes of hoarseness was noted during the assessment.
Tone and Sensation[edit | edit source]
Tone was assessed using the Modified Ashworth Spasticity Scale (MAS). Findings include:
| Muscle Group | Grade |
|
3 |
|
1+ |
Sensation was assessed using the dermatomal pattern for light touch along upper and lower extremities, as well as vibration sense in lower extremities. There was no abnormality in sensation found.
Muscle Strength[edit | edit source]
Global muscle strength for the upper and lower extremities was measured using manual muscle testing (MMT). See tables below.
Upper Extremity MMT:
| Muscle Group | Grade: Left | Grade: Right |
| Shoulder Flexors | 4/5 | 5/5 |
| Shoulder Extensors | 4/5 | 5/5 |
| Shoulder Abductors | 4/5 | 5/5 |
| Shoulder Adductors | 4/5 | 5/5 |
| Shoulder Internal Rotators | 4-/5 | 5/5 |
| Shoulder External Rotators | 4/5 | 5/5 |
| Elbow Flexors | 4+/5 | 5/5 |
| Elbow Extensors | 4/5 | 5/5 |
Lower Extremity MMT:
| Muscle Group | Grade: Left | Grade: Right |
| Hip Flexors | 4/5 | 4/5 |
| Hip Extensors | 4-/5 | 4-/5 |
| Hip Abductors | 4+/5 | 4+/5 |
| Hip Adductors | 4+/5 | 4+/5 |
| Hip Internal Rotators | 4+/5 | 4+/5 |
| Hip External Rotators | 4+/5 | 4+/5 |
| Knee Flexors | 4/5 | 4/5 |
| Knee Extensors | 4-/5 | 4/5 |
| Ankle Plantar Flexors | 3+/5 | 3+/5 |
| Ankle Dorsiflexors | 4/5 | 4/5 |
Active Range of Motion (AROM)[edit | edit source]
AROM was measured using a goniometer.
Upper Extremity AROM:
Limitations in AROM were noted for the movements in the table below. All other movements were within normal limits (WNL) bilaterally.
| Movement | Left | Right |
| Elbow Extension | 10° of flexion | 0° |
Lower Extremity AROM:
Limitations in AROM were noted for the movements in the table below. All other movements were WNL bilaterally.
| Movement | Left | Right |
| Hip Flexion | 90° | 90° |
| Hip Extension | 5° | 5° |
| Knee Flexion | 125° | 125° |
Trunk AROM:
All movements for trunk AROM were WNL bilaterally.
Passive Range of Motion (PROM)[edit | edit source]
PROM was measured using a goniometer. Measurements obtained were recorded as WNL when moved slowly (i.e. completing the full movement within 4-5 seconds). All movements in the upper and lower extremity were WNL bilaterally, with the exception of elbow extensors:
- Spastic increase in elbow flexors when moved rapidly (<0.5s) through joint PROM of elbow extension, resulting in 10 degrees off from full extension.
Balance[edit | edit source]
Balance was assessed using the Berg Balance Scale (BBS) at baseline. Score achieved: 46/56. This score indicates the patient is independent and should not require assistance to ambulate safely. However, this score does indicate that Mr. Parker is at an increased risk for falls.
Gait[edit | edit source]
Noted wide-based stance, slowed gait, and driving feet into the ground with each step. Left arm kept in full flexion and absence of arm swing on left side (an attempt to avoid onset of spastic episode of elbow flexors).
