Core stabilisation exercises vs decompression surgery for lumbar spinal stenosis
Top Contributors - Lyle Brennan, Toby Hale, Cindy John-Chu, Malachi Byrne, Anita Vagvolgyi and Kim Jackson
This article or area is currently under construction and may only be partially complete. Please come back soon to see the finished work! (11/10/2023)
Introduction: Lumbar Stenosis [edit | edit source]
-Lumbar spinal stenosis is a medical condition characterized by the narrowing of the spinal canal as a consequence of bone and soft tissue degeneration, including disc herniation, facet and ligamentum flavum hypertrophy, and osteophyte formation (Kreiner et al., 2014). Acquired forms of LSS are further classified as degenerative, spondylolisthesis, iatrogenic (postsurgical), posttraumatic, or combined (Fritz, Erhard and Vignovic, 1997)
Spinal stenosis is a term used to describe a narrowing of the spinal canal. The narrowing may not cause any symptoms. However, the narrowing may progress to cause squeezing (compression) of the spinal nerves. Spinal stenosis can cause back pain and/or leg pain. Most often it occurs when you walk. Weakness of the legs may make you feel unsteady. This may affect both legs and just one leg. (Coppen, 2022)
Image 1 of anatomy & location site of lumbar stenosis: [1]/ 21st Sept 2016
Epidemiology[edit | edit source]
The prevalence of LSS is estimated to be 9% in the general population, and up to 47% in people over age 60 (Kalichman et al., 2009). Spinal stenosis is most common in people over 50 years of age (Fritz, Erhard and Vignovic, 1997). Spinal stenosis is one of the most common causes of non-traumatic spinal cord injuries (Ho et al., 2007).
With the current ageing population in the UK, the prevalence of LSS is likely to continue to rise in the future, increasing the financial burden of lower back pain.
Aetiology[edit | edit source]
Lumbar stenosis is the consequence of degenerative changes in the lumbar spine because of age (wear and tear). As the aging process increases in the individual, the incidence for lumbar stenosis to occur, increases in likelihood (Genevay and Atlas, 2010).
Normal ageing, the intervertebral disc loses proteoglycans and water, leading to diminished disc height and circumferential disc bulging. (Tseng, 2021)
1. Biomechanical stress on the facet joints leading to facet hypertrophy and thickening of the ligamentum flavum, along with collagen and calcium deposition.
2. These changes reduce the diameter of the spinal canal and neural foramina and the space available for the dural sac and exiting nerve roots.
3. Intrusion of the disc anteriorly within the spinal canal and of the hypertrophied facets and ligamentum flavum laterally and posteriorly creates a characteristic triangular or trefoil appearance on cross-section of the canal.
Risk Factors[edit | edit source]
- age over 40 years, (Yabuki et al., 2013)
- previous back injury or surgery (Aiki et al., 2005)
Symptoms[edit | edit source]
Symptoms include reduced walking distance because of impairment of neurogenic claudication (pain in buttocks and/or legs) (Verbiest, 1954). Walking or prolonged standing aggravates symptoms (Cook et al., 2020) and forward bending (the shopping cart sign) or sitting relieves symptoms typically in most patients with lumbar spinal stenosis (Cook et al., 2020).
Additionally, lower back pain is common (Tomkins-Lane et al., 2016) and pain is bilateral in central lumbar spinal stenosis and can be accompanied with paraesthesia, or weaknesses on lower extremities including the buttocks or thighs.
The table below indicates key clinical features for a diagnosis of lumbar stenosis; however, it is important to be aware that not all symptoms are present in those with lumbar spinal stenosis.
|
(Tomkins-Lane et al., 2016)
Diagnosis[edit | edit source]
The diagnosis of the condition is based on clinical history, a thorough physical examination to rule out other possible aetiologies of presenting symptoms, and consistent radiographical findings demonstrating narrowing of the spinal canal and/or neuroforamen.
Red flags must be assessed and monitored throughout the patients plan of care.
Diagnosis options can include: 1. Patients History, 2. Examination, 3. Objective findings: MRI, X-Rays, CT scanning, Electrodiagnosis. The more objective forms of diagnosis are typically used more frequently for pre surgery patient and patients with excessive and consistently reported pain symptoms. In addition, many patients are asymptomatic to typical symptoms of lumbar stenosis and so equipment such as MRI can be used to confirm a clinician’s findings. (Tseng, 2021)
Link to imaging of MRI scan for Lumbar Stenosis [2] (Lee et al., 2015)
Severities – 3 systematic graded systems for severity of lumbar stenosis (Walther, 2023)[edit | edit source]
Lumbar spinal stenosis grading refers to systems for classifying the severity of central spinal canal narrowing around the cauda equina nerve roots.
