William's Syndrome
Original Editors - Julie Frederick from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Julie Frederick, Kim Jackson, Admin, 127.0.0.1, Elaine Lonnemann, Wendy Walker, WikiSysop, Shaimaa Eldib and Lucinda hampton
Definition/Description[edit | edit source]
William’s Syndrome was first recognized as a unique disorder in 1961.[1] J.C.P. Williams observed in four patients an association between supravalvular aortic stenosis and the common physical and mental characteristics of this patient population and stated that it “may constitute a previously unrecognized syndrome”[1]. Later, A.J. Beuren described eleven new patients with the characteristics described by Williams and the disorder became known as Williams-Beuren Syndrome.[1] Diagnosis of the syndrome can be made at birth based on physical characteristics, but a true medical diagnosis is confirmed following a diagnostic test called fluorescence in situ hybridization (FISH).[1] The test reveals a recurring micro-deletion, with a size of 1,551,83 Mb, on chromosome band 7q11.23, which contains 24-28 genes.[2][3][4] The deleted part of the chromosome band includes the elastin gene, which leads to serious cardiovascular complications.[5] Elastin is a protein responsible for tissue elasticity.
Other common names for William's Syndrome[6]:
- Williams-Beuren Syndrome
- Beuren Syndrome
- Elfin Facies Syndrome
- Elfin Facies with hypercalcemia
- Hypercalcemia-Supravalvar Aortic Stenosis
- Infantile hypercalcemia
- Supravalvar aortic stenosis syndrome
- WBS
- WMS
- WS
Prevalence[edit | edit source]
Stated as being anywhere from 1/7,500 to 1/50,000.[1][2][7][8][9] William’s Syndrome occurs sporadically and spontaneously and is found equally in all ethnicities, races, socioeconomic backgrounds and genders.[1][5][8]
Characteristics/Clinical Presentation[9][edit | edit source]
The above video was put together as a slide show presentation by Boston University School of Medicine and can be accessed at the following web address: http://www.youtube.com/watch?v=Dgfm8iWrY_4 . The video displays children with William's Syndrome and the common facial characteristics distinct to the genetic disorder.
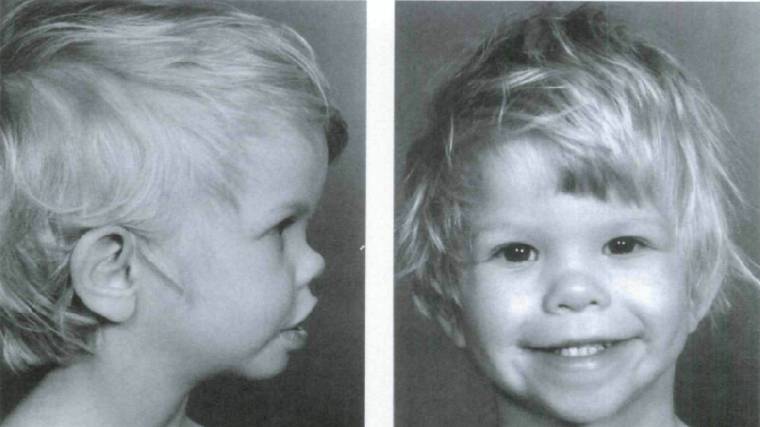
The above two pictures are from the following souce: Kaplan P, Wang P, Francke U. Williams (Williams Beuren) syndrome: a distinct neurobehavioral disorder. Journal Of Child Neurology. (2001, Mar); 16(3): 177-190.
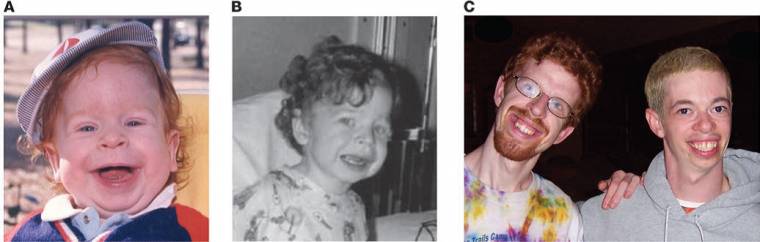
The above series of 3 pictures A, B, C was from the following journal article: Pober B, Johnson M, Urban Z. Mechanisms and treatment of cardiovascular disease in Williams-Beuren syndrome. Journal of Clinical Investigation. (2008, May); 118(5): 1606-1615.
