Epidermolysis Bullosa
Original Editors - Cody Mumaw and Rachel Kirchgessner from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Top Contributors - Rachel Kirchgessner, Cody Mumaw, Elaine Lonnemann, 127.0.0.1, WikiSysop, Lucinda hampton, Evan Thomas and Kim Jackson
Definition/Description[edit | edit source]
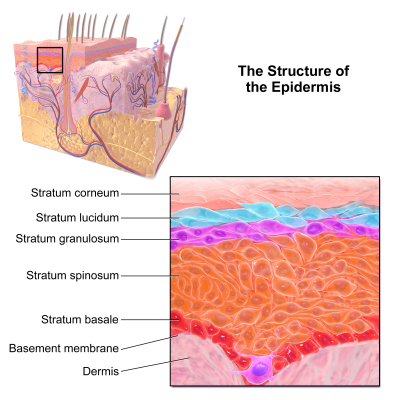
Epidermolysis bullosa (EB) consists of a rare group of genetically determined skin fragility disorders, categorized by blistering skin and mucosa in response to little or no apparent trauma, with some forms leading to substantial morbidity and increased mortality[1]. The fragility of skin and mucosa within this disease is due to defects in structural proteins within the epidermis, specifically at the epidermal-dermal junction, that cause a deficiency of cellular structures that normally stabilize the adhesion of the epidermis (see Cytoskeleton) These, in turn, result from abnormalities in the genes encoding various proteins that define EB into specific categories[2]. Accordingly, EB has been classified into three major different subtypes based on mode of inheritance, location of lesions, and clinical features which include the following three major forms: EB simplex (EBS), junctional EB (JEB), and dystrophic EB (DEB)[1]. Each are based on the level of blistering of the skin, although the classification of EB continues to evolve with recognition of up to 30 clinical subtypes[2].
Prevalence[edit | edit source]
All types and subtypes of EB are rare. Estimates of prevalence and incidence of EB have been endeavored by many different sampling techniques in numerous populations worldwide, but the most accurate and up to date epidemiological data is derived from the National EB registry (NEBR) from the USA. This registry is a cross-sectional and longitudinal epidemiological study of patients diagnosed with EB across the entire U.S. Over 16 years (1986-2002), 3,300 patients were identified, enrolled, classified, characterized, and followed for outcomes[3]. Among this study, the overall incidence and prevalence of inherited EB, within the United States, was found to be approximately 19.60 per one million live births and 8.22 per one million population, respectively. When analyzing the different classifcations of EB, the incidence and prevalence rates for EB simplex was found to be 10.75 and 4.65, for junctional EB 2.04 and 0.44, for dystrophic EB recessive type 2.04 and 0.92, and dystrophic EB dominant type 2.86 and 0.99[1].
Characteristics/Clinical Presentation[edit | edit source]
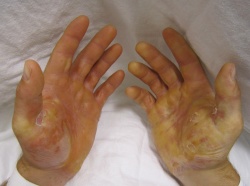
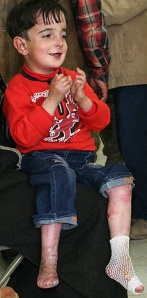
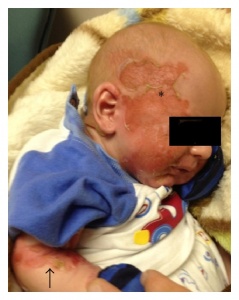
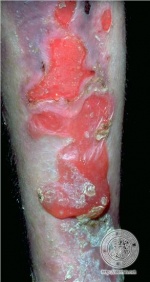
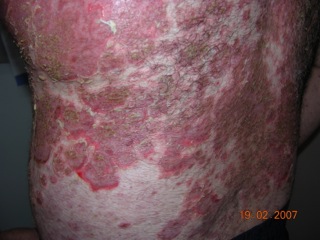
The primary symptom of EB is lesions to the skin. These lesions appear in multiple forms, most commonly in the form of blisters or open wounds[4]. Areas of appearance of skin lesions can be anywhere on the body but tend to arise on parts of the body that are subject to repeated friction or minor trauma, however the amplitude of this stress is not proportionate to the amount of skin deformation or blister/wound size. EB is classified into three primary groups, determined by the depth at which the skin is impacted. These groups are EB simplex, junctional EB, and Dystrophic EB[4].
