Complex Regional Pain Syndrome in the Foot
Top Contributors - Ewa Jaraczewska and Jess Bell
Introduction[edit | edit source]
Complex Regional Pain Syndrome (CRPS) is a clinical diagnosis of a syndrome. Its aetiology is multifactorial and it has several contributing factors. These include peripheral and central mechanisms and factors related to traumatic or surgical events. Successful outcomes depend on an early diagnosis and a comprehensive management approach, which includes pharmacological therapy, physiotherapy, therapeutic exercise, neurorehabilitation, and psychological and educational interventions for chronic pain management.[1]
History and Definition[edit | edit source]
History[edit | edit source]
Complex Regional Pain Syndrome is also known as algodystrophy.[2] Its history is summarised in an article by Iolascon et al.[1]
- Ambroise Paré (1510-1590) was the first to describe a condition that resembles the current concept of CRPS in the 16th century.
- British surgeon Alexander Denmark provided the first written description of CRPS in the 19th century.[1]
- Silas Weir Mitchell (1829-1914) discovered "causalgia" was a complication of gunshot wounds sustained by soldiers during the American Civil War.[1][3]
- Paul Sudeck (1866-1945) described acute inflammatory bone atrophy, known as "Sudeck’s atrophy.”
- In 1917, Rene Leriche (1879-1955) highlighted the key role of the sympathetic nervous system in the onset of the disease and came up with the term "sympathetic neuritis".[1]
- Around 1947, James A. Evans from Massachusetts, USA, renamed causalgia as "Reflex Sympathetic Dystrophy" (RSD) and supported the idea that the condition was caused by the sympathetic nervous system.[1]
- Philip S. Foisie (1896-1996) found that low-grade arterial spasms due to soft tissue injury could cause a severe pain syndrome with allodynia, oedema, muscle atrophy, osteoporosis, joint stiffness and reduced mobility. In 1948, he argued that RSD should be named ‘traumatic arterial vasospasm.’[1]
- In the 1950s, a new branch of anesthesiology called algology was born. An anesthesiologist, John J. Bonica (1917-1994), proposed the staging of CRPS.[1]
- The second consensus conference for The International Association for the Study of Pain (IASP) was held in 1993 in Orlando, Florida, to create shared criteria supporting the diagnosis of Reflex Sympathetic Dystrophy. Two forms of CRPS were defined: one form characterised by the evidence of apparent nerve damage (CRPS type II, corresponding to causalgia) and the second form without demonstrable nerve lesions (CRPS type I).[1]
- In 1999, two articles were published by two members of the IASP Task Force on CRPS: Norman Harden (Pain Medicine Specialist, Chicago, IL) and Stephen Bruehl (Pain Medicine Specialist, Nashville, TN). They proposed adding clinical signs to the diagnostic criteria of CRPS.[4]
- In 2003, during a medical conference in Budapest, a new classification for CRPS was proposed. [1]
Definition[edit | edit source]
CRPS is a "chronic pain condition characterized by autonomic and inflammatory features."[3] It usually begins in a distal extremity and is "disproportionate in magnitude or duration to the typical course of pain after similar tissue trauma."[3]
Classification[edit | edit source]
The International Association for the Study of Pain (IASP) offers the following final classification:[1]
- CRPS I (old name: reflex sympathetic dystrophy): used if the onset of symptoms have an uncertain history of a causative nerve injury.
