Physiotherapy Management of Traumatized Diaphragm
Introduction[edit | edit source]
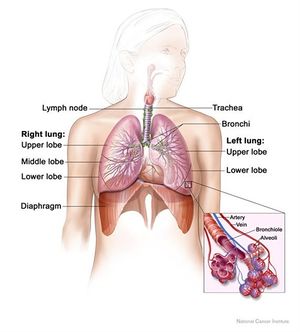
The diaphragm is a double-domed musculoskeletal partition separating the thoracic and abdominal cavities.It is crucial for respiration and maintenance of intra-abdominal pressure, and injuries can result in significant ventilatory compromise. Traumatic diaphragmatic injuries (TDI) include wounds and diaphragm ruptures due to thoraco-abdominal blunt or penetrating traumas.[1]
Traumatic diaphragmatic injuries are usually occult and can easily be missed. They occur in a context of multiple trauma making diagnosis difficult.[2] In developing countries where initial care of severely injured patients and diagnostic facilities are less than optimal, blunt TDI may go undiagnosed .[3] An accurate diagnosis requires a high index of suspicion as missed TDI may result in herniation and strangulation of intra-abdominal viscera into the thoracic cavity .[4] This herniation can interfere with breathing, and blood supply can be cut off to organs that herniated through the diaphragm, damaging them .[5] In 1579, Paré first described a diaphragmatic rupture in a french artillery captain, who initially survived a gunshot wound of the abdomen but died 8 months later of a strangulated gangrenous colon that herniated through a small diaphragmatic defect the size of a fingertip.[6] It was not until the end of the nineteenth century that surgical procedures for this condition were being undertaken. Despite having been recognised early in the history of surgery, blunt traumatic diaphragm rupture was a rarely reported condition before the twentieth century.
Etiology[edit | edit source]
Traumatic diaphragmatic injuries may be caused by:
- Blunt trauma (road traffic accidents and falls from height are the most common causes[3][7])
- Penetrating trauma with direct injury to the diaphragm is more common and accounts for about two-thirds of cases. It is frequently caused by gunshot injuries, stab wounds or impalement lesions. [8]
- Iatrogenic causes, for example during surgery to the abdomen or chest. .
Clinicians are trained to suspect diaphragmatic rupture, particularly if penetrating trauma has occurred to the lower chest or upper abdomen.[9] With penetrating trauma, the contents of the abdomen may not herniate into the chest cavity right away but they may do so later, causing the presentation to be delayed [7]. Since the diaphragm moves up and down during breathing, penetrating trauma to various parts of the torso may injure the diaphragm.[10] In extremely rare cases, the patient may have phrenic nerve injury leading to diaphragmatic paralysis.
Incidence and Epidemiology[edit | edit source]
The Incidence of TDI ranges from 0.8 to 8%, but the true incidence is likely to be higher due to missed or delayed diagnosis.[11]
- Penetrating TDI is highest among thoraco-abdominal gunshot wounds.[12] It accounts for 5 to 10% of admissions at most trauma centers.[11][13]
- While most blunt TDI are located in the central or posterolateral diaphragm and are related to embryologic weakness, penetrating TDI can occur
anywhere on the diaphragm.[14]
- Right-sided TDI is less common with most series reporting rates of 35-49%, however, they are much harder to diagnose due to coverage by the liver, and the true incidence is almost certainly higher than reported.[12]
- Left-sided TDI is more common, representing 75% of cases.[15] It have more frequent Injuries to associated organs, and have a higher morbidity and mortality risk.[13]
- Bilateral TDI is an extremely rare occurrence, reported as 2 to 8% overall but is seen almost exclusively with blunt mechanisms.[16]
- Traumatic diaphragmatic injuries can be missed even during exploratory surgery; with one series demonstrating 14% of TDI missed at an initial laparotomy.[17] This is likely due to the lack of a high index suspicion and the difficulty with direct visualisation of the hemi-diaphragms [12].
Relevant Anatomy[edit | edit source]
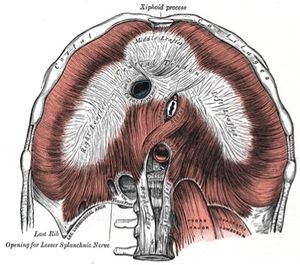
The diaphragm is a bi-domed structure, which separates the contents of the thorax from the abdominal cavity.It is composed of a thin leaflet of central aponeurosis in continuity with peripheral striated muscle, and is divided into two parts: the lumbar diaphragm and costal diaphragm. It originates anteriorly from the xiphoid, sternum, and the ribs and costal cartilages of ribs 7–12, as well as posteriorly from the lumbar vertebrae by means of the left and right crura.