►EB simplex
Caused by a mutation in the Keratin 5 or 14 genes, the simplex form is the most common type of EB[5]. This type of EB affects the epidermisand generally the blistering of the skin is most severe on the feet and hands. Other characteristics of EB simplex are a strong association with the dominant gene and, in most cases, is not life threatening, however the Dowling Meara variety could be fatal[5].
►Junctional EB
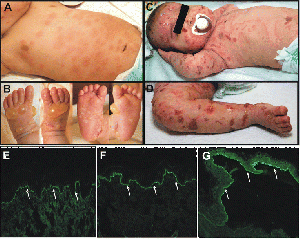
Junctional EB (JEB) occurs in the lamina lucida of the dermal-epidermal junction[6]. There are two forms of JEB, Herlitz and non-Herlitz. The Herlitz subtype occurs in infants and currently has no cure with fatality rate around 90%, while the non-Herlitz form is compatible with a normal life-span. Characteristics of JEB include fragile skin and mucous membrane resulting in blistering of the skin[6]. In non-Herlitz JEB the most common problems include nail dystrophy and loss, corneal erosion and scarring, dental problems, and fragility of the genitourinary tract leading to urethral stenosis
►Dystrophic EB
Caused by a mutation in the COL7A1 gene, dystrophic epidermolysis bullosa (DEB) occurs at the dermal-epidermal junction below the basal lamina[7]. DEB can be broken down further into two categories by the way it is inherited, either by the dominant or recessive gene. A defect in the dominant gene resulting in DEB usually presents with more mild symptoms, while inheritance by a mutation in the recessive gene has more severe symptoms[7]. Patient’s with DEB typically present with blistering of skin over a large portion of their body, particularly on the extensor surfaces of the extremities and the torso[4].
Associated Co-morbidities[edit | edit source]
►Contractures and scaring
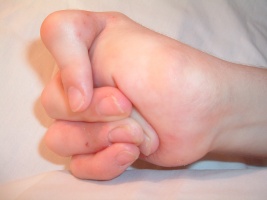
Due to the extensive damage to the skin, contractures and atrophic scarring are common to occur on the hands and feet of individuals with EB. Contractures are typically treated with surgical release and if severe enough amputation of digits may be indicated[4].
►Anemia
Due to the open wounds and blistering of the skin that occurs with EB, blood loss results and occasionally leads to chronic anemia. Treatment includes increased dietary intake of iron or supplemented with iron in liquid form[4].
►Gastrointestinal issues
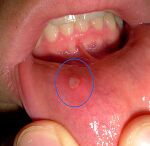
Oral mucous lesions cause pain in the mouth and result in the refusal to eat[8]. This begins the viscous cycle of adverse effects in the GI system. Malnutrition is very detrimental to these patients, as this disease requires an elevated need for nutritional supplements due to blood loss and chronic inflammation. Other gastrointestinal issues associated with EB include dysphasia and chronic constipation[1].
►Cardiopulmonary complications
Hoarseness, stridor, and respiratory distress can be effects of EB due to the chronic inflammation of the airways[8]. Cardiomyopathy has also been associated with EB, especially in children, however the connection between these two conditions in not well understood[1].
►Genitourinary complications
Both DEB and JEB may cause fragility, scarring, and infection in and around the genitourinary tract. Conditions such as glomerulonephritis, renal amyloidosis, and immunoglobulin A nephropathy have all contributed to renal failure in cases of EB[4].
►Psychological complications
Depression and other emotional issues are common with EB. Contributing factors to psychological issues include unpleasant cosmetic features due to skin blistering, pain, and other complications affecting other body systems[4].
►Pain
Pain in EB may be a result of multiple factors including chronic wounds/blisters, contractures, osteoporosis, oral mucous lesions, inflammation or blistering of the esophagus, corneal ulcerations, and chronic constipation[4].
►Cancer
Squamous cell carcinoma is associated with severe forms of DEB. This occurs with multiple sites of primary metastases, most commonly on bony prominences[4].
