Dupuytren’s Contracture
Original Editor - Gwen Fritsche and Lauren Leimbach from Temple University's Evidence Based Practice Project
Top Contributors - Lauren Leimbach, Gwen Fritsche, Admin, Kim Jackson, Lucinda hampton, Rachael Lowe, Chrysolite Jyothi Kommu, Kirenga Bamurange Liliane, Vidya Acharya, Laura Ritchie, Cindy John-Chu, Scott A Burns, WikiSysop, Fasuba Ayobami, Khloud Shreif, 127.0.0.1, Carina Therese Magtibay, Lennert De Henau and Evan Thomas
Introduction[edit | edit source]
Dupuytren contracture is a benign, progressive disease of the palmar fascia which results in shortening, thickening, and fibrosis of the fascia and aponeurosis of the palm driven by myofibroblastic proliferation.[1]
- Dupuytren disease is predominantly a myofibroblastic disease that affects the hand/fingers and results in contracture deformities.
- Commonly affected digits are those farthest from the thumb i.e. the fourth (ring) and fifth (pinky) digits.
- The disease begins in the palm as painless nodules that form along longitudinal lines of tension.
- The nodules form cords that produce contracture deformities within fascial bands and tissues of the hand.
- Dupuytren contracture is usually seen in Caucasians and the disorder often presents bilaterally. When unilateral, the right side is more likely to be involved compared to the left.
- In many individuals, there is a family history with males being more susceptible to the condition than females.[2][3]
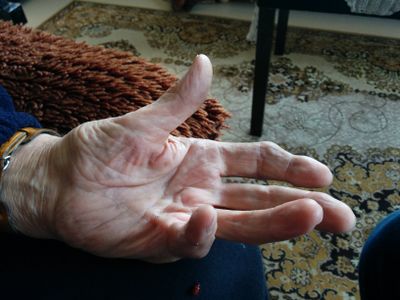
Image 1: Clinical presentation of Dupuytren contracture[4]
Etiology[edit | edit source]
Dupuytren disease is a genetic disorder expressed in an autosomal dominant fashion, but most frequently seen with a multifactorial etiology. It is associated with diabetes, seizure disorders, smoking, alcoholism, HIV, and vascular disease[5].
Ectopic manifestations beyond the hand can be seen in Plantar Fibromatosis (plantar fascia), 10% to 30%; Peyronie disease (Dartos fascia of the penis), 2% to 8%; and Garrod disease (dorsal knuckle pads), 40% to 50%.[5]
Epidemiology[edit | edit source]
This condition is common in populations of Northern European/Scandinavian descent. It is relatively uncommon in Southern European and South American populations and is rare in Africans and Asians. The disease affects men more severely than women. Males are affected by a 2:1 ratio compared to women. Younger age of onset is also associated with increased severity of disease progression. In Asian populations, the palm is more likely to be involved than the digits and thus often goes unnoticed.[5]
Pathological Process[edit | edit source]
The pathophysiology of Dupuytren disease involves abnormal myofibroblastic growth in the hand.
- Type III collagen predominates, which under a non-disease state would be Type I collagen.
- Dupuytren contracture progresses through three phases: (1) proliferative, (2) involution, and (3) residual. The proliferative phase has a characteristically high concentration of immature myofibroblasts and fibroblasts arranged in a whorled pattern. In the involution phase, fibroblasts become aligned in the longitudinal axis of the hand following lines of tension. In the residual phase, relatively acellular collagen-rich chords remain causing contracture deformity.
- The disorder is not always progressive and in at least 50-70% of patients, it may stabilize or even regress.
Several cords can develop which can cause unique deformities of the hand.
