Kienbock's Disease
Original Editors - Fien Selderslaghs, Mirabella Smolders, Liese Magnus, Laura Van Der Perren and Jessica Worrell
Top Contributors - Jessica Worrell, Lucinda hampton, Admin, Laura Ritchie, Naomi O'Reilly, Shreya Pavaskar, WikiSysop, Fasuba Ayobami and Evan Thomas
Introduction[edit | edit source]
Kienböck disease refers to avascular necrosis of the lunate carpal bone, known as lunatomalacia (coined by Robert Kienbock to describe softening of the bone in the acute process). Kienbock's disease (KD) is defined by avascular necrosis of lunate with a predictable pattern of lunate collapse, carpal change, and degeneration, resulting from an apparent combination of vascular, anatomic and traumatic insults. (KD) was first described in 1983 by Peste through his cadaveric work and observations of lunate collapse[1]
Clinically Relevant Anatomy[edit | edit source]
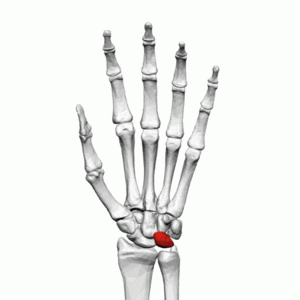
The proximal carpal row of the mid-carpal joint is largely responsible for wrist motion, as opposed to the relatively fixed distal carpal row. The lunate is the central bone in the proximal row, and it articulates with the scaphoid, capitate, triquetrum, and occasionally the hamate. More proximally, the lunate is a component of the radio-carpal joint, and it also articulates with the ulna via the triangular fibrocartilage complex (TFCC). Of note, nearly 10% of the axial-radial/ulnar/carpal load is transmitted through the TFCC and subsequently to the ulna, and 35% through the radio-lunate articulation[2]
Image 1: Lunate Bone Left Hand
Epidemiology[edit | edit source]
Kienböck disease is the second most common type of avascular necrosis of the carpal bones, preceded only by avascular necrosis of the scaphoid.[2]
The age distribution for Kienböck disease depends on gender.
- The condition is most common within the dominant wrist of young adult men where it appears to be due to repeated loading of the lunate.
- In women, Kienböck Disease typically occurs in middle age and is equally divided between the dominant and non-dominant wrist.[3]
Etiology[edit | edit source]
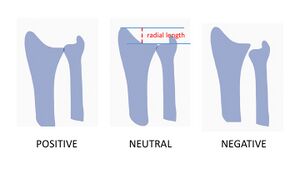
There is a significant association between negative ulnar variance and Kienbock disease. Ulna variance refers to a disproportionately shortened ulna when compared to the radius.[2]
Image 2: Relationship between radial length and ulnar variance. Radial length is the measure from distal ulna to radial styloid process. When ulnar variance is neutral radial length should be between 9-12mm.
A causal association is difficult to prove, however, the effectiveness of decompressive procedures such as radial shortening or ulnar lengthening in relieving pain and preventing further collapse of the lunate is supportive. Overall, the negative ulnar variance is present as a predisposing factor in around 75% of cases of Kienbock disease[3].
Trauma, negative ulnar variance, genetic factors are observed but there haven't been much studies to support it.
Characteristics/Clinical Presentation[edit | edit source]
Typical symptoms of Kienböck’s disease are activity-related dorsal wrist pain and weakness, often accompanied by swelling dorsally around the lunate. The complaints are in most cases longstanding and increase progressively. The pain can be described from just mild and occasional to severe and debilitating. The range of motion of the joint is nearly always decreased, with loss of flexion and extension. Compared to the unaffected side there is loss of grip strength.[4] Symptoms are most commonly in the dominant wrist and a history of trauma may be present.[5]
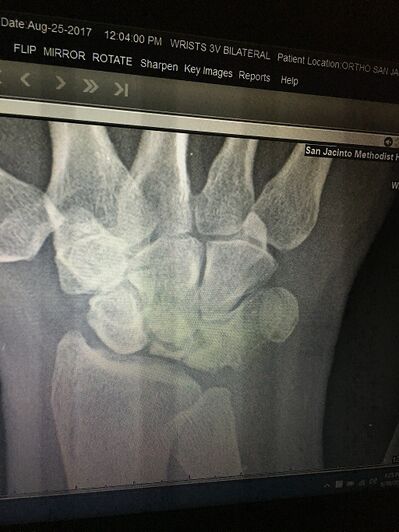
Image 3: X-ray on right wrist showing Kienböck's Disease. A collapsed lunate is present.
The length of the two bones of the forearm, in particular the ulna and the radius, is not usually equally. We can make a subdivision among the length between the distal articular surfaces of the radius and ulna, called the ulnar variance. It may be negative, positive or neutral. Negative ulnar variance indicates that the distal articular surface of the ulna is located more proximally than the radius, positive ulnar variance indicates the stage where the distal articular surface of the ulna is located more distally compared with the surface of the radius and at last neutral ulnar variance means that the ulnar and radial surfaces are at the same level.[6]
Staging of Kienböck disease is essential for treatment planning and divides into morphological staging.