Medications[edit | edit source]
Currently no drugs are known to correct the primary molecular effects in EB. Recently, the use of topical opiates for pain management has been proven to reduce the need for powerful systemic analgesia. Amitriptyline, as well, has also been found to be useful in both children and adults in reducing pain. As far as systemic treatment, no agents so far have proven to be effective in controlling blisters in patients diagnosed with EB. Prolonged use of corticosteroids are contraindicated because of the high risk of complications associated with this drug. No other medication, including phenytoin and tetracycline, have improved the blistering or epithelial disadhesion in EB. Thus, there is no current reliable clinical trial evidence for any type of treatment with medication[1].
Diagnostic Tests/Lab Tests/Lab Values[edit | edit source]
Diagnosing EB is achieved by one of three primary methods including: transmission electron microscopy (TEM), immunofluorescence antigen mapping (IF), and mutational analysis. The TEM method was the first and has remained the gold standard tool for diagnosis EB. Drawbacks of this method are that it is expensive, time consuming, and requires a skilled operator. IF is just as reliable as TEM and is more time efficient and easier to use. This method uses poly or monoclonal antibodies to detect structural proteins in keratinocytes or in the dermal-epidermal junction. Mutational analysis uses IgG antibodies to diagnosis all three primary forms of EB. For EBS mutational analyzes cytokeratin 5 and 14, plectin, alpha-6 and beta-4 integrin within the epidermis by cytolysis of basal keratinocytes. Mutational analysis examines type XVII collagen and laminin 332 in diagnosing JEB. Diagnosing DEB is done by using type IV collagen antibody to visualize the level of blister formation, and a reaction on the roof of the blister indicates dermolytic blistering which confirms the diagnosis. Mutational analysis is the ultimate means of determining the mode of inheritance, level of blister formation, and type of molecular mutation[1].
Etiology/Causes[edit | edit source]
With epidermolysis bullosa, the fragility of skin and mucosa in which erosions occur because of slight mechanical trauma are inadvertently the result of defects in structural proteins within the epidermis. This, in turn, occurs because of abnormalities in the genes encoding these various proteins[2]. Based on pathophysiology, EB has been classified into three major subtypes that include their own genetic abnormalities:
►EB simplex
In EB simplex, the gene that helps make keratin, a fibrous protein in the top layer of skin, is faulty. Though it does not cause scarring, this subtype causes blister cleavage to occur within the epidermis[9].
►Junctional EB
In junctional EB, defects in the basement membrane, specifically laminins, cause blister cleavage below the epidermis but above the basallamina. This in turn, causes tissue separation and blistering in this deep layer of skin[9].
►Dystrophic EB
In dystrophic EB, collagen VII mutations affect the anchoring fibrils and cause dermal blisters below the basal lamina within the papillary dermis. Because of this mutation, layers of the skin don’t join properly. This type can be either dominant or recessive[9].
Systemic Involvement[edit | edit source]
Although the most common form of EB (EB simplex) is rarely life threatening and does not commonly affect multiple body systems, dystrophic and sometimes junctional EB may both lead to death[10]. As discussed in the associated comorbities section above, these two forms of EB can cause damage and lead to pathological conditions in multiple body systems.
Medical Management [edit | edit source]
Currently there is no effective therapy or cure for EB[1]. Listed below is medical management that was proven to be the most effective in managing EB.
►Preventing blisters
Avoidance of aggravating factors for blistering remains the backbone of management. Measures to reduce these factors by using heat and humidtiy to lower the threshold for blistering are important[1].
►Nursing Care
Also deemed important is successful management through nursing care. Mothers should be educated to nurse their children on thick foam pads to prevent them from inducing blisters from unnecessary trauma. As for older children, they should be educated upon certain precautions such as using adhesive tapes, sphygmomanometer cuffs, tourniquets, and other instruments that may shear their skin or mucous membranes. The adhesions should be cleaned with sterile saline and afterwards covered with nonadherant dressings. Because there is a risk of development of antibiotic resistant bacteria, topical antibiotics are usually avoided. Constipation needs to managed with a sufficient amount of fiber as well as stimulant laxatives. Balloon dilation is also required for esophageal strictures[1].
►Dental Care
Good dental hygiene is vital for patients with EB, as regular visits to the dentist are recommended. This should be initiated as quickly as tooth eruption and oral mucosal involvement begins. Good oral hygiene, crown placements, and tooth implants should be maintained to preserve function[1].