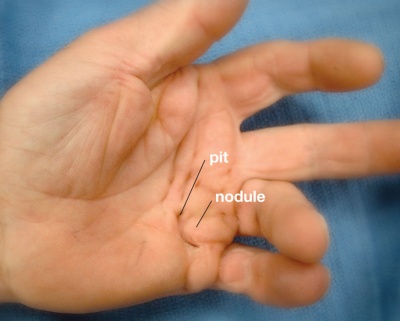
- Pretendinous cords cause skin pitting and metacarpal phalangeal (MCP) joint contracture.
- Natatory cords are responsible for webspace contractures.
- Spiral cords are the most important in the disease process and can cause proximal interphalangeal (PIP) contracture.
Risk factors for increased severity and recurrence of disease after treatment include- male gender; onset before age 50; bilateral disease; sibling/parent involvement; the presence of Garrod pads, Ledderhose, or Peyronies diseases.[2]
Clinical Presentation[edit | edit source]
Dupuytren contracture occurs slowly and typically progresses over the course of several years, but can also develop more rapidly over weeks or months.[6]
It typically affects older men of European descent. This condition most commonly begins with thickening of the skin on the palm, resulting in a puckering or dimpled appearance. As the condition progresses, bands of fibrotic tissue form in the palmar area and may travel distally toward the fingers. This tightening and shortening eventually lead to the affected fingers being pulled into flexion. Dupuytren contracture typically occurs bilaterally, with one hand being more severely affected than the other.
Physical findings:
- Blanching of the skin when the finger is extended
- Proximal to the nodules, the cords are painless
- Pits and grooves may be present
- The knuckle pads over the PIP joints may be tender
- If the plantar fascia is involved, this indicates a more severe disease (Plantar Fibromatosis)
- The patient may not be able to place the palm flat on the table[2]
Diagnostic Procedures[edit | edit source]
- X-rays of the hand should be obtained to examine for other contributing, bony abnormalities that may contribute to the loss of range of motion.
- Laboratory workup to rule out diabetes is recommended.
- Ultrasound may demonstrate thickened palmar fascia and the nodules.[2]
Differential Diagnosis[edit | edit source]
Dupuytren disease should be distinguished from other diseases of the hand including stenosing flexor tenosynovitis, ganglion cysts, and soft tissue masses[2].
Outcome Measures[edit | edit source]
- Range of motion measurements of the metacarpophalangeal (MCP), proximal interphalangeal (PIP), and distal interphalangeal (DIP) joints should be recorded ( flexion and extension of these joints, with measurements of passive and active range of motion). Take as a baseline measure and then throughout the treatment process, can help stage the severity of the contractures.
- Measure hand function by tests and measures such as eg Disabilities of the Arm, Shoulder, and Hand Questionnaire (The DASH), or its shorter version The Quick Dash.
Medical Management[edit | edit source]
Indications for treatment are based on the effects of disease on the patient's quality of life. Many patients with a positive tabletop test, MCP contracture of 30 degrees, or PIP contracture of 15 to 20 degrees will elect to have treatment.
Treatment options consist of observation, needle aponeurotomy, collagenase injection, and/or surgical resection and fasciectomy.
Observation is appropriate for individuals with painless stable disease and no impairment in function. Follow up every 6 months may be done to assess the progression of the disorder.
- Physical and occupational therapy including ultrasound waves and heat can help during the early stage of the disease. Some patients may also benefit from a brace/splint to stretch the digits. The range of motion of the fingers is necessary to prevent adhesions.
- Corticosteroid injections may be beneficial for some patients eg those with painful nodules. Steroid injections do not work in all patients and a 50% recurrence has been reported. Corticosteroid injections can lead to fat atrophy, pigmentation change and there is the potential to cause rupture of the tendons.
- Other treatments that have been tried include-tamoxifen; anti-tumor necrosis factor agents; 5 fluorouracil, imiquimod; botulinum toxin. No evidence exists to say any of these treatments are superior or work for everyone.