| Grading system | Lee
(Guen et al., 2011) |
Schizas
(Schizas et al., 2010) |
Miskin-Mandell
(Miskin et al., 2021) |
| Tier 1 | No stenosis
The anterior CSF space is not obliterated |
No / minor stenosis
CSF is clearly visible in the dural sac but the distribution is heterogeneous A1: rootlets lie dorsally, occupying less than half the dural sac area. A2: rootlets lie dorsally, in contact with the dura and in a horseshoe configuration. A3: rootlets lie dorsally, occupying more than half the dural sac area. A4: rootlets lie centrally, occupying more than half the dural sac area |
Normal
Nerve roots are freely distributed, without crowding; the anterior margin of the thecal sac is flat or convex |
| Tier 2 | Mild stenosis
The anterior CSF space is mildly obliterated but all cauda equina can be clearly separated from each other |
Moderate stenosis
Rootlets occupy the whole of the dural sac but they can still be individualised; some CSF is still present, giving a grainy appearance to the sac |
Mild spinal stenosis Slight crowding of the nerve roots; the anterior margin of the thecal sac is flat or slightly concave; the nerve roots remain distinguishable from the CSF |
| Tier 3 | Moderate stenosis
The anterior CSF space is moderately obliterated and some of the cauda equina are aggregated (impossible to visually separate) |
Severe stenosis
No rootlets are recognised; no CSF is visible, giving a homogeneous gray signal to the sac, while epidural fat remains posteriorly |
Moderate stenosis Crowding of the nerve roots, resulting in a homogeneously “speckled” appearance of CSF interspersed with nerve roots; the anterior margin of the thecal sac is concave |
| Tier 4 | Severe stenosis
The anterior CSF space is severely obliterated so as to show marked compression of the dural sac and the cauda equina appears as one bundle (none can be visually separated from each other) |
Extreme stenosis
In addition to no rootlets being recognizable, the epidural fat posteriorly is obliterated |
Severe spinal stenosis Complete effacement of CSF, resulting in nerve roots being not individually distinguishable; the anterior margin of the thecal sac is concave or not discernible |
Effects on patients and health service[edit | edit source]
ADL’s and Lower back pain
ADL’s = Active Daily Livings
Pain in legs and difficulty walking can limit function and participation in daily activities, which can have negative psychological effects. (Ammendolia et al., 2017)
Patient’s perspective: (Jensen et al., 2021)
“I was in incredible pain every single day and sometimes had to walk bending forward. The thought of having to walk the 400 metres to the bus was a nightmare. Sometimes I could not walk more than 100 metres and I had to stand still for quite a long time before I was able to walk again. I was worried about whether I could reach the bus and I didn’t like to feel pathetic”.
Effects upon health service[edit | edit source]
Musculoskeletal conditions such as back pain cost the National Health Service (NHS) £4.76 billion each year.
Effects on health services within the NHS. These include:
1. increased healthcare utilization (Increased focus on MDT quality of communication + teamwork
2. the need for diagnostic tests and imaging, a range of treatment options (including surgeries)
3. rehabilitation and follow-up care.
In fact, musculoskeletal (MSK) conditions account for approximately 30% of GP in England. This imposes additional costs on the NHS financial budget. They are estimated to cost the National Health Service (NHS) £4.76 billion each year, and this is likely to increase. (Office for Health Improvement and Disparities, 2022)
Current practice[edit | edit source]
Due to the heterogeneity of the condition, and the differences in clinical presentations of individuals with lumbar spinal stenosis with patients ranging from asymptomatic to completely debilitating pain, the management of the condition varies largely between patients, and there is no gold standard management that is directed to every patient.a
What do the guidelines suggest?[edit | edit source]
The North American Spine Society clinical practice guidelines (Kreiner et al, 2011) highlight that there is a paucity in literature surrounding the impact of physical therapy on lumbar spinal stenosis, but that it should be included as part of a complex treatment strategy. However, surgical intervention, particularly decompression surgery, was positively indicated for individuals with moderate or severe symptoms of lumbar spinal stenosis.
The NASS guidelines also highlighted evidence for the following interventions:
- Caudal epidural injections – effective in short to medium term if administered in a multiple injection regimen.
- Lumbosacral bracing – effective in reducing pain and facilitate increased walking distance. No evidence to suggest that results are maintained once brace removed.
The Danish Health Authority (Rousing et al, 2019) produced a clinical guideline established for the management of lumbar spinal stenosis in patients over the age of 65. In similarity with the NASS guidelines, surgical decompression is recommended for consideration in those experiencing severe symptoms for an extended period (3-6months), and if previous conservative treatment has been unsuccessful. In contrary to NASS, for patients with increased spinal instability, spinal fusion could be considered. The DHA highlights the importance of exercise for improvements to general health, however there is no evidence to implicate a positive impact on neurogenic pain. Additionally, due to the increased risk of secondary complications this patient population are placed at, the guidelines recommend against the use of NSAIDs.
In the contrary to the above two sets of guidelines, the Japanese Orthopaedic Association (JOA) clinical practice guidelines (Kawakami et al, 2022) highlighted the benefits of exercise therapy guided by physiotherapists in alleviating pain, improving function and improving quality of life, in patients with lumbar spinal stenosis. If conservative intervention fails as initial treatment, it is recommended that surgical decompression is considered in patients with severe symptoms, to increase cost-effectiveness, and produce better clinical outcomes.