Ambulates with a single-point cane with 2-point step through pattern for longer distances. Does not use it daily.
| Outcome Measure | Score | Interpretation |
| Gait Speed Test (4 meters) | 0.82 meters/second | Indicates that Mr. Parker may safely ambulate in the community independently, but will need interventions in place to reduce his falls risk. |
| Tandem Gait Test | 10 steps | Mr. Parker completed 10 steps of the tandem walk without the use of his cane in an unsteady and uncoordinated manner, having to widen his stance with almost every step. This is indicative of mild balance and gait disturbances. |
DIAGNOSIS[edit | edit source]
Mr. Parker is a previously active and independent 56-year-old right-hand dominant retired construction worker referred to PT by his neurologist for management of progressive PLS, diagnosed in 2022. Mr. Parker was initially misdiagnosed with ALS in 2019. Since then, his functional abilities have been slowly deteriorating. Mr. Parker presents with bilateral weakness in the lower extremities, painful spastic posturing in left arm, intermittent dysphagia, decreased independence with Activities of Daily Livings (ADLs), and decreased activity tolerance affecting his ability to participate in meaningful activities. Due to the progressive nature of PLS, there is low likelihood Mr. Parker will recover complete functional independence. Mr. Parker is a suitable candidate for PT aimed at maintaining lower extremity strength and functional mobility, managing pain, education on self-management strategies, and activity modification to optimize participation in day-to-day life and previously enjoyed activities.
PROBLEM LIST[edit | edit source]
PATIENT GOALS[edit | edit source]
Short-Term Goals (STG): 0-6 Weeks[edit | edit source]
- Mr. Parker will be able to describe three fall prevention strategies to use at home without prompts from the therapist in 1 week (education).
- Mr. Parker will be able to independently complete 12 or more consecutive steps in tandem walking without using the stepping strategy in 3 weeks (activity).
- Mr. Parker will maintain the strength of all lower extremity global muscle groups relative to baseline assessment MMT scores in 6 weeks (body structure and function).
Long-Term Goals (LTG): 7-17 Weeks[edit | edit source]
- Mr. Parker will be able to perform a vertical transfer from ground to chair with less than two errors while maintaining safety in 8 weeks (activity).
- Mr. Parker will be able to participate in his daily walks with his wife around the neighborhood blocks (ambulating approx. 1500 meters) with the use of a single-point cane with a 2-point step through gait pattern in 11 weeks (participation).
- Mr. Parker will be able to ascend and descend 12 stairs safely to get to the basement of his house with the support of one railing and no use of a mobility aid without loss of balance in 17 weeks (activity).
INTERVENTION[edit | edit source]
Management Program[edit | edit source]
PLS treatment strategies are aimed at managing patient symptoms and maintaining functional abilities. A goal-oriented and functional intervention program was created for Mr. Parker that considered his disease progression, preferences, and goals. The program highlights activities that will be performed in an outpatient neurological rehabilitation clinic in Toronto and activities that he will be coached to perform at home as part of his home exercise program. Education, strengthening, and flexibility are the central aspects of the treatment plan. Additionally, growing research has shown that getting patients with neurological disorders to stand more often is beneficial for ROM, spasticity, and psychological well-being [11]. Standing has been shown to promote the maintenance of anti-gravity muscles in the trunk and lower limbs [11]. For this reason, Mr. Parker is encouraged to perform as many of these exercises in standing as tolerated.
Out-patient Neurorehabilitation Treatment Plan[edit | edit source]
Education[edit | edit source]
The teach-back method was used to confirm patient understanding.
| Topic | Description |
| Fall Prevention Strategies |
|
| Spasticity |
|
| Pain Management |
|
Intervention[edit | edit source]
| Topic | Description | Parameters |
| Balance and Fall Prevention Strategies |
|
F: 2 days/week
I: As tolerated T: 3 sets of 30 seconds reps |
| Behavioral Therapy |
|
Ongoing |
| Virtual Reality System (VRS) |
|
Introduced after 4 weeks in PT
F: 2 days/week T: 10 minutes |
| MR Cube Monitored Rehab Tool | F: 1-2 days/week
I: as tolerated T: 10-15 minutes | |
| OmniStand Dynamic Balance system |
|
F: 2 days/ week in combination with VRS from week 4.