►Dietary Care
Lack of vitamins and trace metals are common with patients who receive gastrostomy feeding. It is important to do regular nutritional evaluations with patients who have EB as well as provide them with dietary advice and appropriate supplementation[1].
►Molecular Therapy
Gene therapy is probable to become a reality sometime in the future. With gene therapy, there are two broad principles-ex vivo and in vivo. Ex vivo is a specific type of gene therapy that uses cultured cells such as keratinocytes, specifically from the affected individuals, which are propagated and transduced in culture to re-express the gene that is malfunctioning in the patients. Contrarily, the in vivo gene introduces the transgene directly into the target tissue by either direct injection of the genetic material into the skin or mucous membranes or by topical application. The objective of molecular therapy is to replace one of the two mutant alleles in recessive forms of EB by introducing one normal allele into somatic cells. The first successful gene therapy for EB was reported by Michele De Luca. With this, areas of skin on the anterior thighs of an adult male patient with junctional EB were “cured” by transplanting genetically modified epidermal sheets grown in culture with a total of 500cm² on both legs. These epidermal sheets were taken from palm biopsies and transfected ex vivo, with a retroviral expressing normal lamina 5-3, that was grown from the patient’s own laminin 5-3-chain-deficient epidermal stem cells. De Luca’s team is now planning the replacement of the big proportion of the patient’s skin surface with this genetically modified skin. These current advances in molecular genetics has allowed prenatal and preimplantation genetic diagnosis to be possible, broadening many options with patients in management of EB[1].
Physical Therapy Management [edit | edit source]
Physical therapy is very practical in management of epidermolysis bullosa. When providing this treatment of care, it is always necessary to ask for input from clinicians in virtually all areas of medicine, including pediatricians, surgeons, dentists, gastroenterologist, hematologists, otorhinolaryngologists, and dietitians. Because EB is rare, patients suffering from this disease feel abandoned, neglected, and frustrated that clinicians may know very little. Thus, staying up to date on current medicine and practice is vital in providing optimal care that is suitable for these patients[2]. When providing treatment to patients suffering from EB, initiating early exercises to help reduce the severity of contractures, which are almost unavoidable, is necessary in maintaining adequate range of motion to complete all activities of the daily living. Upon surgical release of the contracted fingers, it is important for the patient suffering from EB to also get strength and sufficient ROM back. Also, being observant of the provoking factors that may be causing blisters with the patient’s posture in sitting, when doing transfers, and movement during daily skills can be prevented by providing compensatory change with skilled thought. Patient education on the importance of appropriate surfaces to prevent shearing of the skin as well as adding modalities such as as heat and moisture can lower the threshold for blistering and potentially lessen the occurrence. Also, if the therapist is well cultured in the area of wound care, providing the patient with proper education of signs and symptoms of an infection and ways to put on and take bandages off without causing shearing to the skin is important in patient management[1].
Differential Diagnosis[edit | edit source]
The size or validity of the differential diagnosis presented with a child or adult with blistering of the skin is most certainly a reflection of the level of training and expertise of the physician. In almost all situations the diagnosis of EB should be apparent to a dermatologist, with only a marginal number of cases needing more of wide-ranging differential diagnoses before tissue confirmation[3].
►Pemphigus vulgaris
Pemphigus vulgaris (PV) is a rare autoimmune, intraepithelial, blistering disease that is associated with a very encumbering
quality of life. PV is characterized by autoantibodies against desmoglein 3 and desmoglein 1 of keratinocytes. It clinically, however, is characterized by extensive blisters that affect the skin and mucous membranes[3]. This is found in the mucous membrane in 95% of cases (oral, pharyngeal, esophageal, nasal, and genital), followed by skin lesions, crusting, and purple stains on the anterior chest, back, and abdomen, with oral lesions being common as well. Characteristics of these blisters include a diameter consisting of millimeters to centimeters that is isolated or in groups that appear to be fragile and flaccid, breaking in eroded areas, becoming bloody and wet, and covered with bloody crusting. Areas such as the face, scalp, neck, sternum, armpit, groin and periumbicals may experience a burning sensation[4]. The treatment goal of PV is to prevent new blister formation, heal old wounds, and eventually complete the tapering of treatment. As of now, there has been been no treatment strategy for PV according to the international consensus. The best treatment strategy for PV remains unclear as higher quality RCTs are needed in the future to explore other unstudied interventions[3].