The diaphragm inserts into the central tendon which is fused with the pericardium.There are three openings permitting the passage of three large structures.The inferior vena cava and the right phrenic nerves pass through an opening at the level of T8, the oesophagus and vagus nerves pass through an opening at the level of T10 and the abdominal aorta, thoracic duct and azygous vein pass through an opening at T12.The lumbocostal trigone, a thin area of degenerative muscle on the left diaphragm above the lateral arcuate ligament, represents an embryological transitional region between the costal and lumbar diaphragm.Nerve supply is via the phrenic nerves (C3, C4 and C5) which supplies sensory and motor innervation.Its actions during respiration are responsible for 70% of the work during inspiration.
Clinical Presentation[edit | edit source]
Clinical presentation varies depending on the mechanism of injury (i.e. blunt vs. penetrating) and the presence of associated injuries.[18]
Blunt traumatic rupture commonly occur in poly-traumatized patient and in most cases, early symptoms are missed, and the injury may be detected only at exploratory laparatomy for other suspected intra-abdominal organ injuries.[19]
In the post-traumatic period, presenting symptoms are usually due to herniation of intra-abdominal organs such as stomach, omentum, intestines, spleen and liver into the thoracic cavity.[19] Hence, patients may experience chest pain, recurrent shortness of breath and gastro-intestinal symptoms such as nausea and vomiting, epigastric discomfort or abdominal pains.[3] Bowel sounds may also be heard in left-sided hernia associated with bowel loops in the chest.
Commonly associated Injuries are visceral lacerations of the thoracal or abdominal organs and bone fractures.For visceral lacerations, splenic ruptures, pulmonary contusion, pneumothorax, liver laceration and cardiac contusion were most frequent.[20] The most likely associated fractures were pelvic fractures and rib fractures.[21]
Multiple classification systems have been used when referring to the natural history of TDI. An especially appropriate system described by Grimes [22] divides the presentation into three phases,
- the acute phase,The acute phase extends from the time of original trauma to the apparent recovery from the primary injuries. It is at this phase that the diagnosis of ruptured diaphragm is frequently missed, because of the presence of shock, respiratory insufficiency, visceral injuries and coma
- the latent phase, it begins as the intra-abdominal viscera occupy the defect and variably herniates into the thoracic cavity.
- the obstructive phase. it begins with the signs of visceral obstruction or ischemia as in other hernias.If herniation causes significant lung compression, it can lead to tension pneumothorax. Diaphragmatic paralysis also may occur. The box below depicts the clinical features of the three phases:[23]
Acute Phase
|
Latent Phase
Pain, left shoulder
Abnormal chest x-ray film findings |
Obstructive Phase
|
Diagnosis[edit | edit source]
A timely diagnosis of diaphragmatic injury can be problematic.This is due to other distracting and possibly life-threatening injuries that come with it. Patients present with shock 50 – 60% of the time.[2] Although delayed diagnosis is common,classical symptoms and signs can lead to a speedy diagnosis.
Information on the mechanism of injury should be obtained from the patient and pre-hospital care personnel. Physical findings can be either thoracic or abdominal.Thoracic signs include decreased breath sounds,fractured ribs,flail chest,and signs of haemothorax or pneumothorax.
Auscultation of bowel sounds in chest is pathognomic of diaphragmatic rupture,occurring due to herniation of bowel contents.
Abdominal signs include abdominal pain, guarding, absence of bowel sounds, and abdominal swelling, depending on the extent of injuries. Occasionally, physical examination can be relatively normal.
If diagnosis is delayed to months or years after the injury, symptoms are generally less severe,and are due to size reduction in the chest cavity (dyspnoea, orthopnea, respiratory distress), and partial or complete obstruction of herniated abdominal contents (nausea, vomiting, abdominal and chest pain).