Further treatment interventions recommended within these guidelines include:
- Surgical decompression with spinal fusion – beneficial to patients with spinal instability, though accompanies an increased cost and increased risk of complications.
- Nerve block injection – short term pain relief and improvements in quality of life were noted. Improved further when combined with steroids.
Figure 1
Table displaying clinical practice guidelines, their treatment recommendations and the plausibility of evidence.
| Clinical Practice Guideline | Treatment Recommendations | Plausibility of evidence |
| NASS (2011) | Physical therapy/exercise
Surgical decompression Caudal epidural injections Lumbosacral bracing |
low
high moderate low |
| DHA (2019) | Surgical decompression
Surgical decompression with spinal fusion Supervised exercise |
high
low very low |
| JOA (2021) | Exercise therapy
Surgical decompression Surgical decompression with spinal fusion Nerve block injection |
moderate
high moderate high |
Anderson et al (2021) highlighted that the clinical practice guidelines available lacked sufficient supporting evidence, however, they noted that injections and surgery prompted increased positive recommendations, with percentages 93.2% and 66% respectively, compared to non-surgical treatments (28.6%).
How is lumbar spinal stenosis currently managed?[edit | edit source]
Typically, the first line of treatment is a conservative approach; involving the use of physiotherapy, medication and injections.
Physiotherapy management
Recent evidence suggests that physiotherapists should develop a comprehensive training programme and involve manual therapy within the treatment sessions. A recent systematic review by Katz et al (2022) highlighted the benefit of physiotherapy compared to home exercise and self-directed training (figure 2).
Figure 2
Table by Katz et al (2022) displaying effectiveness of physiotherapy intervention.
A comprehensive training programme should include the following: (Bagley et al, 2019)
- Core strengthening and stability exercises
- Flexibility training
- Stability/balance exercises
- Aerobic exercise
The evidence for physiotherapy on the improvement of LSS patients in the long term is limited, however there is significant evidence which highlights the importance of physiotherapy post-surgical intervention (Bagley et al, 2019).
Medication and injections
- Pain relief medication – gabapentin and tramadol, there is evidence to suggest that when combine with rehabilitation, better outcomes are produced compared to rehabilitation alone (Kirker et al, 2023).
- Epidural steroid injections – moderate evidence to implicate that, when compared to surgery, epidural steroid injections are more effective in alleviating pain symptoms.
- NSAIDs – limited evidence on the benefits to pain relief, more randomised controlled trials are required to make conclusive recommendations (Katz et al, 2022).
Surgery
A prospective multicentre study by Burgstaller et al (2020) investigating long term effects of surgical vs non-surgical treatment in LSS patients highlighted the superiority of the surgical groups of participants, demonstrating reduced pain, improved function and improved quality of life.
Lumbar spinal decompression surgery is the most commonly utlised surgical intervention (Lurie and Tomkins-Lane, 2016), however there are different approaches: (Overdevest et al, 2014)
- Open bilateral laminectomy
- Minimally invasive laminectomy
- Endoscopic laminectomy