T: 10 minutes |
| Rhythmic Cycling |
|
F: 2 days/week
I: Moderate intensity T: 15-20 minutes, or as tolerated T: Aerobic |

Home Exercise Program[edit | edit source]
Stretching[edit | edit source]
| Parameter | Description | Exercises |
| Frequency | 2-3 days/week; encouraged daily | Upper Extremity:
Lower Extremity:
|
| Intensity | To the point of tightness or slight discomfort; no pain | |
| Time | Hold stretch for 30 seconds, for an accumulation of 60 seconds/day | |
| Type | Static |
Strengthening[edit | edit source]
| Parameter | Description | Exercises |
| Frequency | 2-3 days/week |
|
| Intensity | 8-12 reps (to muscle fatigue, but not pain)
2-4 sets 70-80% 1RM | |
| Time | 2-3 min rest between sets | |
| Type | Hypertrophy |
Balance Training[edit | edit source]
| Parameter | Description | Exercises |
| Frequency | 2-3 days/week | Internal Perturbations:
External Perturbations:
|
| Intensity | As tolerated | |
| Time | 10-12 reps | |
| Type | Balance |
Aerobic Training[edit | edit source]
| Parameter | Description | Exercises |
| Frequency | 5-7 days/week |
|
| Intensity | Light-to-moderate intensity | |
| Time | 10-30 minutes, as tolerated | |
| Type | Aerobic |
OUTCOMES - Reassessment After Four Months[edit | edit source]
While there are currently no standardized outcome measures for PLS, there are generalized outcome measures that can be used to assess different components of a patient’s presentation and needs. Clinical judgment was used to select which outcome measures to use.
The following is the list of outcome measures used to assess the degree of improvement or change in patient status post-intervention. The post-intervention period was 4 months.
| Outcome Measure | Justification | Pre-Intervention Score | Post-Intervention Score
(4 months) |
| Gait Speed Test
(4 meters) |
To assess walking speed over a predefined distance (4 meters), and monitor functional mobility. No mobility aid was utilized during the test. | 0.82 meters/second | 0.81 meters/second |
| Interpretation: Minimal reduction in gait speed by 0.01 meters/second. Indicative that the patient is able to ambulate safely in the community without further assistance. May use an assistive device for endurance purposes or to increase patient confidence with ambulation. | |||
| Tandem Gait Test | To assess balance and gait disturbances. | 10 steps | 14 steps |
| Interpretation: Able to complete an increased number of tandem steps in a more controlled and steady manner. Patient had to widen his stance to complete the test, both pre- and post-intervention. Indicative of potential balance and gait disturbances. | |||
| Manual Muscle Testing (MMT) | To monitor muscle strength of the lower extremity. | Bilaterally:
Hip Extensors: 4-/5 Knee Extensors: 4-/5 Ankle Dorsiflexors: 4-/5 |
Bilaterally:
Hip Extensors: 3+/5 Knee Extensors: 4-/5 Ankle Dorsiflexors: 4-/5 |
| Interpretation: Maintenance of muscle strength in knee extensors and ankle dorsiflexors. Decreased hip extensor strength post-intervention. Muscle strength in the upper extremity and remaining segments of the lower extremity maintained an MMT score of 4/5 pre- to post-intervention. | |||
| Berg Balance Scale (BBS) | To assess balance and its correlation to predict falls risk. | 46/56 | 44/56 |
| Interpretation: Both pre-and post-intervention scores correlate with an increased falls risk. BBS score decreased by 2 points post-intervention, indicative of a greater falls risk. | |||
| Modified Ashworth Scale (MAS) | To assess and monitor the level of muscle spasticity in the limbs. The MAS is valid for the central nervous system lesion population, which includes PLS [15]. | Upper Extremity
Left: Elbow Flexors: 3 Elbow Extensors: 0 Right: Elbow Flexors: 1+ Elbow Extensors: 0 |
Upper Extremity
Left: Elbow Flexors: 2 Elbow Extensors: 0 Right: Elbow Flexors: 1+ Elbow Extensors: 0 |
| Lower Extremity
Bilaterally: Hip Flexors: 3 Hip Extensors: 1+ Knee Flexors: 1+ Knee Extensors: 0 |
Lower Extremity
Bilaterally: Hip Flexors: 2 Hip Extensors: 1 Knee Flexors: 1+ Knee Extensors: 0 | ||
| Interpretation:
Upper Extremity: Reduced muscle spasticity in left elbow flexors post-intervention, and maintenance of minimal spasticity in right elbow flexors and no spasticity in elbow extensors bilaterally. Lower Extremity: Reduced muscle spasticity in hip flexors and extensors bilaterally post-intervention. No change in score of muscle spasticity in knee flexors and extensors bilaterally post-intervention. | |||
Mr. Parker’s condition has remained relatively stable four months post-intervention. The outcome measures will continue to be monitored throughout his time at PT. Further assessments of upper and lower extremity muscle weakness, spasticity, and ROM will continue to be done, in addition to balance and gait cycle measures.