The physical signs often present in a diaphragmatic hernia include: - diminished expansion of the chest, impairment of resonance, adventitious sounds, cardiac displacement, circulatory collapse, cyanosis, dyspnoea and asymmetry of the hypochondrium.[4]
Radiography
Plain chest radiographs are useful screening tool in cases of suspected diaphragmatic injury. Occasionally, the Chest X-ray can show pathognomic signs of a diaphragmatic injury.[2] It has an unsatisfactory accuracy in detecting TDI; indeed, it may appear normal or it may show only nonspecific changes in 20 – 50% of the patients affected by diaphragmatic rupture (Langdorf et al, 2015). However, as indicated by Bell and Radswiki,[24] the following signs are helpful in making the diagnosis:
- Inability to trace the normal hemi diaphragm contour
- Intrathoracic herniation of a hollow viscus (stomach, colon, small bowel) with or without focal constriction of the viscus at the side of the tear (collar sign)
- If large, the positive mass effect may cause a contralateral mediastinal shift
- Visualization of a nasogastric tube above the hemidiaphragm on the left side (as shown in figure 2)
- Left hemidiaphragm much higher than the right
Computerized Tomography (CT) scanning is perhaps the most useful diagnostic tool in the polytrauma patient. Not only can it provide information about diaphragmatic injuries, but also reveal other associated injuries. However, the patient does need to be haemodynamically stable. CT offers high accuracy (61% - 87% sensitivity and 72% - 100% specificity) in diagnosing TDI.[25] Helical CT has been shown to improve the early diagnosis of TDI in polytrauma patient. It has a sensitivity of 71% and a specificity of 100%.[26] Conventional CT has a variable sensitivity of between 14 and 61% and a specificity of between 76 and 99%. Multidetector CT allows thinner slicing and more detailed imaging in a shorter time, and a more flexible image reconstruction. It is nowadays becoming the imaging modality of choice in trauma patients and also for diagnosing TDI. The findings on CT demonstrating diaphragmatic rupture include:-
· Diaphragm discontinuity or segmental non-recognition.
· Intrathoracic herniation of abdominal contents.
· Constriction of herniated abdominal viscera
· Visualisation of the herniated viscera against the posterior chest wall, the “dependent viscera sign”. [27]
Magnetic resonance imaging (MRI) warrants higher tissue contrast resolution than CT and enables to clearly depict the diaphragm, but its use in an emergency setting is limited by the longer acquisition times and by the need for patient collaboration. Moreover, the original advantage of MRI in comparison with CT in diagnosing TDI i.e its multiplanarity, has been overwhelmed by the development of multidetector CT scanners.[28]
Surgical
Considering the difficulty in confirming diaphragmatic injury radiologically, it is unsurprising that the diagnosis is often unsuspected, and only found at laparotomy. This can account for the diagnosis of up to 50% of blunt ruptures.[2] With the increasing utilisation of laparoscopy and thoracoscopy, more diaphragmatic injuries are being correctly diagnosed and repaired. Diagnostic laparoscopy remains an excellent tool for the detection of haemoperitoneum, solid organ damage and diaphragmatic lacerations.[29]
Complications[edit | edit source]
The most serious complication of TDI is perforation of viscera into the thoracic cavity, leading to infections like pneumonia, empyemas and subphrenic or intra-abdominal abscesses. To avoid this, the vigorous irrigation of the thoracic and abdominal cavity with adequate drainage is recommended.[30] The morbidity includes complications like suture-line dehiscence, hemi diaphragmatic paralysis secondary to iatrogenic phrenic nerve injuries, respiratory insufficiency, empyemas and subphrenic abscess. The underlying trauma and associated injuries may be the cause of late morbidity.[4]
Management[edit | edit source]
Medical Management[edit | edit source]
First line of management would focus on resuscitating the patient. Patients with diaphragmatic injury require standard resuscitation and evaluation, including airway control and ventilation as well as restoration of circulation by stopping external haemorrhage and effective volume restoration. Ensure a patent airway, assist ventilation if required, and begin fluid resuscitation if necessary.