- Radiation therapy may be beneficial during the early phase of the disease but has associated complications. It aims to slow down or halt the abnormal cell growth, being particularly effective when finger bending is minimal. A review of several studies indicated varied responses; some patients saw no change or improvement, while others experienced disease progression[7]. A long-term study revealed many patients remained stable or even improved, with minor late side effects like skin atrophy and dry skin being observed in some patients, yet no increased cancer risk was noted[8]. However, more research is needed to fully grasp the treatment's benefits and risks[9].
- Needle aponeurotomy is typically reserved for mild contractures. The procedure is minimally invasive and is often performed in an office setting.
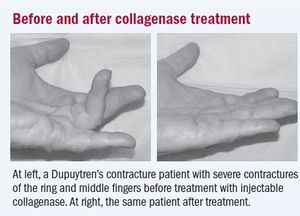
- Collagenase injections provide a minimally invasive treatment derived from Clostridium histolyticum. Night extension splinting is maintained for 6 months. Collagenase injections result in a 75% contracture reduction with a 35% recurrence rate. Complications include edema, skin tearing, tendon rupture, complex regional pain syndrome, and pulley rupture. Before and after collagenase treatment image at R [10]
- Surgical fasciectomy can be either limited or radical. The recurrence rate at 1 to 2 years is 30%, 15% at 3 to 5 years, and less than 10% after ten years.
- Total palmar fasciectomy can also be performed but is infrequently used as it requires resection of all palmar and digital fascia, including nondiseased tissue.
- Complications of fasciectomy include skin necrosis, hematoma (most common complication), flare reaction, neurovascular injury, digital ischemia, swelling, and infection.
Irrespective of the treatment, recurrence is common with all of them, approaching 20-50% at 5 years.[2]
Physical Therapy Management[edit | edit source]
Conservative Approach[edit | edit source]
Physical therapy may include ultrasound waves: heat (early stage of the disease); brace/splint to stretch the digits; a range of motion of the fingers to prevent adhesions.
Postoperative Care/Rehabilitation[edit | edit source]
Patients often enter hand therapy to :
- Maintain the range of motion of the hand and fingers is important (for many activities of daily living), see hand exercises
- Extension splints often are used in conjunction with other modalities.
- Odema and scar interventions.[11]
- Should be undertaken for at least 3 months to prevent contractures.
- Maximal benefits of surgery are not immediate, only become obvious after 6-8 weeks.[2]
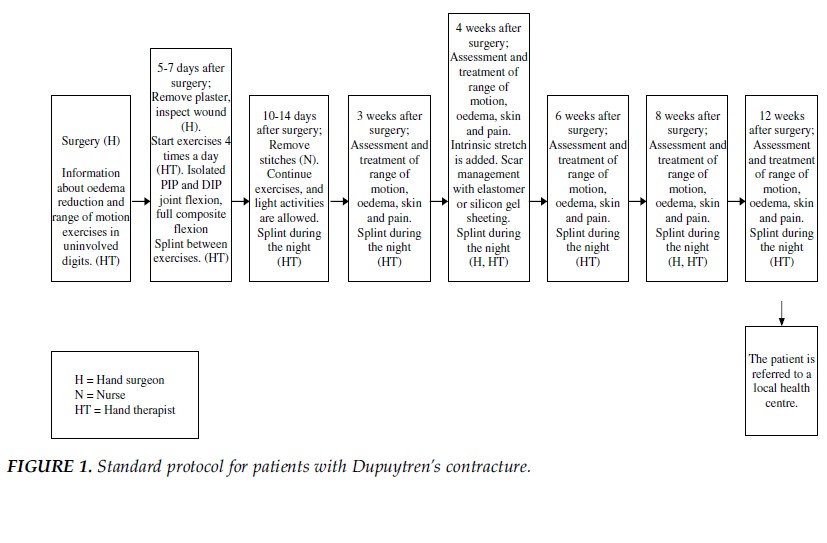
A standard protocol for postoperative management of Dupuytren disease is shown below (Engstrand et al. in 2009).[11]
- Within the initial 5 days postoperative, the primary interventions are to educate the patient on decreasing edema and the importance of performing a range of motion exercises on the uninvolved fingers.