Progress in Other Interventions:
- Behavioral therapy: Behavioral therapy has shown promising results post-intervention. Patient reports that he is better able to manage and recognize what triggers his emotional outbursts.
- Fall Prevention and Balance Strategies: Patient is well-educated on various fall prevention and balance strategies, and is able to effectively demonstrate them as needed. Patient also reports a decreased fear of falling which he owes to his increased confidence with a mobility aid and fall prevention education.
REFERRALS[edit | edit source]
While conversations were had with Mr. Parker and his wife early on with regards to referrals to other health care professionals, it was only recently decided to further explore other available services - including speech-language pathology (SLP), psychiatry, and occupational therapy (OT).
- Speech-Language Pathology (SLP): To assess swallowing impairments and monitor speech abilities. Services provided may include the application of speech synthesizers, swallowing therapy, improvement of larynx elevation, and strengthening of tongue and suprahyoid muscles [16][17].
- Psychiatrist: To assess and utilize cognitive-behavioral therapy (CBT) to aid in the management of behavior and cognitive changes experienced by the patient. Provide emotional support to the patient and family.
- Occupational Therapy (OT): To assess and practice day-to-day tasks, suggest home equipment and assistive devices. To assist in the elimination of hazards in the patient’s environment by providing feedback on changes to the layout of the home, installation of bathroom equipment (i.e.., grab bars, bath bench, etc.), and ensure adequate lighting to aid in fall prevention.
- Medical/Pharmaceutical:
- To prescribe oral medications and the appropriate dosage for management of spasticity such as tizanidine or valium [18].
- Growing research has shown the benefits of using stem cell in slowing the progression of ALS [19]. Considering the similarities between ALS and PLS, there has been a move to consider stem cell therapy as an alternate therapeutic strategy for PLS [19].
DISCHARGE PLAN[edit | edit source]
Due to the progressive nature of Mr. Parker’s condition, a discharge plan is not indicated and ongoing assessments with his physiotherapist are recommended to monitor and manage his symptoms and physical functioning.
Going forward, less frequent PT visits are required as Mr. Parker is managing his symptoms well, is compliant with the treatment protocol, and has developed appropriate self-management strategies that he can utilize at home and in the community.
DISCUSSION[edit | edit source]
PLS is a rare and sporadic UMN disease that is often mistaken for ALS [20]. In addition to impeding functional activities, muscle weakness, spasticity, and loss in ROM, this condition can negatively impact one’s outlook on life. This comprehensive fictional case study was developed to increase the awareness of PLS and to integrate evidence-based practice in the assessment and management of this condition.
Mr. Parker is a 56-year-old male who was initially diagnosed with ALS three years ago in 2019. Upon further assessment of his symptoms, he was diagnosed with PLS in 2022. Prior to his diagnosis, Mr. Parker was an active and independent individual. He regularly participated in recreational running, CrossFit training, and working out at the gym until the presentation of his symptoms. His PT assessment revealed global muscle weakness in the lower extremity, increased tone in left elbow flexors and bilateral hip flexors, painful spastic posturing, and reduced ROM in his left arm. He also reported episodes of dysphagia and issues with balance contributing to his fear of falling. Additionally, Mr. Parker reported that it has been difficult for him to adjust to the new trajectory of his life and the adaptions he has had to make.