Place an NG tube when possible, as this will help in diagnosis if the NG tube appears in the chest on chest radiograph. Aspiration of gastric contents also helps to decompress any abdominal herniation and lessen the abdomino-peritoneal gradient that favors herniation into the chest. Consider placing a chest tube to drain any associated hemothorax or pneumothorax. Perform this with caution to prevent injury to herniated abdominal contents within the pleural cavity. Most surgeons recommend chest tube placement prior to transfer to another facility. If this is not required immediately in the definitive care institution, it may be delayed and completed in the operating room. Performing chest radiography before intubation may yield a better result (because it is more likely to show associated herniation).[18]
Surgical Management[edit | edit source]
Surgical repair via laparatomy or thoracotomy is necessary, even for small tears, because the defect will not heal spontaneously. Acute cases of TDI are better managed via a laparatomy as this also rules out and treat associated intra-abdominal organ injuries. Delayed cases, however, are better managed via a thoracotomy or thoraco-abdominal approach because of intra-thoracic adhesions.[31] Repair of diaphragmatic defect is usually achieved with interrupted non-absorbable sutures like nylon and prolene in a single or double-layered fashion. Mesh repair is used for large defect.
Physiotherapy Management[edit | edit source]
Assessment[edit | edit source]
Following Laparatomy and thoracotomy, there is overwhelming evidence of changes in lung function and associated clinical manifestations. These changes include characteristic reduction in lung volume which is primarily restrictive in nature, reduction in functional residual capacity, slowing of mucociliary clearance, and abnormalities in gaseous exchange.[32] Other frequently observed postoperative complications are post-thoracotomy pain syndrome and ipsilateral reduction in upper extremity range of motion and strength.[33]
Assessment is primarily focused on physical examination, chest expansion ABG analysis, pulmonary function test, chest X-ray, SpO2 (oxygen saturation), peripheral muscle strength and cardiopulmonary exercise testing.[34] In the preoperative phase, physical examination should address the presence of dyspnoea, exercise tolerance, cough, and expectoration. Examination should also focus on respiratory rate, pattern of breathing, and wheezing. Patient may show either normal or altered breathing pattern on physical examination. Postoperatively, patients usually present with monotonous shallow breathing without spontaneous deep breaths, increase in respiratory rate, decreased tidal volume, and significant change in minute ventilation. Wheeze, rales, or prolonged breath sounds will be revealed on auscultation.
Aims of Intervention[edit | edit source]
Generally the main aims in the postoperative phase are to maintain adequate ventilation, to assist in the removal of any excess lung secretions and to aid in the general positioning, bed mobility and early ambulation of the patient. Prevention of reduced joint movements or poor posture secondary to incisions or tubes, monitoring of adequate pain relief and appropriate oxygen therapy and humidification are also very important.
Physiotherapy Techniques[edit | edit source]
Physiotherapy techniques which help to achieve these aims include:
Early mobilization: With the development of laparoscopic surgery, improved anaesthetic and pain management many patients are often able to mobilize independently from a very early stage postoperatively. Some patients will require assistance because of the presence of the various drips and drains and it is sometimes safer to have two people assisting for the first stand or walk because of the patient's general fatigue and the risk of postural hypotension. A graduated walking programme adapted to suit each patient should be encouraged with the introduction of stair climbing at an appropriate stage. Hanekom et al [35] reported early mobilization to be a beneficial intervention for patients following upper abdominal surgeries. It is established that delaying early mobilization caused an increase in post-operative pulmonary complications.[36]
Breathing Exercises: Deep breathing exercises are taught to the patients and can help to obtain full expansion of the chest wall during spontaneous breathing. This is essential to help restore lung function and to prevent subsequent chest deformity. The patient is instructed to relax the shoulder and upper chest, take a slow deep breath in through the nose to fill up the lungs as fully as they can, hold breath for a few seconds (3 seconds), and breathe out slowly through the mouth. This should be done five times every hour. Patients are encouraged to practice breathing exercise frequently even when physical therapist isn’t present.
Active cycle of breathing (ACBT)
Thoracic expansion exercises
Coughing and Huffing Technique
Transcutaneous electrical nerve stimulation (TENS): TENS is a non-invasive analgesia technique that produces a significant reduction in pain. The use of TENS has been shown to decrease pain from shoulder flexion in patients undergoing axillary thoracotomy.[37]
Conclusion[edit | edit source]
Traumatic injuries of the diaphragm are often hidden and can be masked by other violent injuries associated with polytrauma. A high index suspicion and the use of relevant radiological investigation should help in early diagnosis. Mobilization, deep breathing exercise, thoracic expansion exercise should also be integrated early into patients care.
References[edit | edit source]
- ↑ Thiam O, Konate I, Gueye ML, Seck M, Cisse M, Diop B, Dirie ES, Ka O, Thiam M, Dieng M, Dia A. Traumatic diaphragmatic injuries: epidemiological, diagnostic and therapeutic aspects. Springerplus. 2016 Dec;5(1):1-6.