- Unilateral laminectomy
- Total laminectomy
However, surgery must only be considered if indicated, and contraindications must be examined for.
What are the contraindications for surgery?[edit | edit source]
In most cases of lumbar spinal stenosis, the outcome of treatment is not impacted by delayed surgery. Therefore, decisions must consider severity of symptoms and the clinician’s opinion, while ensuring the patient is educated with sufficient information to make an informed decision.
Contraindications for lumbar spinal surgery include: (Ju et al, 2022)
- Back pain with the absence of neurogenic symptoms
- Spinal instability that requires stabilisation
- Severe spinal infection
- Cauda equina syndrome
- Complex foraminal stenotic lesions
Core stabilisation exercise[edit | edit source]
When talking about core stability, this is referencing multiple groups of muscles that surrounding the lumbo-pelvic-hip complex. These muscles play a vital role in everyday life by: (Kibler, Press and Sciascia, 2006)
- Allowing bodies to remain stable by controlling the position and motion of the trunk over the pelvis
- Offload excessive loads from the spine
- Allows for the control of force transfer and motion along the kinetic chain
- Prevent injury
Main inner core muscles:
- Pelvic floor
- Transversus abdominis
- Internal abdominals obliques
- Multifidus
Main outer core muscles:
- Rectus abdominus
- External obliques
- Quadratus lumborum
- Glutes
These above muscles produce high torque to counterbalance external forces impacting the spine; thus, this group of muscles is secondarily responsible for maintaining spinal stability, aiding in improving function and reducing pain (Chang, Lin and Lai, 2015)
What's the aim?[edit | edit source]
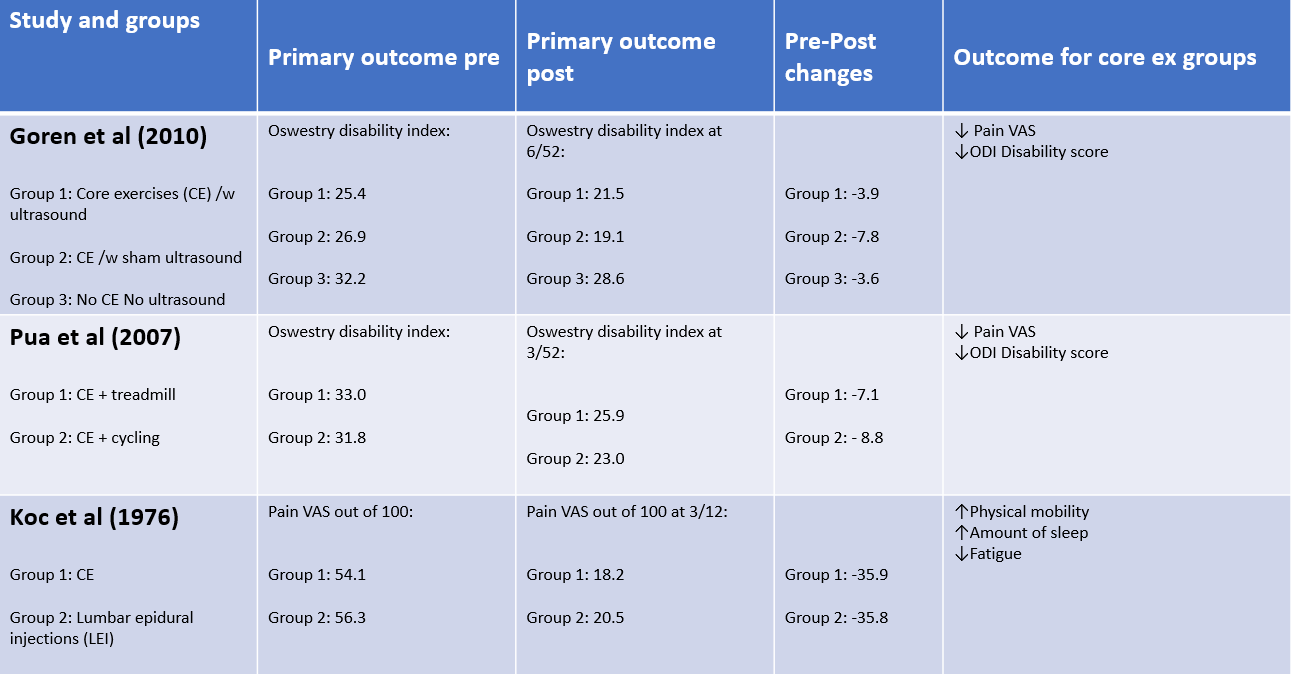
When treating patients with stenosis it’s important to keep in mind exercises does not directly alter the stenotic changes. The idea is that these exercises increase the support on the spinal column (Matsuwaka and Liem, 2018). Supporting the previous point, Goren et al., 2010 found positive effect on pain relief, muscle power, motor skill and improved daily activity from core stabilisation exercises.
Exercise in general for patients with lumbar pathologies gain much more than physiological benefits. Sculco et al., 2001 found clinically significant beneficial on psychosocial barriers such as anger, fear or confusion which prevent activities of daily living for patients with spinal stenosis.
Do core exercises improve outcomes in stenosis?[edit | edit source]
A SR review by (Slater et al., 2015) looks into perceived influence of exercise on pain and disability in patients with lumbar stenosis. This study includes 5 of 310 RCTS including 238 patients with lumbar stenosis and similar demographics. Methodological quality of all studies was assessed with PEDro averaging 6.2/10 with a range of 2-8 with only 1 study not having blinded subjects.
The 3 studies included that focused on core stability exercises are as follows:
What can be concluded from the overall study is that core exercises offer a low-cost clinically and statistical effective intervention for patients with stenosis who present with pain and reduced disability.
Does stenosis severity impact the effectiveness of core exercise?[edit | edit source]
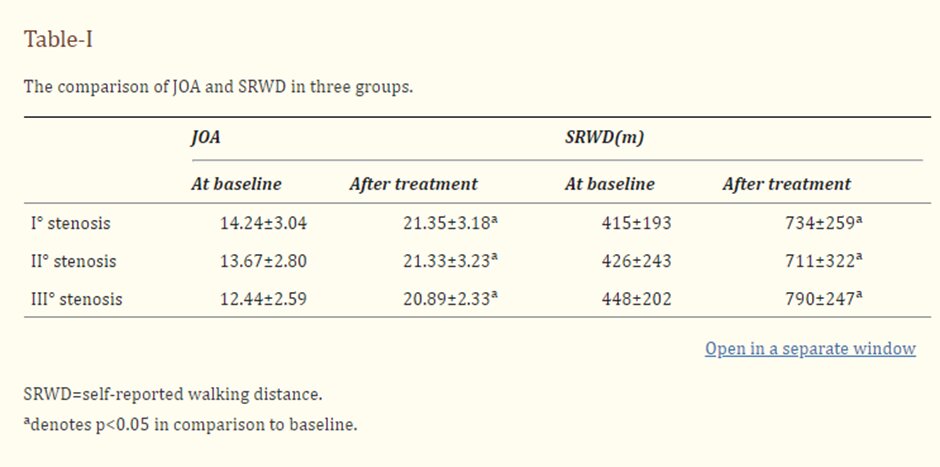
This question is explored by Chen et al., 2017, looking if core exercise effectiveness correlates with the severity of spinal stenosis.