- After 5-7 days postoperative, the primary interventions shift to a range of motion exercises and splinting.
- The exercises are adapted to each subject’s individual goals and are based on their impairment, physical status, and competency.
- The types of splints used included volar splints, dynamic extension splint, dynamic flexion splints, exercise splints, and wrist splints.
The video below gives a good summary of the condition and physiotherapy treatment (less than 4 minutes)
Conclusion[edit | edit source]
The key fact to appreciate is that not all patients need treatment.
- There are many treatments available for Dupuytren contracture and none is ideal or works consistently.
- Only symptomatic patients should be offered treatment because all treatments have complications.
- The patient must be educated about the potential complications of treatments, which are worse than the disorder itself.
- Close communication between the team is essential in order to improve outcomes.
- Overall, only a few patients achieve a desirable result.
- In many cases, prolonged physical therapy is required to restore functionality[2]
References[edit | edit source]
- ↑ E Soreide, M H Murad, J M Denbeigh, E A Lewallen, A Dudakovic, L Nordsletten, A J van Wijnen, S Kakar.Treatment of Dupuytren's contracture: a systematic review.PubMed.gov.National Library of Medicine. National Centre for Biotechnology Information.2018 Sep;100-B(9):1138-1145.doi: 10.1302/0301-620X.100B9.BJJ-2017-1194.R2.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Walthall J, Rehman UH. Dupuytrens Contracture. InStatPearls [Internet] 2019 Feb 19. StatPearls Publishing.Available from:https://www.ncbi.nlm.nih.gov/books/NBK526074/ (last accessed 4.4.2020)
- ↑ Bayat A. A nonsurgical therapy for Dupuytren disease. Rheumatology. 2010;6:7-8.
- ↑ Dupuytren’s Disease. American Society for Surgery of the Hand Web site. http://www.assh.org/Public/HandConditions/Pages/DupuytrensDisease.aspx. 2010. Accessed March 19, 2011.
- ↑ 5.0 5.1 5.2 Walthall J, Anand P, Rehman UH. Dupuytren Contracture. StatPearls [Internet]. 2020 Sep 14.
- ↑ Mayo Foundation for Education and Research. The Dupuytren's Contracture Page. http://www.mayoclinic.com/health/dupuytrens-contracture/DS00732. Updated May 15, 2010. Accessed March 14, 2011.
- ↑ Kadhum M, Smock E, Khan A, Fleming A. Radiotherapy in Dupuytren’s disease: a systematic review of the evidence. Journal of Hand Surgery (European Volume). 2017 Sep;42(7):689-92.
- ↑ Betz N, Ott OJ, Adamietz B, Sauer R, Fietkau R, Keilholz L. Radiotherapy in early-stage Dupuytren's contracture. Strahlentherapie und Onkologie. 2010 Feb 1;186(2):82.
- ↑ Solie P, Stump B, Cashner C, Lenards N, Hunzeker A, Zeiler S. A case study of radiotherapy treatment for Dupuytren's contracture and Ledderhose disease. Medical Dosimetry. 2023 Apr 12.
- ↑ Harvard University. Nonsurgical approach unlocks contracted fingers. Harvard Women’s Health Watch. 2009:6-7.
- ↑ 11.0 11.1 Engstrand C, Boren L, Liedberg GM. Evaluation of activity limitation and digital extension in Dupuytren’s contracture three months after fasciectomy and hand therapy interventions. J Hand Ther. 2009;22:21-27.
- ↑ Physio vibes DUPUYTREN'S CONTRACTURE & PHYSIOTHERAPY MANAGEMENT Available from: https://www.youtube.com/watch?v=a8KMCAFx8xw (last accessed 5.4.2020)