- ↑ 2.0 2.1 2.2 2.3 Bosanquet D, Farboud A, Luckraz H. A review diaphragmatic injury. Respiratory medicine CME. 2009;2(1):1-6.
- ↑ 3.0 3.1 3.2 Kidmas AT, Iya D, Isamade ES, Ekedigwe E. Delayed presentation of blunt traumatic diaphragmatic hernia: a case report. Nigerian journal of surgical research. 2005;7(3):323-4.
- ↑ 4.0 4.1 4.2 Petrone P, Leppäniemi A, Inaba K, Søreide K, Asensio JA. Diaphragmatic injuries: challenges in the diagnosis and management. Trauma. 2007;9(4):227-36.
- ↑ Senent-Boza A, Segura-Sampedro JJ, Olivares-Oliver C, Padillo-Ruiz FJ. Hepatothorax Caused by a Late Post-Traumatic Diaphragmatic Rupture. Cirugía Española (English Edition). 2015;9(93):e101.
- ↑ Bhatia S, Kaushik R, Singh R, Sharma R, Attri A, Dalal U, Dalal A, Bansiwal R. Traumatic diaphragmatic hernia. Indian Journal of Surgery. 2008;70(2):56.
- ↑ 7.0 7.1 Scharff JR, Naunheim KS. Traumatic diaphragmatic injuries. Thoracic surgery clinics. 2007;17(1):81-5.
- ↑ Morgan BS, Watcyn-Jones, T, Garner JP. Traumatic diaphragmatic injury. Journal of the Royal Army Medical Corps. 2010;156(3):139-144.
- ↑ Asensio JA, Roldán G, Petrone P, Rojo E, Tillou A, Kuncir E, Demetriades D, Velmahos G, Murray J, Shoemaker WC, Berne TV. Operative management and outcomes in 103 AAST-OIS grades IV and V complex hepatic injuries: trauma surgeons still need to operate, but angioembolization helps. Journal of Trauma and Acute Care Surgery. 2003;54(4):647-54.
- ↑ Fleisher GR, Ludwig S, editors. Textbook of pediatric emergency medicine. Lippincott Williams & Wilkins; 2010.
- ↑ 11.0 11.1 Lopez PP, Arango J, Gallup TM, Cohn SM, Myers J, Corneille M, Stewart R, Dent DL. Diaphragmatic injuries: what has changed over a 20-year period?. The American Surgeon. 2010;76(5):512-6.
- ↑ 12.0 12.1 12.2 DeBarros M, Martin MJ. Penetrating traumatic diaphragm injuries. Current Trauma Reports. 2015;1(2):92-101.
- ↑ 13.0 13.1 Zarour AM, El-Menyar A, Al-Thani H, Scalea TM, Chiu WC. Presentations and outcomes in patients with traumatic diaphragmatic injury: a 15-year experience. Journal of Trauma and Acute Care Surgery. 2013;74(6):1392-8.
- ↑ Dirican A, Yilmaz M, Unal B, Piskin T, Ersan V, Yilmaz S. Acute traumatic diaphragmatic ruptures: a retrospective study of 48 cases. Surgery today. 2011;41(10):1352.
- ↑ Hanna WC, Ferri LE. Acute traumatic diaphragmatic injury. Thoracic surgery clinics. 2009;19(4):485-9.
- ↑ Ties JS, Peschman JR, Moreno A, Mathiason MA, Kallies KJ, Martin RF, Brasel KJ, Cogbill TH. Evolution in the management of traumatic diaphragmatic injuries: a multicenter review. Journal of Trauma and Acute Care Surgery. 2014;76(4):1024-8.
- ↑ Esme H, Solak O, Sahin DA, Sezer M. Blunt and penetrating traumatic ruptures of the diaphragm. The Thoracic and cardiovascular surgeon. 2006;54(05):324-7.
- ↑ 18.0 18.1 Welsford. 2015. [online] Emedicine.medscape.com. Available at: https://emedicine.medscape.com/article/822999-treatment [Accessed 15 Feb. 2020]
- ↑ 19.0 19.1 Mercadante E, De TG, Rendina EA, Venuta F, Moretti M, Aratari MT, Furio GC. Diagnostic delay in post-traumatic diaphragmatic ruptures. Minerva chirurgica. 2001;56(3):299-302.