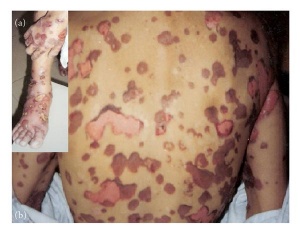
►Bullous systemic lupus erythematosus
Bullous systemic lupus erythematosus (BSLE) is a rare and distinct subtype of SLE, occurring mostly in the third decade. This subepidermal blistering disease occurs in patients that have been diagnosed with systemic lupus erythematosus (SLE), with a low occurrence of only 1%. Clinically, patients diagnosed with BSLE present with a rapid, widespread small vesicles or large tense blisters that are filled with fluid[5]. These lesions may affect the trunk and limbs, or even face and mucous membranes, involving any area of the body. Diagnosis of BSLE includes the following: a past diagnosis of SLE based on American College Criteria (ACR), presence of vesicles and bullae most commonly located in sun-exposed sites, histopathology findings, and deposition of immunoglobulins at the basement membrane zone. Patient evaluation is critical, as a prompt diagnosis may prevent further SLE complications and change the prognosis and method to treatment[6]. In regards to treatment for BSLE, dapsone, corticosteroids, and/or immunosuppressant’s are the first treatment options to consider. If symptoms do not go away rituximab might be appropriate to consider[5].
►Bullous Pemphigoid
Bullous pemphigoid (BP), idiopathic in origin, is the most common autoimmune subdermal blistering disease of the skin and mucous membranes, occurring from antibodies directed against the proteins BPAG1 and BPAG2[7]. This disease is most commonly seen in elderly individuals and is characterized with blistering of the skin as well as intense pruritus[8]. Systemic corticosteroids, prednisone, in doses of 1 mg/kg/day, with dose tapering according to the therapeutic control of disease was shown to be the treatment of choice with the highest level of evidence for BP[9].
►Dyshidrotic eczema
Dyshidrotic eczema is a chronic, recurrent skin disease that effects the palms and soles symmetrically. It often is a very intense and painful condition that can have a very devastating impact on quality of life. Although the etiology is unclear, dyshidrotic eczema is often triggered by emotional stress, smoking, seasonal changes, fungal infections, atopy, nickel allergy, hyperhidrosis, and intravenous immunoglobulin therapy[11]. Much like many other types of eczema, this is a benign chronic inflammatory disease that may occur at intervals of 3 to 4 weeks for months or years, or even progress to longer irregular intervals. Dyshidrotic eczema has no impact on survival as well as very few effective treatment options[1].
►Linear IgA Bullous Dermatosis
Linear IgA Bullous Dermatosis (LAD) is an autoimmune, chronic bullous disease affecting mainly young children and adults. Sub-epithelial blister formation with neutrophils along the basement membrane zone are typically histological characteristic features in LAD.
Childhood onset LAD is characterized by vesicles and bulla mainly around the mouth, eyes, lower abdomen, thighs, buttocks, genitals, wrists, and ankles. Subjective symptoms range from mild pruritus to severe burning. The adult onset form presents with lesions on the trunk and occasionally head and limbs. The most often used treatment modalities in both children and adults are corticosteroids, dapsone, and sulpapyridine[2].
Case Reports/ Case Studies[edit | edit source]
Schmidt E, Benoit S, Bröcker EB, Zillikens D, Goebeler M. Successful adjuvant treatment of recalcitrant epidermolysis bullosa acquisita with anti-CD20 antibody rituximab. Archives of dermatology. 2006 Feb 1;142(2):147-50.
►http://archderm.jamanetwork.com/article.aspx?articleid=402587&resultclick=1
Daka A. K, Hasbahta T. S, Gärqari A, Krasniqi M. V, Daka Q. Neonate with Severe Complications of Epidermolysis Bullosa and Bilateral Clubfoot: An Unusual Case Presentation and Treatment. Journal Of Nepal Paediatric Society [serial on the Internet]. (2015, May), [cited April 8, 2016]; 35(2): 180-183. Available from: Academic Search Complete.