- Stage 1: Normal radiograph, lunate signal intensity changes on MRI.
- Stage 2: Lunate sclerosis on radiography with or without fracture lines. The lunate shape is normal.
- Stage 3: Collapse of the lunate articular surface. 3A: with preserved carpal alignment and height. 3B: with scaphoid flexion and loss of carpal height. 3C: with an associated coronal fracture
- Stage 4: Stage 3B + radio-carpal or mid-carpal arthrosis[2].
Differential Diagnosis[edit | edit source]
Radiologic imaging, such as MRI and CT-scans, in case of Kienböck’s disease is generally the only way to give a correct diagnosis.[7]
- Lunate Bone Contusion/Acute Fracture: This type of lesion can hardly be differentiated from stage I Kienböck’s Disease. Having a history of acute episodes of severe trauma to the hand is probably the only characteristic of acute lunate bone fracture that can be separated from Kienböck’s.[8]
- Lunate Bone Subluxation
- Ulnocarpal Impaction Syndrome
- Bone contusion: May be difficult to distinguish early-stage Kienböck disease on MRI. A history of recent trauma and concomitant injuries to the wrist/hand, in this case, would aid in making the diagnosis
- Arthritis: Bone marrow signal changes in inflammatory or degenerative arthritides may mimic those seen in Kienböck disease. Distinguishing features include demographics, clinical presentation, and absence of ulna minus in arthritis[2]
Diagnostic Procedures[edit | edit source]
Kienböck disease is a clinical and imaging diagnosis. Both radiography/computed tomography and magnetic resonance imaging (MRI) are highly specific. However, MRI is the most sensitive and detects radiographically occult cases.
- Radiography
- CT-scan
- MRI: Is declared to be the best imaging technique for Kienböck’s. It is extremely sensitive and specific to detect osteonecrosis.[9] MRI is most significant in the early stages of the disease (particularly in stage I, when the plain radiographs show a rather normal outcome).[8][10]
Outcome Measures[edit | edit source]
- Range of motion[11][12][13]
- Disabilities of the Arm, Shoulder, and Hand (DASH) questionnaire[14][11][12]
- Grip Strength[11][12][13]
- Pain Scales (VAS/NPR)[11][12][13]
Examination[edit | edit source]
Typically some swelling is visible on the dorsal side of the wrist, along with synovitis.[15] The patient can experience a certain pain situated to the crucifixion fossa, which can be seen between the tendons of both the Extensor Digitorum and Extensor Carpi Radialis.[16] Furthermore, stiffness and tenderness may develop over the lunate bone.[15]
On physical inspection, the range of motion is quite sensitive, especially when extending the wrist. Movements such as flexion and extension are shrunken, sometimes combined with a progressively loss of grip strength.[16][15] However, the rotation of the forearm is still maintained.
Management[edit | edit source]
The goal of treating Kienböck disease is pain relief, wrist motion preservation, and preservation of grip strength. Treatment of Kienböck disease depends on the stage of the disease and its causative factors.
- Stage I is always treated with splinting or cast immobilization.
- Stage II can also be treated with immobilization if necrosis is incomplete.
- Stages II with complete necrosis, III, and IV require "joint-leveling" surgery possibly coupled with vascular bone grafting or transfer of branches from adjacent arteries.
- Later stages with lunate collapse and secondary wrist degenerative arthrosis may also require proximal row carpectomy or intercarpal arthrodesis.
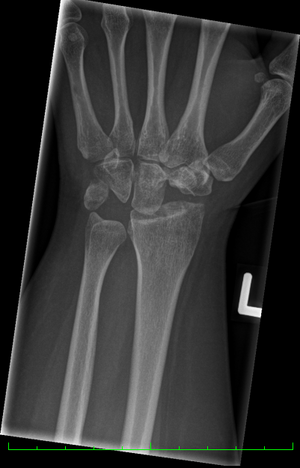
Image 5: Left Hand Post Proximal Row Carpectomy
Treatment may improve symptoms and functionality without affecting imaging findings in the advanced stages.[2]
Physical Therapy Management[edit | edit source]
Physical therapy is not a common treatment for this disease. It is clear that there are too few studies about physical management of this condition. Study findings
- Combined a bone marrow transfusion (medical), low-intensity pulsed ultrasound (PT) and an external fixation (surgery). Most of the patients in this study, wrist pain improved to a pain-free level, wrist flexion improved and average grip strength increased. This method can be used as a less invasive surgical alternative for Kienböck’s disease compared to surgical procedures.[17]
- A study compared different therapies after surgery. One group was treated according to a treatment protocol consisting of kinesiotherapy, electrotherapy, thermotherapy, massage therapy, therapeutic activities and domiciliary guiding. The other group was treated by non-specialized Departments in Hand Therapy and received only thermotherapy and exercises without a direct guidance from the therapist. After therapy, 90% of group one were satisfied with the results of the treatment and 66% of the second group. Also group 1 scored better on pain, muscular strength, prehension force, forearm articular range of motion for pronation and supination movements, wrist articular range of motion in abduction and adduction movements and manual function performance.[18]
- A study is a case study, reported on a 23 year old professional badminton player who had limited and painful wrist movements and was subsequently diagnosed with Kienböck’s disease. In this case study, the patient was treated surgically. For five weeks after the surgery, the wrist was splinted at all times and local anti-inflammatory and ice were used. Over the next eight weeks, stretching was increased gradually. Regular massage to the wrist extensor and flexor muscles was also done to decrease tone, which can affect joint function. At four months post-surgery, the wrist had painless 0-45° range of extension and 0-70° range of flexion. The patient started his regular training program with tape on his wrist for support. This is a case study thus the findings cannot be extrapolated to all patient populations.[19]
Prognosis[edit | edit source]
Kienbock disease is invariably progressive, and joint destruction occurs within 3-5 years of onset.