This study included:
- 42 participants
- 3 groups - varying degree of spinal stenosis
- daily core stability exercises over 6 weeks
Outcomes:
- Japanese Orthopaedic association score (JAO)
- Self-reported walking capacity.
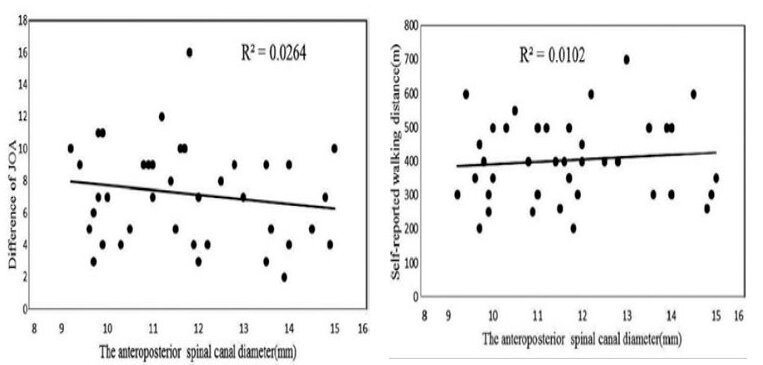
The outcome of the results of this study were able to confirm that the severity of stenosis diameter does not have any correlation to the effectiveness of core exercises when looking at disability and walking distance improvements. Understanding this allows us to vastly increase the scope of appropriate patients. It is however important to note that the severity of stenosis was determined by physiological factors alone without inclusion of psychosocial elements. Further study would be required to include severity of this demographic to further increase ecological validity of the trial.
What core stability exercises are used?[edit | edit source]
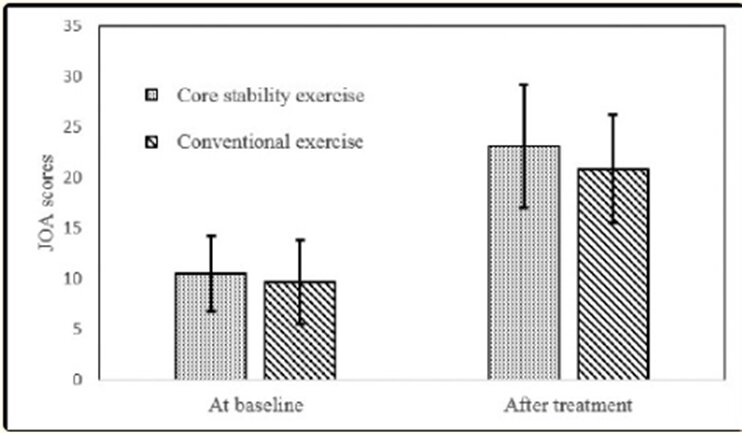
It is unfortunately common for studies to not publish in depth information of the exact exercise programmes they utilise in their studies. Due to this lack of information, there is no one consensus about which are the best set of exercises to use for patients with lumbar stenosis. Mu et al., 2018 published what exercises he used when comparing core exercises to conventional exercise in stenotic patients in an RCT containing 33 participants. Mu found that plank, side plank, bridge, and modified push-up all found to provide higher functional outcomes than the conventional group.
See https://www.physio-pedia.com/Core_Stability and https://www.physio-pedia.com/Core_Strengthening for a list of viable exercises and prescription methods.
Lumbar decompression surgery[edit | edit source]
Definition[edit | edit source]
Lumbar decompression surgery is a surgery to treat spinal stenosis by removing the tissues that compress the nerve to restore the space in the spinal canal and alleviate pain and improve function. It is an outpatients procedure performed by a neurosurgeon or an orthopaedic surgeon with experience in spinal surgery under general anaesthetic usually as an open surgery and takes over an hour to perform. The aim of the surgery is to create space around the compressed nerve which can be achieved by either laminectomy, discectomy, spinal fusion or a combination of these depending on the structure causing the compression.
Theory of lumbar decompression[edit | edit source]
The aim of the lumbar decompression surgery is to remove the structures causing pressure on the spinal cord and create space around the compressed nerve. Compression can be caused by a disc herniation, spondylosis, spondylolisthesis, arthritis, hypertrophied ligaments or tumour. By removing the compression the irritated nerve can recover.
Depending on the structure causing the compression, one or more procedures can be used:
- Laminectomy – a part of the lamina is removed
- Discectomy – a part of the disc is removed
- Spinal fusion – 2 or more vertebrae are fused together to prevent excessive movement
Criteria for surgery[edit | edit source]
Spinal stenosis is at first managed conservatively. Conservative management includes pain medication, physiotherapy and spinal injections. Surgery may be an options for those that struggle with ADLs or their quality of life is affected and it may be considered for those experiencing serious side effects of medication. It may be considered after around 4 months of failed conservative management. Patients would have to be healthy for surgery to be considered.