- ↑ Hofmann S, Kornmann M, Henne-Bruns D, Formentini A. Traumatic diaphragmatic ruptures: clinical presentation, diagnosis and surgical approach in adults. GMS Interdisciplinary plastic and reconstructive surgery DGPW. 2012;1.
- ↑ Pantelis D, Burger C, Hirner A, Wolff, M. Indirect Traumatic Diarrheal Rupture. The Surgeon. 2006;77 (4):360-366
- ↑ Grimes OF. Traumatic injuries of the diaphragm: Diaphragmatic hernia. The American Journal of Surgery. 1974;128(2):175-81.
- ↑ Shah R, Sabanathan S, Mearns AJ, Choudhury AK. Traumatic rupture of diaphragm. The Annals of thoracic surgery. 1995;60(5):1444-9.
- ↑ Bell, D. and Radswiki. Diaphragmatic rupture. [online] Radiopaedia.org. 2015. Available at: https://radiopaedia.org/articles/diaphragmatic-rupture [Accessed 15 Feb. 2020]
- ↑ Panda A, Kumar A, Gamanagatti S, Patil A, Kumar S, Gupta A. Traumatic diaphragmatic injury: a review of CT signs and the difference between blunt and penetrating injury. Diagnostic and interventional radiology. 2014 ;20(2):121.
- ↑ Iochum S, Ludig T, Walter F, Sebbag H, Grosdidier G, Blum AG. Imaging of diaphragmatic injury: a diagnostic challenge?. Radiographics. 2002;22(suppl_1):S103-16.
- ↑ Sangster G, Ventura VP, Carbo A, Gates T, Garayburu J, D’Agostino H. Diaphragmatic rupture: a frequently missed injury in blunt thoracoabdominal trauma patients. Emergency radiology. 2007;13(5):225-30.
- ↑ Bonatti M, Lombardo F, Vezzali N, Zamboni GA, Bonatti G. Blunt diaphragmatic lesions: Imaging findings and pitfalls. World journal of radiology. 2016;8(10):819.
- ↑ Asensio JA, Petrone P. Diaphragmatic injury. Current surgical therapy. 8th ed. Philadelphia: Elsevier Mosby Co. 2004:946-55.
- ↑ Yilmaz M, Isik B, Ara C, Yilmaz S, Kutlu R, Kocak Ö, Ugras M, Kirimlioglu V. Gastric perforation during chest tube placement for acute diaphragmatic rupture and review of the literature. Injury Extra. 2006;37(2):71-5.
- ↑ Edino ST, Alhassan S, Ajayi OO. Traumatic diaphragmatic rupture with gastro-pleuro-cutaneous fistula: A case report and literature review. Nigerian J Surg. 2002;8:18-20.
- ↑ Reeve J.2008. Physiotherapy interventions to prevent postoperative pulmonary complications following lung resection. What is the evidence? What is the practice?.
- ↑ Belda J, Cavalcanti M, Iglesias M, Gimferrer JM, Torres A. Respiratory infections after lung cancer resection. Clinical Pulmonary Medicine. 2006;13(1):8-16.
- ↑ Sultanpuram S, Alaparthi GK, Krishnakumar SK, Ottayil ZC. Physiotherapy practice patterns for management of patients undergoing thoracic surgeries in India: a survey. Surgery research and practice. 2016;2016.
- ↑ Hanekom, S.D., Brooks, D., Denehy, L., Fagevik-Olsén, M., Hardcastle, T.C., Manie, S. and Louw, Q., 2012. Reaching consensus on the physiotherapeutic management of patients following upper abdominal surgery: a pragmatic approach to interpret equivocal evidence. BMC medical informatics and decision making, 12(1), p.5.
- ↑ Haines KJ, Skinner EH, Berney S, Austin Health POST Study Investigators. Association of postoperative pulmonary complications with delayed mobilisation following major abdominal surgery: an observational cohort study. Physiotherapy. 2013;99(2):119-25.
- ↑ González PE, Novoa NM, Varela G. Transcutaneous Electrical Nerve Stimulation Reduces Post-Thoracotomy Ipsilateral Shoulder Pain. A Prospective Randomized Study. Archivos de Bronconeumología (English Edition). 2015;51(12):621-6.