- Craniofacial dysmorphic features[1][2][3][4][5][8]
- Full lips[4][8], short nasal bridge[4], large forehead[4], long philthrum[8], epicanthal folds[8], hypertelorism[8], mandibular hypoplasia[8]
- Mild to moderate mental retardation[1][2][3][4][5][8]
- Average IQ = 55-60 but can range from 40-90[1]
- Mild to moderate learning disabilies2[2]
- Associated systemic disorders (especially cardiovascular)[1][2][3][4][5][7][8]
- Mild growth retardation[1][3], short stature[5][8]
- Commonly overweight as adults[1]
- Deficient visuo-spatial abilities[3][4][5]
- Global processing deficits[4]
- Overfriendly personalities[2][3][5]
- Very sociable[4]
- Empathetic[10]
- High anxiety[10]
- Attention deficit disorder[9]
- Hyperacusis or algiacusis[3]
- White star shape in the iris of the eye[11]
- Increase prevalence of left hand and eye dominance (especially in males)[12]
- Interest and connection with music and sound[11]
Characteristics commonly seen during Infancy[9]:
- Often born post-term
- Small for the family background
- Feeding difficulties - Gastro-esophageal (G-E) reflux, disordered suck and swallow, textural aversion, vomiting
- Failure to thrive
- Prolonged colic (>4 months)
- Chronic constipation
- Idiopathic hypercalcemia
- Strabismus
- Chronic otitis media
- Rectal prolapse
- Inguinal hernia
- Hypotonicity
- Hyperextensible joints
- Delayed motor milestones
Systemic Involvement[1][9][edit | edit source]
Cardiovascular pathologies[2][3][4][5][7]
• Supravalvular aortic stenosis[1][2][4][5][7][9]
• Ventricular septal defect[7]
• Patent ductus arteriosus[7]
• Stenosis of outlying arteries (renal, cerebral, carotid, coronary, brachiocephalic, subclavian, mesenteric, lung)[7]
• Stenosis of the abdominal aorta[14]
• Arteriovenous shunt[7]
• Interruption of the aortic arch[7]
• Vein aplasia[7]
• Aortic aneurysm[7]
• Pulmonary arterial stenosis[5]
• Left and right ventricular hypertrophy[5]
• Aortic coarctation[5]
• Mitral valve prolapse[5]
• Aortic valvular insufficiency[5]
Endocrine abnormalities[5]
• Hypercalcemia[4][7]
• Abnormal glucose metabolism
• Thyroid hypoplasia[8]
• Hypothyroidism[8]
Dental anomalies
• Small, abnormally shaped teeth
• Absent teeth
• Malocclusion
Gastrointestinal problems
• Dysmotility
• Reflux
• Constipation
• Diverticular disease
Musculoskeletal anomalies
• Joint laxity[9]
• Joint stiffness as patient ages[13]
• Tight heel cords[15]
• Scoliosis
• Cervical kyphosis[15]
• Exaggerated lumbar lordosis[15]
• Radioulnar synestosis[15]
• Gait - wide BOS, toe-walk, forward posture[15]
Senses
• Sensorineural hearing loss
• Farsightedness[13]
• Hyperopia[9]
• Strabismus[9]
• Otitis media[9]
Genitourinay anomalies[5]
• Urinary frequency
• Bladder diverticuli
Neurological problems
• Abnormal tone
• Hyperreflexia
• Cerebellar dysfunction
• Developmental delays[16]
• Learning disabilities[16]
• Seizures[16]
Medications[edit | edit source]
There are no medications for William’s Syndrome itself, however, common medications for systemic complications may include: thyroxine for hypothyroidism and thyroid hypoplasia[8], medication for attention deficit disorder[9], medication for hypertension[9], oral corticosteroids and/or intravenous pamidronate for hypercalcemia[9].