- CRPS II (old name: causalgia): "defined earlier with electrodiagnostic or other definitive evidence of a significant nerve lesion."[1]
- CRPS-NOS (not otherwise specified): "partially meets CRPS criteria; not better explained by any other condition."[1]
The current classification for the diagnosis of CRPS relies solely on clinical findings.[1]
Pathophysiology[edit | edit source]
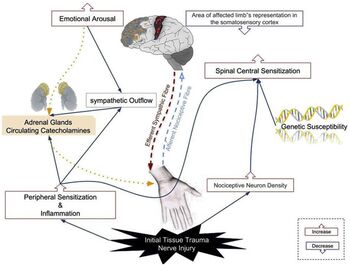
The aetiology of Complex Regional Pain Syndrome is multifactorial and it has several contributing factors.[1] It is a complex condition due to the involvement of the peripheral and central nervous systems.[5]
CRPS I can present differently in patients because of the various processes which contribute to its development. They include:
- Inflammatory mechanisms: patient presents with signs of inflammation, including heat, pain, redness, and swelling.[6]
- Altered cutaneous innervation: some studies suggest an initial nerve trauma is a significant trigger for the CRPS cascade.[6]
- The sympathetic nervous system: vasoconstriction causes skin discolouration and is considered a contributing factor to pain development.[6]
- Role of circulating catecholamines (hormones made by the adrenal glands and released into the body in response to physical or emotional stress.): higher sensitivity to circulating catecholamines.[6]
- Autoimmunity: presence of immunoglobulin G (IgG) autoantibodies in the serum of CRPS patients.[6]
- Brain plasticity: neuroimaging testing shows a decreased response in the region of the somatosensory cortex representing the CRPS-affected body. The level of sensory damage may be significantly correlated with the pain intensity and degree of hyperalgesia.[6]
- Genetic events: a genetic predisposition for developing CRPS was found in some family-based studies.[6]
- Psychological influence: there is a proposed linked between development of CRPS and psychological disorders, including anxiety and depression. [6]
Risk Factors[edit | edit source]
Higher rates of CRPS are associated with specific population and condition factors.
Population factors include:[7]
- Female gender
- Caucasian race
- Higher median household income
- Presence of comorbidities such as depression, drug abuse, and headache
- Male CRPS patients will likely have depression and kinesiophobia and use passive pain coping strategies[7]
Condition factors include:[7]
- Extremity injuries: fractures and sprains
- Surgical events: bunionectomy, tarsal tunnel release, and heel-spur surgery
- Blunt trauma to the foot with or without fracture or ankle sprain
- Fibromyalgia
- Rheumatoid arthritis
- Long-term disability
- Multiple pain diagnoses
CRPS is not only an adult condition. It also affects the paediatric and adolescent populations.
In the adolescent population, CRPS occurs more frequently in girls than boys. Blood work should be completed to rule out rheumatoid arthritis. Watch for risk factors, including stress at school, overachieving attitude, and inappropriate roles in the family, where the family focuses on the child's performance. It may take 6-8 months for children to recover from CRPS, and psychotherapy to help cope with this condition is a must.[8]
Diagnostic Criteria[edit | edit source]
CRPS I is diagnosed based on the modified Harden/Bruehl Criteria results, which became The Budapest Research Criteria. The diagnosis is confirmed when at least one sign in two of four categories and at least one symptom in each of the four categories are present, and "there should be no other condition able to explain the presented signs and symptoms":[6][9]
- Continuing pain is disproportionate to any inciting event.
- Must report at least one symptom in each of the four following categories:
- Sensory: report of hyperesthesia (i.e. the "increased sensitivity to stimulation, excluding the special senses"[10])
- Vasomotor: reports temperature asymmetry and/or skin colour changes and/or skin colour asymmetry.
- Sudomotor/oedema: reports oedema and/or sweating changes and/or asymmetry.
- Motor/trophic: reports decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin).
- Must display at least one sign in two of four of the following categories:
- Sensory: evidence of hyperalgesia (to pinprick) and/or allodynia (to light touch).
- Vasomotor: evidence of temperature asymmetry and/or skin colour changes and/or asymmetry.
- Sudomotor/oedema: evidence of oedema and/or sweating changes and/or perspiration asymmetry.
- Motor/trophic: evidence of the decreased range of motion and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin).
- There must be no other diagnosis that better explains the signs and symptoms.
Clinical Presentation[edit | edit source]
The key symptom of CRPS is prolonged and intense pain that can become constant. Patients usually describe the pain as burning, shooting, constant, stabbing, like an electric shock or it feels as if someone is squeezing the affected limb. The pain is out of proportion with the activities undertaken and it might spread to the entire leg.[6] Patients may often progress from an acute stage, where they have painful, warm, and oedematous limbs, to a chronic stage, where there is no warmth and oedema, but pain still affects function. Specific CRPS I characteristics in the foot include:
- Foot hypersensitivity - regular contact with the skin is excruciating, i.e. allodynia, which is defined as pain resulting from a stimulus, such as a light touch, that would not normally provoke pain.
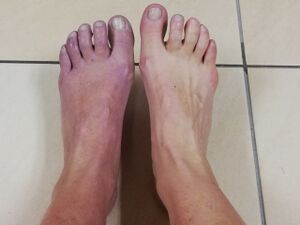
- Skin discolouration: the skin becomes purple and blotchy,[8] and it may appear shiny and thin.