Maitra S, Das N, Lal N. Peeling skin in newborn with abdominal distension. Indian Journal Of Paediatric Dermatology [serial on the Internet]. (2016, Jan), [cited April 8, 2016]; 17(1): 29-31. Available from: Academic Search Complete.
Resources[edit | edit source]
- Debra of America – Dystrophic Epidermolysis Bullosa Research Association - http://www.debra.org
- Epidermolysis Bullosa Medical Research Foundation - http://www.ebkids.org/about-ebmrf/
- EB Info World - http://blog.ebinfoworld.com
- The Future of Treatments for EB- https://youtu.be/DIpKZFCu3Q0?list=PLB4B29A07FFFC00CE
References[edit | edit source]
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 Sarkar R, Bansal S, Garg V. Epidermolysis bullosa: Where do we stand? Indian Journal of Dermatology, Venereology and Leprology 2011 Jul;77(4):431-8.
- ↑ 2.0 2.1 2.2 2.3 2.4 Mitsuhashi Y, Hashimoto I. Genetic abnormalities and clinical classification of epidermolysis bullosa. Arch Dermatol Res 2003 04;295:S29-33.
- ↑ 3.0 3.1 3.2 3.3 Fine J. Inherited epidermolysis bullosa. Orphanet Journal of Rare Diseases 2010;5:12.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 4.9 Pillay E. Epidermolysis bullosa. Part 1: causes, presentation and complications [corrected] [published erratum appears in BR J NURS;2008 Apr 10-23;17(7):413]. British Journal Of Nursing [serial on the Internet]. (2008, Mar 13), [cited April 5, 2016]; 17(5): 292-296 5p. Available from: CINAHL with Full Text.
- ↑ 5.0 5.1 5.2 5.3 Kiritsi D, Pigors M, Tantcheva-Poor I, Wessel C, Arin M, Has C, et al. Epidermolysis Bullosa Simplex Ogna Revisited. Journal Of Investigative
- ↑ 6.0 6.1 6.2 Yan E, Paris J, Ahluwalia J, Lane A, Bruckner A. Treatment decision-making for patients with the Herlitz subtype of junctional epidermolysis bullosa. Journal Of Perinatology [serial on the Internet]. (2007, May), [cited April 5, 2016]; 27(5): 307-311 5p. Available from: CINAHL.
- ↑ 7.0 7.1 7.2 Yang C, Lu Y, Farhi A, Nelson-Williams C, Kashgarian M, Choate K, et al. An Incompletely Penetrant Novel Mutation in COL7A1 Causes Epidermolysis Bullosa Pruriginosa and Dominant Dystrophic Epidermolysis Bullosa Phenotypes in an Extended Kindred. Pediatric Dermatology [serial on the Internet]. (2012, Nov), [cited April 5, 2016]; 29(6): 725-731. Available from: Academic Search Complete.
- ↑ 8.0 8.1 8.2 Schachner L, Feiner A, Camisulli S. Epidermolysis Bullosa: Management Principles for the Neonate, Infant, and Young Child. Dermatology Nursing [serial on the Internet]. (2005, Feb), [cited April 6, 2016]; 17(1): 56-59. Available from: Academic Search Complete.
- ↑ 9.0 9.1 9.2 9.3 Formsma SA, Maathuis CBG, Robinson PH, Jonkman MF. Postoperative Hand Treatment in Children with Recessive Dystrophic Epidermolysis Bullosa. Journal of Hand Therapy 2008 Jan;21(1):80-4; quiz 85.
- ↑ Kim J, Kim S. Epidermolysis bullosa acquisita. Journal Of The European Academy Of Dermatology And Venereology: JEADV [serial on the Internet]. (2013, Oct), [cited April 8, 2016]; 27(10): 1204-1213. Available from: MEDLINE.
- ↑ Markantoni V, Kouris A, Armyra K, Vavouli C, Kontochristopoulos G. Remarkable improvement of relapsing dyshidrotic eczema after treatment of coexistant hyperhidrosis with oxybutynin. Dermatologic Therapy [serial on the Internet]. (2014, Nov), [cited April 5, 2016]; 27(6): 365-368. Available from: MEDLINE.