IMPORTANT NOTE: Children with William's Syndrome should not be given a daily pediatric multivitamin because of the dose of vitamin D contained in the vitamin. This can lead to hypercalcemia.[9]
Diagnostic Tests/Lab Tests/Lab Values[edit | edit source]
The most common test used to diagnose William’s Syndrome is fluorescence in situ hybridization (FISH)[1], but targeted mutation analysis can also be used. Targeted mutation analysis may include any of the following tests: Real-time quantitative PCR, Genomic microarray analysis, and Heterozygosity testing.
Other tests to diagnose systemic complications include: electrocardiogram, echocardiogram, aortography[14], ultrasonography[8], Tc-pertechnetate thyroid scintigraphy[8], thyroid function tests[8], MRI[9], polysomnography[9]
There is also prenatal testing available for couples with a family history of Williams syndrome who may want to conceive.[13]
Etiology/Causes[edit | edit source]
William's Syndrome is a result of a spontaneous microdeletion of a band on chromosome 7.[1] The deletion can be inherited from either the mother or father of a person with William's Syndrome and a person with William's Syndrome has a 50% chance of having a child with the genetic disorder.[7][13] However, the disorder mostly occurs spontaneously and equally amongst all races, ethnicities, socioeconomic backgrounds and genders. With William's syndrome penetrance is 100%, thu<span id="fck_dom_range_temp_1302733639308_887" />s 100% of people with the gene deletion will have William's syndrome.[9] However, expression of the phenotypic features is variable, thus not every person with William's syndrome will have all of the common characteristics typically seen in those with the disorder.[9]
Associated Co-morbidities[edit | edit source]
- Supravalvular aortic stenosis
- Hypercalcemia
Medical Management[9][edit | edit source]
- Early intervention programs
- Special education programs
- Vocational training to address developmental disabilities
- Speech/language therapy
- Physical therapy
- Occupational therapy
- Sensory integration therapy
- Psychologic evaluation
- Psychiatric evaluation
- Behavioral counseling - often used to manage behavior problems, especially attention deficit disorder and anxiety
- Psychotropic medication - often used to manage behavior problems, especially attention deficit disorder and anxiety
- Surgery may be required for supravalvular aortic stenosis (required in 30% of children with the disorder[9]), mitral valve insufficiency, or renal artery stenosis
- Treatment of hypercalcemia - diet modification, oral corticosteroids, and/or intravenous pamidronate
- Referral to a nephrologist - for management of nephrocalcinosis, persistent hypercalcemia and/or hypercalcuria
- Infants may benefit from feeding therapy
- Corrective lenses - hyperopia
- Patching one eye of surgery - strabismus
- Tympanotomy tubes - otitis media
- Dental cleanings every 3 months
Yearly Check-ups for children with William's syndrome should include the following:
- Medical evaluation
- Vision screening
- Measurement of blood pressure
- Calculation of calcium/creatine ratio in a random spot urine
- Urinalysis
Periodic evaluations for children with William's syndrome should include:
- Serum concentration of calcium
- Thyroid function
- Hearing
- Renal and bladder ultrasound
Periodic evaluations for adults with William's syndrome should include:
- Glucose tolerance
- Cardiac evaluation - mitral valve prolapse, aortic insufficiency, arterial stenosis and other associated cardiac pathologies
- Ophthalmologic evaluation - cataracts
| Interval/Age | Test/Measurement |
|
Annually |
•Medical evaluation •Vision screening to monitor for refractive errors and strabismus •Monitoring of blood pressure in both arms •Measurement of calcium/creatine ratio in a random spot urine and urinalysis |
| Every 2 years | •Serum concentration of calcium |
| Every 3 years | •Thyroid function and TSH level |
| Every 5 years | •Audiologic examination |
| Every 10 years | •Renal and bladder ultrasound examination |
| In adults |
•Oral glucose tolerance test (OGTT) starting at age 30 years to evaluate for diabetes mellitus •Evaluation for mitral valve prolapse, aortic insufficiency, and arterial stenoses •Evaluation for cataracts
|
Physical Therapy Management[edit | edit source]
Dr. Colleen A. Morris suggested adding exercise and joint ROM into the treatment plan for children and adults with William's syndrome in order to help maintain proper blood glucose levels to prevent insulin resistance or the development of diabetes and prevent contractures.[9]
A case study that examined the motor development of children with William's syndrome suggests that due to the deficits found, "early evaluation of motor development, strengthening of motor performance and upper limb dexterity may be vitally important during pre-school and school age."[17]
Source: Sen-Wei T, Shyi-Kuen W, Ying-Ming L, San-Ging S. Early development in Williams syndrome. Pediatrics International. (2008, Apr); 50(2): 221-224.