PLS has a multidimensional impact on the emotional, physical, and social aspects of an individual's life. An intervention program focusing on education, maintaining strength and ROM, reducing spasticity, and improving balance was created to improve Mr. Parker’s quality of life and ability to participate in meaningful activities. Additionally, Mr. Parker has been taught fall prevention strategies and has received CBT. Referrals were made to SLP, psychiatry, and OT, as well as follow-ups in regards to the management of medications were encouraged.
While other progressive diseases like ankylosing spondylitis, muscular dystrophy, and Alzheimer’s disease, have disease-specific treatment plans, there is yet to be a treatment plan specific to PLS. Despite this, when designing a treatment plan for a patient with PLS, it can be suitable to use diagnostic tools, outcome measures, and interventions that are specific to UMN disorders and other related constructs. In the case of Mr. Parker, the goal of his PT treatment was to manage and maximize his function within the contrasts of his progressive pathology. The ICF framework was a key component for identifying the main problems that needed to be addressed (i.e., activity limitations, participation restrictions, etc.). The patient-centered approach allowed for the development of an individualized and comprehensive care plan. The focus was placed on promoting patient autonomy and creating meaningful and realistic goals with the patient.
Evidence-based recommendations in terms of diagnostic tools, outcome measures, and treatment specific to PLS is limited, as indicated in this case study. Therefore, it is paramount for further research to take place regarding the assessment and treatment of PLS. Building upon the current research on PLS will aid in our understanding of the condition, appropriate management strategies, and improved treatment interventions. Additionally, there should be some consideration towards investigating proactive measures that may delay onset of the disease itself. Aspects of future research may also be applicable to other UMN diseases.
References[edit | edit source]
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 Physiopedia. Primary Lateral Sclerosis. Available from: https://www.physio-pedia.com/Primary_Lateral_Sclerosis [last accessed 09/05/22]
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 Mayoclinic. Primary lateral sclerosis (PLS). Diseases and Conditions. Available from: https://www.mayoclinic.org/diseases-conditions/primary-lateral-sclerosis/symptoms-causes/syc-20353968 [last accessed 09/05/22]
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 Turner MR, Barohn RJ, Corcia P, Fink JK, Harms MB, Kiernan MC, Ravits J, Silani V, Simmons Z, Statland J, van den Berg LH; Delegates of the 2nd International PLS Conference, Mitsumoto H. Primary lateral sclerosis: consensus diagnostic criteria. J Neurol Neurosurg Psychiatry. 2020 Apr;91(4):373-377. doi: 10.1136/jnnp-2019-322541.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 Gordon PH, Cheng B, Katz IB, Pinto M, Hays AP, Mitsumoto H, Rowland LP. The natural history of primary lateral sclerosis. Neurology. 2006 Mar;66(5):647-53. doi: 10.1212/01.wnl.0000200962.94777.71