Risks of lumbar decompression surgery[edit | edit source]
Just like with any other surgery, there are potential complications including
- Infection
- Blood clot/DVT/pulmonary embolism
- Pain
- Bleeding
- Scarring
- Difficulty passing urine
- Chest infection
Complications of a lumbar decompression surgery includes
- Facial sores and loss of vision
- No relief of the symptoms – continued pain and numbness
- Symptoms return within a few years of the surgery
- Dural tear
- Leakage of cerebrospinal fluid
- Bleeding in the spinal column
- Damage to the spinal nerves or cord/nerve damage
- Loss of bladder or bowel control
- Paralysis
- Death
Risk factors include smoking and obesity. If a repeat surgery is needed, there is a higher risk of complications.
Evidence for efficacy of lumbar decompression surgery[edit | edit source]
The leg pain should improve immediately after surgery. Most patients are discharged after 1-4 days and it takes around 4-6 weeks to reach the expected level of mobility and function. Physiotherapy is completed after the operation to regain strength and movement. Most patients return to work in 4-8 weeks.
A Cochrane review (2016) including 24 RCTs amongst 2352 patients found that those who had decompression plus fusion didn’t have a better outcome than those who only had decompression surgery. There were no differences between different forms of decompression.
A study by Srinivas et al. (2019) looked at the improvement in lower back pain after lumbar decompression for spinal stenosis. 1221 patients were included in the analysis. Lower back pain significantly improved at 3 months and the improvements were sustained at 24 months. Those who didn’t use narcotics, had compensation claims or had an increased severity of lower back pain before surgery achieved better outcomes.
In a review by Sunderland et al. (2021) lumbar decompression surgery was found to be effective at improving quality of life. It was effective at improving leg pain and to a lesser extend back pain at 3 months which was sustained at the 2 year follow up.
A study by Anjarwalla, Brown and McGregor (2007) looked at the outcome of spinal decompression surgery for lumbar stenosis. 51 participants completed the 5 year follow up and significant improvement was noted in back and leg pain and physical function at 6 weeks, 1 year and 5 years, however some deterioration in pain levels were noted at 5 years but still a significant improvement on baseline levels and initially in social function, however those improvements diminished at the 5 year follow up.
Conclusion[edit | edit source]
Lumbar stenosis is a debilitating condition that can cause pain and reduced function, affecting the quality of life of millions of people worldwide. The two primary interventions for lumbar stenosis are core stability exercises and lumbar decompression surgery.
Core Stability Exercises:
Core stability exercises are an effective intervention for patients with lumbar stenosis. Such exercises aim to improve the function of the muscles that support the spine. They can improve psychosocial barriers such as fear or confusion, which prevent activities of daily living for patients with spinal stenosis. Studies have shown that core stability exercises can help reduce pain and improve mobility in patients with lumbar stenosis (Sculco et al., 2001)
Lumbar Decompression Surgery:
Lumbar decompression surgery is an option for those who have failed conservative management and struggle with activities of daily living. The surgery aims to remove structures causing pressure on the spinal cord and creates space around the compressed nerve. Although it is an effective intervention, it is not without risks. Risks of the surgery include infection, bleeding, scarring, and nerve damage.
Evidence:
Research has shown that lumbar decompression surgery can improve leg pain and quality of life (Anjarwalla, Brown and McGregor, 2007). However, it is worth noting that decompression plus fusion does not have a better outcome than decompression alone (Machado GC, 2016). It is essential to note that surgery is not always the best option for all patients. The decision to undergo surgery should be made on a case-by-case basis, considering the severity of the condition, the patient's age, and overall health.
Physiotherapy:
Physiotherapy is a vital component of post-op care for patients who undergo lumbar decompression surgery. It is essential to regain strength and movement after surgery and prevent any complications. Patients who undergo surgery should follow a comprehensive rehabilitation program that includes physiotherapy to regain their quality of life.
In conclusion, both core stability exercises and lumbar decompression surgery are effective interventions for patients with lumbar stenosis. However, the decision to undergo surgery should be made based on an individual's case and should be weighed against the risks and benefits of surgery. Physiotherapy is an essential component of post-op care and should be followed to regain movement and strength.
References[edit | edit source]
Aiki, H. et al. (2005) ‘Adjacent segment stenosis after lumbar fusion requiring second operation’, Journal of Orthopaedic Science, 10(5), pp. 490–495. Available at: https://doi.org/10.1007/s00776-005-0919-3.
Ammendolia, C. et al. (2017) ‘The physical and psychological impact of neurogenic claudication: The patients’ perspectives’, Journal of the Canadian Chiropractic Association, 61(1), pp. 18–31.