At Combine Sections Meeting (CSM) 2011, The Children's Hospital of Philadelphia gave a presentation on connective tissue disorders and how to treat them with both land-based and water-based exercise programs. William's syndrome was one of the disorders included in this presentation. The following information about physical therapy treatment for children with William's syndrome was taken from the aforementioned presentation given by Michael Dilenno, PT, DPT, CSCS and Abby Leibrand, PT, DPT.[18]
Typical areas of weakness found:
- Trunk/core
- Shoulder
- Hand
- Hip
- Foot and ankle
Therapeutic Exercises used to treat the above weaknesses include:
- Strengthening
- Stretching
- Neuromuscular Re-education
- Neuromuscular Stabilitization
- Functional Training (self-care and home management)
- Functional Training in job/school/play
- Devices and equipment
The following muscles or muscle groups were targeted during strengthening treatment sessions:
- Abdominal exercise
–Transverse Abdominus
–Obliques
–Lower Abdominals
- Back
–Extensors
–Latissimus dorsi
- Shoulder girdle and attachments
–Middle Trapezius
–Lower Trapezius
–Rhomboids
–Serratus Anterior
- Muscles of or influencing the elbow, hand, hip, knee, ankle and foot
Examples of methods for treating weaknesses include using the following:
- For infants, toddlers, children
–Physioball activities for trunk and core
–Increase resistance with t-band, weight, objects
- For older children, adolescents, young adults
–Physioball
–Resistance bands
–Free weights
–Machines
Support Group Music Camp[edit | edit source]
Many individuals have great talent in music so places offer summer music camps for individuals with William's Syndrome to attend and utilize their strength in music to help alleviate any weaknesses, such as anxiety or fear of what others think about them.[11] Vanderbilt Kennedy Center of Vanderbilt University in Nashville, TN holds a summer camp every year called ACM Lifting Lives Music Camp, where participants, who have William's Syndrome or other developmental disabilities and are at least 16 years of age, reside at the camp for a week. Participants of the camp "celebrate music by participating in a songwriting workshop, recording session, songwriter's night and a live performance on the stage of the Grand Ole Opry."[11] They are also invited to partipate in research that is being conducted about developmental disabilities. Goals of the camp include[11]:
- Learning and practicing new skills
- Music enrichment and appreciation
- Performing with other musicians
- Socializing with mentors and peers
- Feeling connected and empowered
- Family support and information
The camp is sponsored by American Country Music and many country singers volunteer and share their experiences with campers. Below is a picture of an up-and-coming country star, Chris Young, with a camper who attended the summer camp in 2010.