- Changes in temperature due to abnormal microcirculation, which is caused by damage to the nerves controlling blood flow and temperature.[5]
- Lower leg oedema.[5]
- Great difficulty or inability to recruit the foot muscles.[6]
- Sensory abnormalities within the pathologic zones of the skin. These abnormalities are significantly different in patients, so there is no specific abnormality that can be used to diagnose CRPS I.[11]
- Functional impairment and disability due to nociceptive, vascular, and autonomic changes which exceed the expected clinical course of the inciting injury in proportion and duration.[6]
- Hypertrophic and later atrophic skin, along with nail texture and hair growth changes. Osteopenia may be observed in radiographic studies.[12]
- Ankle stiffness.[6]
Diagnostic Procedures[edit | edit source]
The current diagnosis of CRPS I is based on the physical examination and an analysis of the patient's history. However, certain diagnostic approaches have been adopted to assist with the diagnosis of CRPS. These include:[13]
- Plain film x-ray to identify bone loss.
- Three-phase bone scan for identifying bone loss or resorption by CRPS (these changes may appear only temporarily).
- Magnetic resonance imaging (MRI) allows for the examination of pathologic changes in musculoskeletal tissues (although its effectiveness has not been fully established).
Differential diagnosis[6][edit | edit source]
- Nerve lesion excluded by conduction velocity study
- Small fibre dysfunction (peripheral neuropathy affecting the small nerve fibres in the skin)
- Severe skin infections
- Chronic rheumatic Diseases
Management / Intervention[edit | edit source]
The following are the fundamental management principles for CRPS I:
- TEAM management includes chronic pain specialists, psychotherapists, orthopaedic surgeons, physiotherapists, and occupational therapists.[14]
- The patient's family must be on board from the beginning.[14]
- One size that does not fit all. The treatment approach must be personalised.[8]
- Rehabilitation should focus on the overall deficit of the affected and contralateral limb.[15]
- Slow, easy, short bursts of activities.[8]
- Find joy in activities. Find out what type of activities the patient would like to participate in.[8]
- Patient to keep a diary about progress.[8]
Goals[edit | edit source]
When establishing goals, the clinician must:[8]
- Collaborate with the patient regarding goal setting
- Choose achievable goals
- Manage the patient's expectations
Physiotherapy intervention should focus on:
- Pain management
- Quality of life improvement
The following are key components of the physiotherapy intervention:
- Patient and family education
- Desensitisation
- Strength and proprioceptive exercises
"You cannot move on with rehabilitation if you haven't dealt with the CRPS and that pain... And, you know, once the CRPS is dealt with, [the patient] will take off and they will do well." But prepare your patients for setbacks along the way. Helene Simpson
Education[edit | edit source]
- Learn about the patient's beliefs and fears[8]
- Review the patient's history of trauma[8]
- Avoid scary words[8]
- Assure the patient that you see them and hear them[8]
Desensitisation[edit | edit source]
- Touching, brushing, tapping, self-massage: firm pressure, but not too painful, one minute at a time.[8]
- Mirror therapy
Additional Therapeutic Interventions[edit | edit source]
Additional therapeutic interventions with mixed results:
- Low‐level laser therapy combined with individualised active and active assisted exercises, dosed up to pain threshold, demonstrated reduced pain at rest[23] - (low-quality evidence).[17]
- Exercise in combination with manual lymph drainage provided for six weeks, three times a week, to the affected lower limb shows a tendency towards greater pain reduction.[24]
Neuropsychological Changes[edit | edit source]
CRPS can be associated with complex neuropsychological changes that include:[25]
- Distortions in body representation
- Deficits in lateralised spatial cognition
- Non-spatially-lateralised higher cognitive functions
Cognitive changes in CRPS should be addressed separately and during therapy.[25]
Resources[edit | edit source]
- Perez RS, Zollinger PE, Dijkstra PU, Thomassen-Hilgersom IL, Zuurmond WW, Rosenbrand KC, Geertzen JH. CRPS I task force. Evidence-based guidelines for complex regional pain syndrome type 1. BMC Neurol. 2010 Mar 31;10:20.