Anderson, D.B., De Luca, K., Jensen, R.K., Eyles, J.P., Van Gelder, J.M., Friedly, J.L., Maher, C.G. and Ferreira, M.L. (2021) ‘A critical appraisal of clinical practice guidelines for the treatment of lumbar spinal stenosis’, The Spine Journal, 21(3), pp.455-464. doi: https://doi.org/10.1016/j.spinee.2020.10.022
Bagley, C., MacAllister, M., Dosselman, L., Moreno, J., Aoun, S.G. and El Ahmadieh, T.Y. (2019) ‘Current concepts and recent advances in understanding and managing lumbar spine stenosis’, F1000Research, 8. doi: https://doi.org/10.12688/f1000research.16082.1
Burgstaller, J.M. et al. (2020) ‘Long-term results after surgical or nonsurgical treatment in patients with degenerative lumbar spinal stenosis: a prospective multicenter study’, Spine, 45(15), pp.1030-1038. doi: https://doi.org/10.1097/BRS.0000000000003457
Chang, W.-D., Lin, H.-Y. and Lai, P.-T. (2015). Core strength training for patients with chronic low back pain. Journal of Physical Therapy Science, [online] 27(3), pp.619–622. doi:https://doi.org/10.1589/jpts.27.619.
Cook, C.J. et al. (2020) ‘Systematic review of diagnostic accuracy of patient history, clinical findings, and physical tests in the diagnosis of lumbar spinal stenosis’, European Spine Journal, 29(1), pp. 93–112. Available at: https://doi.org/10.1007/s00586-019-06048-4.
Coppen, K. (2022) Spinal Stenosis - NHS TIMS, Spinal Stenosis. Available at: https://www.tims.nhs.uk/wp-content/uploads/2020/06/1.8-TIMS-Spinal-Stenosis.pdf
Fritz, J.M., Erhard, R.E. and Vignovic, M. (1997) ‘A nonsurgical treatment approach for patients with lumbar spinal stenosis’, Physical Therapy, 77(9), pp. 962–973. Available at: https://doi.org/10.1093/ptj/77.9.962.
Genevay, S. and Atlas, S.J. (2010) ‘Lumbar Spinal Stenosis’, Best Practice and Research: Clinical Rheumatology, 24(2), pp. 253–265. Available at: https://doi.org/10.1016/j.berh.2009.11.001.
Goren, A., Yildiz, N., Topuz, O., Findikoglu, G. and Ardic, F. (2010). Efficacy of exercise and ultrasound in patients with lumbar spinal stenosis: a prospective randomized controlled trial. Clinical Rehabilitation, [online] 24(7), pp.623–631. doi:https://doi.org/10.1177/0269215510367539.
Guen, Y.L. et al. (2011) ‘A new grading system of lumbar central canal stenosis on MRI: An easy and reliable method’, Skeletal Radiology, 40(8), pp. 1033–1039. Available at: https://doi.org/10.1007/s00256-011-1102-x.
Ho, C.H. et al. (2007) ‘Spinal Cord Injury Medicine. 1. Epidemiology and Classification’, Archives of Physical Medicine and Rehabilitation, 88(3 SUPPL.1), pp. 1–6. Available at: https://doi.org/10.1016/j.apmr.2006.12.001.
Jensen, R.K. et al. (2021) ‘Lumbar spinal stenosis’, The BMJ, 373, pp. 1–6. Available at: https://doi.org/10.1136/bmj.n1581.
Ju, C.I., Kim, P., Ha, S.W., Kim, S.W. and Lee, S.M. (2022) ‘Contraindications and Complications of Full Endoscopic Lumbar Decompression for Lumbar Spinal Stenosis: A Systematic Review’, World Neurosurgery, 168, pp.398-410.doi: https://doi.org/10.1016/j.wneu.2022.07.066
Kalichman, L. et al. (2009) ‘Spinal stenosis prevalence and association with symptoms: the Framingham Study’, Spine Journal, 9(7), pp. 545–550. Available at: https://doi.org/10.1016/j.spinee.2009.03.005.
Katz, J.N., Zimmerman, Z.E., Mass, H. and Makhni, M.C. (2022) ‘Diagnosis and management of lumbar spinal stenosis: a review’, Jama, 327(17), pp.1688-1699. doi: https://doi.org/10.1001/jama.2022.5921
Kawakami, M., Takeshita, K., Inoue, G., Sekiguchi, M., Fujiwara, Y., Hoshino, M., Kaito, T., Kawaguchi, Y., Minetama, M., Orita, S. and Takahata, M. (2022) ‘Japanese Orthopaedic Association (JOA) clinical practice guidelines on the management of lumbar spinal stenosis, 2021-Secondary publication’, Journal of Orthopaedic Science, 28, pp. 46-91. doi: https://doi.org/10.1016/j.jos.2022.03.013
Kibler, W.B., Press, J. and Sciascia, A. (2006). The Role of Core Stability in Athletic Function. Sports Medicine, [online] 36(3), pp.189–198. doi:https://doi.org/10.2165/00007256-200636030-00001.
Kirker, K., Masaracchio, M.F., Loghmani, P., Torres-Panchame, R.E., Mattia, M. and States, R. (2023) ‘Management of lumbar spinal stenosis: a systematic review and meta-analysis of rehabilitation, surgical, injection, and medication interventions’, Physiotherapy Theory and Practice, 39(2), pp.241-286. doi: https://doi.org/10.1080/09593985.2021.2012860
Kreiner, D.S. et al. (2014) ‘An evidence-based clinical guideline for the diagnosis and treatment of lumbar disc herniation with radiculopathy’, Spine Journal, 14(1), pp. 180–191. Available at: https://doi.org/10.1016/j.spinee.2013.08.003.