For more information about the camp, visit the following website: http://kc.vanderbilt.edu/site/services/page.aspx?id=11
Differential Diagnosis[edit | edit source]
Dr. Colleen A. Morris stated that William's Syndrome "should be distinguished from other syndromes that include developmental delay, short stature, distinctive facies, and congenital heart disease."[9]
Syndromes that should be distinguished from William's Syndrome include[9]:
- Noonan Syndrome
- Deletion 22q11 (DiGeorge syndrome)
- Smith-Magenis syndrome
- Kabuki syndrome
- Fetal Alcohol Syndrome (FAS)
Dr. Colleen A. Morris also stated that "individuals with SVAS (supravalvular aortic stenosis) should be evaluated to determine if William's Syndrome or autosomal dominant SVAS is the appropriate diagnosis"[9]
Case Reports/ Case Studies[edit | edit source]
add links to case studies here (case studies should be added on new pages using the case study template)
- Cerebral Infarction in Williams-Beuren Syndrome: Kalbhenn T, Neumann L, Lanksch W, Haberl H. Spontaneous Intracerebral Hemorrhage and Multiple Infarction in Williams-Beuren Syndrome. Pediatric Neurosurgery. (2003, Dec); 39(6): 335-338.
- Left Hand and Eye Dominance in Williams-Beuren Syndrome: Van Strien J, Lagers-Van Haselen G, Van Hagen J, De Coo I, Frens M, Van Der Geest J. Increased Prevalences of Left-handedness and Left-eye Sighting Dominance in Individuals with Williams-Beuren Syndrome. Journal of Clinical & Experimental Neuropsychology. (2005, Nov); 27(8): 967-976.
- Motor Development in Children with William's Syndrome: Sen-Wei T, Shyi-Kuen W, Ying-Ming L, San-Ging S. Early development in Williams syndrome. Pediatrics International. (2008, Apr); 50(2): 221-224.
The following case study is made up from the documentation and treatment of a child with William's Syndrome. Consent was given by the child's mother and the therapist treating the child.
- Names have been changed to protect the privacy of the therapist and child.
Title: Child with William's Syndrome seen in OP Pediatric Setting
Author: Student Physical Therapist: Julie Frederick
Patient Characteristics
• Date of Initial Evaluation: 07/14/10
• Demographic Information: 3 year old female
• Medical diagnosis: William's Syndrome (diagnosed 03/11/10)
• Co-morbidities: None stated by mother
• Previous care or treatment: Received physical therapy through the state, but discontinued by child's mother in March 2009 due to poor communication with the therapist.
Examination
Subjective: Child’s mother reported that she wants Elizabeth* to be able to climb stairs safely and "just be where she needs to be" in comparison with her peers. She states concerns about Elizabeth’s flat feet. She also mentioned that Elizabeth loves music.
Objective:
• Posture:
Sitting: Unremarkable.
Standing: Moderate pronation bilaterally.
• Tone: Child presents with fluctuating tone. At rest child presents with gross hypotonia; however, during tasks that require increased physical exertion child with ‘spastic’ like tendancies and lack of controlled movement patterns.
• ROM: No restrictions noted.
• Strength: Unable to formally assess child’s strength due to her age; however, functionally child demonstrates at least 3/5 or greater throughout her UEs and 4/5 throughout her LEs.
• Sensation: Unable to formally assess child’s sensation due to her age; however, child responded appropriately to examiner’s light touch throughout all four extremities and trunk.
• Gross Motor Skills:
Gait: Child displayed wide BOS, unequal step lengths and high guard in her UEs.
Stair Management: Child overall unsteady throughout stair management. She reciprocally ascends stairs with use of 1 hand rail and 1 hand-held assist with significant posterior lean. Non-reciprocal descent again with 1 hand rail and 1 hand- held assist.
Running: Child displayed wide BOS, flailing UEs and bilateral foot slap throughout running activities.
Jumping: Child able to jump in place with bilateral take off. At the time of the evaluation child unable to advance jump or jump off 6” step despite multiple verbal, visual and tactile cues. Instead of jumping child steps off step with 1 hand-held assist.
• Balance:
SLS: Child able to briefly stand on one foot when her hands are held; however, child immediately places foot on floor when external support is absent.
• Visual Tracking: Child with poor visual attention during evaluation and thus unable to assess visual tracking at this time.
• Cognition: Child was alert and responded to her name throughout the evaluation.
• Behavior: Child was pleasant and warmed to examiner quickly. Child displayed decreased attention for therapeutic activities; however, was easily motivated with toys and music.