Prognosis depends on:
- Functional Staging: The greater the extent of viable bone, the better the prognosis.[6]
- Negative ulnar variance: The greater the negative variance, the more severe the disease and the more likely it is to progress.
- Age at diagnosis: Patients diagnosed at an older age are more likely to have advanced stage disease and are more likely to progress.
It is significant to note that the severity of symptoms does not always correlate with morphologic stage.[2]
References[edit | edit source]
- ↑ Al Bayati MA, Muttar AY, Al-Khishali TJ, Al Bayati MM, Hameed BM. The outcome of treatment of Kienbock's disease stage III by excisional arthroplasty and palmaris longus spacer.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Nasr LA, Koay J. Kienbock Disease.2019 Available:https://www.statpearls.com/articlelibrary/viewarticle/23892/ (accessed 1.10.21)
- ↑ 3.0 3.1 Radiopedia Kienböck disease Available:https://radiopaedia.org/articles/kienbock-disease-2 (accessed 1.10.2021)
- ↑ De Smet L et al., Treatment options in Kienböck’s disease, Acta Orthop. Belg., 2009, 75: 715-726.
- ↑ Watson HK, Guidera PM. Aetiology of Kienbock's disease. J Hand Surg [Br]. Feb 1997;22(1):5-7.
- ↑ Cerezal L et al., Imaging Findings in Ulnar-sided Wrist impaction Syndromes, RadioGraphics, 2002, 22(1): 105-121
- ↑ Allan CH et al., Kienböck’s Disease: Diagnosis and Treatment, Kienböck’s Disease, Journal of the American Academy of Orthopaedic Surgeons, 2001, 9(2): 128-136.
- ↑ 8.0 8.1 Arnaiz J et al., Imaging of Kienböck Disease, American Journal of Roentgenology, 2014, 203: 131-139.
- ↑ Watanabe K, Nakamura R, Imaeda T. Arthroscopic assessment of Kienbock's disease. Arthroscopy. Jun 1995;11(3):257-62.
- ↑ Schuind F et al., Kienböck’s disease, The Journal of Bone and Joint Surgery, 2008, 90-B: 133-139.
- ↑ 11.0 11.1 11.2 11.3 Stahl et al. Journal of Orthopaedic Surgery and Research (2015) 10:133 DOI 10.1186/s13018-015-0276-7
- ↑ 12.0 12.1 12.2 12.3 Ebrahimzadeh H. M, Moradi A, Vahedi E, Kachooei R A. Mid-term clinical outcome of radial shortening for kienbock disease. J Res Med Sci. 2015 February; 20(2): 146–149
- ↑ 13.0 13.1 13.2 Lindsay Innes, BS, Robert J. Strauch. Systematic Review of the Treatment of Kienböck’s Disease in Its Early and Late Stages. J Hand Surg 2010; 35A:713
- ↑ Martin, G. R., & Squire, D. (2013). Long-term outcomes for Kienböck’s disease. Hand (New York, N.Y.), 8(1), 23–26. http://doi.org/10.1007/s11552-012-9470-9
- ↑ 15.0 15.1 15.2 Lutsky K et al., Kienböck Disease, American Society for Surgery of the Hand, 2012, 37(A): 1942-1952.
- ↑ 16.0 16.1 Fontaine C et al., Kienböck's disease: la maladie de Kienböck, Chirurgie de la Main, 2015, 34(1): 4–17.
- ↑ Ogawa T et al., A new treatment strategy for Kienböck’s disease: combination of bone marrow transfusion, low-intensity pulsed ultrasound therapy, and external fixation, J Orthop Sci., 2013, 18(2): 230-237.
- ↑ Lima SMPF et al., Rehabilitation of patients with Kienböck disease underwent proximal row carpectomy, Acta Ortop. Bras., 2000;8(2): 83-89
- ↑ Karahan AY et al., Kienbock’s Disease That Manifested in a Badminton Player: Case Report, International Journal of Sports Science 2013, 3(4): 132-134.