Kreiner, D.S., Shaffer, W.O., Baisden, J., Gilbert, T., Summers, J., Toton, J., Hwang, S., Mendel, R. and Reitman, C. (2011), ‘Diagnosis and treatment of degenerative lumbar spinal stenosis’, Boulevard, USA: North American Spine Soc. Available at: https://www.spine.org/Portals/0/Assets/Downloads/ResearchClinicalCare/Guidelines/LumbarStenosis.pdf [Accessed 16 May]
Lee, S.Y. et al. (2015) ‘Lumbar stenosis: A recent update by review of literature’, Asian Spine Journal, 9(5), pp. 818–828. Available at: https://doi.org/10.4184/asj.2015.9.5.818.
Lurie, J. and Tomkins-Lane, C. (2016) ‘Management of lumbar spinal stenosis’, The BMJ, 352. doi: https://doi.org/10.1136/bmj.h6234
Matsuwaka, S. and Liem, B. (2018). The Role of Exercise in Treatment of Lumbar Spinal Stenosis Symptoms. [online] doi:https://doi.org/10.1007/s40141-018-0171-3.
Miskin, N. et al. (2021) ‘Simplified Universal Grading of Lumbar Spine MRI Degenerative Findings: Inter-Reader Agreement of Non-Radiologist Spine Experts’, Pain Medicine (United States), 22(7), pp. 1485–1495. Available at: https://doi.org/10.1093/pm/pnab098.
Mu, W., Shang, Y., Mo, Z. and Tang, S. (2018). Comparison of two types of exercises in the treatment of lumbar spinal stenosis. Pakistan Journal of Medical Sciences, [online] 34(4). doi:https://doi.org/10.12669/pjms.344.15296.
Office for Health Improvement and Disparities (2022) Musculoskeletal health: Applying all our health, GOV.UK. Available at: https://www.gov.uk/government/publications/musculoskeletal-health-applying-all-our-health/musculoskeletal-health-applying-all-our-health
Overdevest, G.M., Moojen, W.A., Arts, M.P., Vleggeert-Lankamp, C.L., Jacobs, W.C. and Peul, W.C. (2014) ‘Management of lumbar spinal stenosis: a survey among Dutch spine surgeons’, Acta neurochirurgica, 156, pp.2139-2145. doi: https://doi.org/10.1007/s00701-014-2186-6
Rousing, R., Jensen, R.K., Fruensgaard, S., Strøm, J., Brøgger, H.A., Degn, J.D.M. and Andersen, M.Ø. (2019) ‘Danish national clinical guidelines for surgical and nonsurgical treatment of patients with lumbar spinal stenosis’, European Spine Journal, 28, pp.1386-1396. doi: https://doi.org/10.1007/s00586-019-05987-2
Schizas, C. et al. (2010) ‘Qualitative grading of severity of lumbar spinal stenosis based on the morphology of the dural sac on magnetic resonance images’, Spine, 35(21), pp. 1919–1924. Available at: https://doi.org/10.1097/BRS.0b013e3181d359bd.
Sculco, A.D., Paup, D.C., Fernhall, B. and Sculco, M.J. (2001). Effects of aerobic exercise on low back pain patients in treatment. The Spine Journal, [online] 1(2), pp.95–101. doi:https://doi.org/10.1016/s1529-9430(01)00026-2.
Slater, J., Kolber, M.J., Schellhase, K.C., Patel, C.K., Rothschild, C.E., Liu, X. and Hanney, W.J. (2015). The Influence of Exercise on Perceived Pain and Disability in Patients With Lumbar Spinal Stenosis. American Journal of Lifestyle Medicine, [online] 10(2), pp.136–147. doi:https://doi.org/10.1177/1559827615571510.
Tomkins-Lane, C. et al. (2016) ‘ISSLS prize winner: Consensus on the clinical diagnosis of lumbar spinal stenosis’, Spine, 41(15), pp. 1239–1246. Available at: https://doi.org/10.1097/BRS.0000000000001476.
Tseng, M.D. (2021) Spinal Stenosis. Available at: https://bestpractice.bmj.com/topics/en-gb/191 (Accessed: 18 May 2023).
Verbiest, H. (1954) ‘A radicular syndrome from developmental narrowing of the lumbar vertebral canal’, The Journal of bone and joint surgery. British volume, 36-B(2), pp. 230–237. Available at: https://doi.org/10.1302/0301-620x.36b2.230.
Walther, F. (2023) Lumbar Spinal Stenosis (grading): Radiology reference article, Radiopaedia Blog RSS. Available at: https://radiopaedia.org/articles/lumbar-spinal-stenosis-grading?lang=us
Yabuki, S. et al. (2013) ‘Prevalence of lumbar spinal stenosis, using the diagnostic support tool, and correlated factors in Japan: A population-based study’, Journal of Orthopaedic Science, 18(6), pp. 893–900. Available at: https://doi.org/10.1007/s00776-013-0455-5. [Original source: https://studycrumb.com/alphabetizer]