Assessment: Child is a 3 year old female referred to physical therapy due to her gross motor delays. Child’s PMH includes hernia repair, frequent ear infections, tube placement and recent diagnosis of William’s Syndrome. Child presents with significant delays in her locomotor and object manipulation skills as measured by the PDMS-2. These delays due to decreased dynamic standing balance, reciprocal bilateral coordination and decreased overall strength. Other contributing factors include decreased attention and possible visual deficits. Child’s sensation to light touch and AROM tested WNL throughout all four extremities and trunk. Child will benefit from physical therapy to address the aforementioned deficits and to provide caregiver education to further therapeutic benefit outside of scheduled therapy sessions.
Intervention
Frequency/duration: 1 x week (60 minutes) x 6 months
Therapeutic Procedures, Neuromuscular Reeducation, Gait Training, Therapeutic Activities, Orthotic Management
• Balance/coordination: balance beam, SLS
• Stair management
• Jumping: jump off 6" step
Outcomes
At 6 month re-evaluation on 01/19/11: Child met 1/6 goals set and is steadily progressing with the others. She showed improvement since her initial evaluation in the following activities: ability to jump forward and in place on level ground and off small objects, obtain SLS without UE assistance (although unable to maintain at this time) and ascend/descend steps without use of hand rail.
Discussion
Although Elizabeth only met 1/6 goals set by her therapist, she still showed great improvement in all areas when compared to her initial evaluation. Gains were noted when comparing her PDMS-2 scores. The scores of her initial and 6 month scores are below:
| Subtest | Raw Score | Age Equivalence | Percentile | Standard Scores |
| Stationary | 40 | 28 months | 25 | 8 |
| Locomotion | 94 | 20 months | 2 | 4 |
| Object Manipulation | 14 | 20 months | 5 | 5 |
| GMQ | |
| Quotients | 72 |
| Percentiles | 3 |
| Subtests | Raw Score | Age Equivalence | Percentile | Standard Score |
| Stationary | 45 | 40 months | 50 | 10 |
| Locomotion | 105 | 23 months | 5 | 5 |
| Object Manipulation | 23 | 27 months | 7 | 7 |
| GMQ | |
| Quotients | 83 |
| Percentiles | 13 |
Resources
[edit | edit source]
• Williams Syndrome Association: www.williams-syndrome.org
• Williams Syndrome Foundation: http://www.wsf.org
• Vanderbilt Kennedy Center: http://kc.vanderbilt.edu/site/about/default.aspx
• Genetics Home Reference: http://ghr.nlm.nih.gov/condition/williams-syndrome
• Information provided by an expert on the disorder, Dr. Colleen A Morris: http://www.ncbi.nlm.nih.gov/books/NBK1249/
• Gene Test Locations: http://www.ncbi.nlm.nih.gov/sites/GeneTests/lab/clinical_disease_id/2527
References[edit | edit source]
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 Pober B, Johnson M, Urban Z. Mechanisms and treatment of cardiovascular disease in Williams-Beuren syndrome. Journal of Clinical Investigation. (2008, May); 118(5): 1606-1615.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 John A, Mervis C. Comprehension of the Communicative Intent Behind Pointing and Gazing Gestures by Young Children with Williams Syndrome or Down Syndrome. Journal of Speech, Language & Hearing Research. (2010, Aug); 53(4): 950-960.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Lucena J, Pezzi S, Aso E, Valero M, Carreiro C, Campuzano V, et al. Essential role of the N-terminal region of TFII-I in viability and behavior. BMC Medical Genetics. (2010, Apr 19); 1161.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 Collette J, Xiao-Ning C, Mills D, Galaburda A, Reiss A, Korenberg J, et al. William's syndrome: gene expression is related to parental origin and regional coordinate control. Journal of Human Genetics. (2009, Apr); 54(4): 193-198.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 Del Pasqua A, Rinelli G, Toscano A, Iacobelli R, Digilio C, de Zorzi A, et al. New Findings concerning Cardiovascular Manifestations emerging from Long-term Follow-up of 150 patients with the Williams-Beuren-Beuren syndrome. Cardiology in the Young. (2009, Dec); 19(6): 563-567.