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 5.8 5.9 Le Forestier N, Maisonobe T, Spelle L, Lesort A, Salachas F, Lacomblez L, Samson Y, Bouche P, Meininger V. Primary lateral sclerosis: further clarification. J Neurol Sci. 2001 Apr;185(2):95-100. doi: 10.1016/s0022-510x(01)00469-5.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 Zhao, C, Lange, DJ, Wymer, JP. Management of Primary Lateral Sclerosis. Current Treatment Options Neurology. 2020 Aug; 22(31). https://doi.org/10.1007/s11940-020-00640-6 [last accessed 12/05/22]
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 Wais, V, Rosenbohm, A, Petri, S, Kollewe, K, Hermann, A, Storch, A, Hanisch, F, Zierz, S, Nagel, G, Kassubek, J, Weydt, P, Brettschneider, J, Weishaupt, JH, Ludolph, AC, and Dorst, J. The concept and diagnostic criteria of primary lateral sclerosis. Acta Neurologica Scandinavica. 2016 Oct;136: 204– 211. https://doi.org/10.1111/ane.12713h [last accessed 12/05/22]
- ↑ Faldi Yaputra. Primary Lateral Sclerosis. Available from: https://www.youtube.com/watch?v=dcyAMDAPEeI [last accessed 11/05/22]
- ↑ Stitching PLS. Primary lateral sclerosis, English subtitles Available from: https://www.youtube.com/watch?v=Zpt1-QTUbXE [last accessed 11/05/22]
- ↑ 10.0 10.1 Kuipers-Upmeijer J, Jager AEJde, Hew JM, Snoek JW, Weerden TWvan. Primary lateral sclerosis: Clinical, neurophysiological, and magnetic resonance findings. Journal of Neurology, Neurosurgery & Psychiatry.2001. Available from: https://jnnp.bmj.com/content/71/5/615 [last accessed 10/05/22]
- ↑ 11.0 11.1 Stevenson VL. Rehabilitation in practice: spasticity management. Clin Rehabil. 2010 Apr; 24(4):293-304. doi: 10.1177/0269215509353254
- ↑ 12.0 12.1 12.2 Cano Porras D, Sharon H, Inzelberg R, Ziv-Ner Y, Zeilig G, Plotnik M. Advanced virtual reality-based rehabilitation of balance and gait in clinical practice. Ther Adv Chronic Dis. 2019 Aug 23;10:2040622319868379. doi: 10.1177/2040622319868379.
- ↑ 13.0 13.1 13.2 MR cube - balance rehabilitation system by Mettler Electronics. Medical EXPO. Available from: https://www.medicalexpo.com/prod/mettler-electronics/product-121639-844767.html [last accessed 11/05/22]
- ↑ 14.0 14.1 14.2 14.3 OMNISTAND® - Balance Rehabilitation System by ACP: Medicalexpo. Medical EXPO.. Available from:https://www.medicalexpo.com/prod/acp/product-84231-929447.html [last accessed 11/05/22]
- ↑ Morris S. Ashworth and Tardieu scales: Their clinical relevance for measuring spasticity in adult and paediatric neurological populations. Physical Therapy Reviews. 2002 Mar;7(1),53-62. doi: 10.1179/108331902125001770
- ↑ 16.0 16.1 16.2 Martindale N, Stephenson J, Pownall S. Neuromuscular Electrical Stimulation Plus Rehabilitative Exercise as a Treatment for Dysphagia in Stroke and Non-Stroke Patients in an NHS Setting: Feasibility and Outcomes. Geriatrics. 2019; 4(4):53. https://doi.org/10.3390/geriatrics4040053 [last accessed 11/05/22]
- ↑ 17.0 17.1 17.2 Sura L, Madhavan A, Carnaby-Mann G, Crary M. Dysphagia in the elderly: management and nutritional considerations. Clin Interv Aging. 2012;7:287-298 https://doi.org/10.2147/CIA.S23404 [last accessed 11/05/22]
- ↑ Statland JM, Barohn RJ, Dimachkie MM, Floeter MK, Mitsumoto H. Primary Lateral Sclerosis. Neurologic clinics. 2015; 33(4), 749–760. https://doi.org/10.1016/j.ncl.2015.07.007 [last accessed 11/05/22]
- ↑ 19.0 19.1 Czarzasta J, Habich A, Siwek T, Czaplinski A, Maksymowicz W, Wojtkiewicz J. Stem cells for ALS: an overview of possible therapeutic approaches. Int J Dev Neurosci. 2017 Jan;57:46-55. doi: 10.1016/j.ijdevneu.2017.01.003
- ↑ Turner MR, Talbot K. Primary lateral sclerosis: diagnosis and management Practical Neurology 2020;20:262-269. https://doi.org/10.2147/CIA.S23404 [last accessed 11/05/22]