Pleurodesis
This article is currently under review and may not be up to date. Please come back soon to see the finished work! (20/11/2023)
Description[edit | edit source]
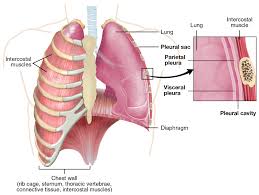
A pleurodesis is a form of pleural surgery which aims to obliterate the pleural space between the visceral and parietal pleura. This involves a chemical or surgical procedure to create an adhesion between the two pleura.
- Chemical pleurodesis: This is done by inserting a sclerosing agent into the pleural cavity via a chest tube. The drug used causes the pleural surfaces to become sticky and bond together, therefore closing the pleural space. The type of sclerosing agent to be used depends on local expertise, availability and the underlying reason why the pleurodesis is required. Drugs used include: sterile medical talc, Tetracyclines (minocycline, doxycycline), Silver nitrate, Iodopovidone, Bleomycin, Corynebacterium parvum with parenteral methylprednisolone acetate, Erythromycin, Fluorouracil, Interferon beta, Autologous blood, Mitomycin C, Cisplatin, Cytarabine, Doxorubicin, Etoposide, Bevacizumab (intravenous or intrapleural) and Streptococcus pyogenes A3 (OK-432)[1]
- Surgical pleurodesis: This is done through medical thoracoscopy, video-assisted thoracoscopy (VATS), or open thoracotomy, where either a sclerosis agent is placed in the pleural cavity or mechanical abrasion (also termed dry abrasion) is achieved by draining the pleural fluid using a tunneled catheter (induces pleurodesis without instillation of a sclerosing agent)[1].
- VATS with Talc pleurodesis stands out as the preferred choice due to its lower complication rates in comparison to alternative procedures, making it the most widely adopted method. [2]
Analgesia such as an NSAID or opiate, is usually given for the duration of the procedure. Patients can expect to be an inpatient for at least 24 hours following the procedure, longer if surgical pleurodesis is performed. Occasionally a single stitch may be used to close the insertion site of the chest drain.
Indication[edit | edit source]
The pleural space between the parietal and visceral pleura usually contains around 50ml of pleural fluid[1]. Under pathological conditions, such as pneumothorax or pleural effusion, air or excess fluid can build up in the pleural space. If this becomes recurrent and significantly symptomatic, pleurodesis may be indicated.
Any condition which causes extra fluid to collect in the pleural cavity may require a pleurodesis. These include: heart failure, pneumonia, tuberculosis, cancer, liver and kidney disease and inflammation of the pancreas[3].
Typically, a recurrent pleural effusion (particularly if malignant) or recurrent or persistent pneumothorax may be treated by pleurodesis.
Clinical Presentation[edit | edit source]
Depending on the reason that the pleurodesis is required, patients may present with signs of pleural effusion or pneumothorax, such as:
- Breathlessness
- Chest pain
- Cough
- Fever
- Tachycardia
- Tachypnoea
- Fatigue
Complications[edit | edit source]
Complications from chemical pleurodesis include:
- Chest pain
- Fever
- Breathlessness (localised inflammatory reaction to the procedure)
- Infection at the insertion site
- Empyema
Complications from surgical pleurodesis include:
- Pneumothorax[3]
- Injury to the chest wall, arteries, or lungs
- Blood clots, pulmonary embolism[3]
In the case of failed pleurodesis, pleurectomy may be required to control malignant pleural effusions[1]. Patients must be good surgical candidates and have a reasonably long expected survival because total radical pleurectomy/decortication requires a thoracotomy and is a major surgical procedure associated with considerable morbidity and some mortality.
Physiotherapy Post-Pleurodesis Surgery[edit | edit source]
Early mobilization[edit | edit source]
Minimum of 60 m be walked four times on day 1, 80 m on day 2, 100 m on day 3 and then continuing or increasing this as able throughout the rest of their admission.[4]
Patients are encouraged to mobilize at a pace where they achieve a breathlessness of 3-4 on the Borg ten-point scale.[5]
Physiotherapists need to be cautious when mobilizing patients with drains, especially when the drains are on suction. In such cases, it is advisable to utilize portable suction to ensure the safety and well-being of the patients during mobilization.
Airway Clearance Techniques[edit | edit source]
The use of airway clearance techniques and lung recruitment techniques such as active cycle of breathing (ACBT), IPPV, cough assist, manual techniques and Forced Expiratory Technique (FET) are used as clinically indicated.
Posture and arm exercises[edit | edit source]
Posture and arm exercises should also be used each day as they have been shown to reduce shoulder dysfunction post-operatively and regain functional activities of daily living.[6]
Wound support[edit | edit source]
Patient should be taught a supported cough to mechanically support the wound to reduce pain and improve confidence in coughing post-operatively, which will ultimately help in clearing the secretions.
Incentive Spirometry[edit | edit source]
The use of incentive spirometry (IS) remains widespread in the post-operative physiotherapy management of major surgical patients; however, several studies and meta-analyses have shown no additional benefit to the use of IS compared with standard physiotherapy of mobilization, breathing exercises and coughing at reducing the incidence of Post Pulmonary Complications or reducing hospital length of stay. [7] [8][9]
Resources[edit | edit source]
Oxford Centre for Respiratory Medicine. Pleurodesis Information for patients [online]. Accessed 31 March 2022
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 Ali M, Surani S. Pleurodesis. 2021. In: StatPearls [Internet]. Accessed 25 Mar 2022
- ↑ Ali M, Surani S. Pleurodesis [Internet]. PubMed. Treasure Island (FL): StatPearls Publishing; 2020.
- ↑ 3.0 3.1 3.2 Healthline. Pleurodesis [online]. Accessed 31 Mar 2022.
- ↑ Baddeley RA. Physiotherapy for enhanced recovery in thoracic surgery. Journal of Thoracic Disease [Internet]. 2016 Feb 1;8(Suppl 1):S107–10. /
- ↑ Ga B. Psychophysical Bases of Perceived Exertion [Internet]. Medicine and science in sports and exercise. 1982.
- ↑ Li WWL, Lee TW, Yim APC. Shoulder function after thoracic surgery. Thoracic Surgery Clinics. 2004 Aug;14(3):331–43.
- ↑ Agostini P, Naidu B, Cieslik H, Steyn R, Rajesh PB, Bishay E, et al. Effectiveness of incentive spirometry in patients following thoracotomy and lung resection including those at high risk for developing pulmonary complications. Thorax. 2013 Feb 21;68(6):580–5.
- ↑ Gosselink R, Schrever K, Cops P, Witvrouwen H, De Leyn P, Troosters T, et al. Incentive spirometry does not enhance recovery after thoracic surgery. Critical Care Medicine. 2000 Mar;28(3):679–83.
- ↑ Carvalho CRF, Paisani DM, Lunardi AC. Incentive spirometry in major surgeries: a systematic review. Brazilian Journal of Physical Therapy. 2011 Oct;15(5):343–50.