- ↑ U.S. National Library of Medicine. Genetics Home Reference. Bethesda, MD: U.S. National Library of Medicine; [updated 2011 April 3; cited 2011 April 5]. Available from: http://ghr.nlm.nih.gov
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 Figueroa J, Olivares Rodríguez L, Pablos Hach J, Ruíz V, Martínez H. Cardiovascular Spectrum in Williams-Beuren Syndrome. Texas Heart Institute Journal. (2008, Sep); 35(3): 279-285.
- ↑ 8.00 8.01 8.02 8.03 8.04 8.05 8.06 8.07 8.08 8.09 8.10 8.11 8.12 8.13 8.14 8.15 8.16 Stagi, Manoni, Salti, Cecchi, Chiarelli. Thyroid Hypoplasia as a Cause of Congenital Hypothyroidism in Williams Syndrome. Hormone Research. (2008, Nov); 70(5): 316-318.
- ↑ 9.00 9.01 9.02 9.03 9.04 9.05 9.06 9.07 9.08 9.09 9.10 9.11 9.12 9.13 9.14 9.15 9.16 9.17 9.18 9.19 9.20 9.21 9.22 9.23 Morris A, Colleen. Williams Syndrome. Bethesda, MD: National Center for Biotechnology Information, U.S. National Library of Medicine; [updated 2006 April 21; cited 2011 April 5]. Available from: http://www.ncbi.nlm.nih.gov.
- ↑ 10.0 10.1 Meyer-Lindenberg A, Hariri A, Munoz K, Mervis C, Mattay V, Berman K, et al. Neural correlates of genetically abnormal social cognition in Williams syndrome. Nature Neuroscience. (2005, Aug); 8(8): 991-993.
- ↑ 11.0 11.1 11.2 11.3 11.4 Vanderbilt Kennedy Center. Williams Syndrome Research: A whole person approach. Nashville, Tennessee: Vanderbilt Kennedy Center; [updated 2010 January 21; cited 2011 April 5]. Available from: http://kc.vanderbilt.edu.
- ↑ Van Strien J, Lagers-Van Haselen G, Van Hagen J, De Coo I, Frens M, Van Der Geest J. Increased Prevalences of Left-handedness and Left-eye Sighting Dominance in Individuals with Williams-Beuren Syndrome. Journal of Clinical & Experimental Neuropsychology. (2005, Nov); 27(8): 967-976.
- ↑ 13.0 13.1 13.2 13.3 13.4 MedlinePlus. Williams Syndrome. Bethesda, MD: U.S. Department of Health and Human ServicesNational Institutes of Health; [updated 2011 March 28; cited 2011 April 5]. Available from: http://www.nlm.nih.gov/medlineplus.
- ↑ 14.0 14.1 Rose C, Wessel A, Pankau R, Partsch C, Bürsch J. Anomalies of the abdominal aorta in Williams-Beuren syndrome – another cause of arterial hypertension. European Journal of Pediatrics. (2001, Nov); 160(11): 655.
- ↑ 15.0 15.1 15.2 15.3 15.4 Kaplan P, Wang P, Francke U. Williams (Williams Beuren) syndrome: a distinct neurobehavioral disorder. Journal Of Child Neurology. (2001, Mar); 16(3): 177-190.
- ↑ 16.0 16.1 16.2 Kalbhenn T, Neumann L, Lanksch W, Haberl H. Spontaneous Intracerebral Hemorrhage and Multiple Infarction in Williams-Beuren Syndrome. Pediatric Neurosurgery. (2003, Dec); 39(6): 335-338.
- ↑ Sen-Wei T, Shyi-Kuen W, Ying-Ming L, San-Ging S. Early development in Williams syndrome. Pediatrics International. (2008, Apr); 50(2): 221-224.
- ↑ DiIenno M, Leibrand A. Connective Tissue Disorders in Children: Evaluation and Treatment on Land and in the Water. Combined Sections Meeting. February 2011.