- Harden RN, Oaklander AL, Burton AW, Perez RS, Richardson K, Swan M, Barthel J, Costa B, Graciosa JR, Bruehl S. Reflex Sympathetic Dystrophy Syndrome Association. Complex regional pain syndrome: practical diagnostic and treatment guidelines, 4th edition. Pain Med. 2013 Feb;14(2):180-229.
- Strauss S, Barby S, Härtner J, Pfannmöller JP, Neumann N, Moseley GL, Lotze M. Graded motor imagery modifies movement pain, cortical excitability and sensorimotor function in the complex regional pain syndrome. Brain Commun. 2021 Sep 25;3(4):fcab216.
References[edit | edit source]
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 Iolascon G, de Sire A, Moretti A, Gimigliano F. Complex regional pain syndrome (CRPS) type I: historical perspective and critical issues. Clin Cases Miner Bone Metab. 2015 Jan-Apr;12(Suppl 1):4-10.
- ↑ Giannotti S, Bottai V, Dell'Osso G, Bugelli G, Celli F, Cazzella N, Guido G. Algodystrophy: complex regional pain syndrome and incomplete forms. Clin Cases Miner Bone Metab. 2016 Jan-Apr;13(1):11-4.
- ↑ 3.0 3.1 3.2 Bruehl S. Complex regional pain syndrome. BMJ. 2015 Jul 29;351:h2730.
- ↑ Harden RN, Bruehl S, Galer BS, Saltz S, Bertram M, Backonja M, Gayles R, Rudin N, Bhugra MK, Stanton-Hicks M. Complex regional pain syndrome: are the IASP diagnostic criteria valid and sufficiently comprehensive? Pain. 1999 Nov;83(2):211-9.
- ↑ 5.0 5.1 5.2 Bruehl S. An update on the pathophysiology of complex regional pain syndrome. Anesthesiology. 2010 Sep;113(3):713-25.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 Eldufani J, Elahmer N, Blaise G. A medical mystery of complex regional pain syndrome. Heliyon. 2020 Feb 19;6(2):e03329.
- ↑ 7.0 7.1 7.2 Taylor SS, Noor N, Urits I, Paladini A, Sadhu MS, Gibb C, Carlson T, Myrcik D, Varrassi G, Viswanath O. Complex Regional Pain Syndrome: A Comprehensive Review. Pain Ther. 2021 Dec;10(2):875-892.
- ↑ 8.00 8.01 8.02 8.03 8.04 8.05 8.06 8.07 8.08 8.09 8.10 8.11 Simpson H. Complex Regional Pain Syndrome and the Foot. Plus 2022
- ↑ Mesaroli G, Hundert A, Birnie KA, Campbell F, Stinson J. Screening and diagnostic tools for complex regional pain syndrome: a systematic review. Pain. 2021 May 1;162(5):1295-1304.
- ↑ Maldonado RJ, De Jesus O. Hyperesthesia. [Updated 2021 Nov 15]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK563125/
- ↑ Kemler MA, Schouten HJ, Gracely RH. Diagnosing sensory abnormalities with either normal values or values from contralateral skin: comparison of two approaches in complex regional pain syndrome I. The Journal of the American Society of Anesthesiologists. 2000 Sep 1;93(3):718-27.
- ↑ David Clark J, Tawfik VL, Tajerian M, Kingery WS. Autoinflammatory and autoimmune contributions to complex regional pain syndrome. Mol Pain. 2018 Jan-Dec;14:1744806918799127
- ↑ Yoon D, Xu Y, Cipriano PW, Alam IS, Mari Aparici C, Tawfik VL, Curtin CM, Carroll IR, Biswal S. Neurovascular, muscle, and skin changes on [18F] FDG PET/MRI in complex regional pain syndrome of the foot: a prospective clinical study. Pain Medicine. 2022 Feb;23(2):339-46.
- ↑ 14.0 14.1 Harden RN, Oaklander AL, Burton AW, Perez RS, Richardson K, Swan M, Barthel J, Costa B, Graciosa JR, Bruehl S. Reflex Sympathetic Dystrophy Syndrome Association. Complex regional pain syndrome: practical diagnostic and treatment guidelines, 4th edition. Pain Med. 2013 Feb;14(2):180-229.
- ↑ Mouraux D, Lenoir C, Tuna T, Brassinne E, Sobczak S. The long-term effect of complex regional pain syndrome type 1 on disability and quality of life after a foot injury. Disability and rehabilitation. 2021 Mar 27;43(7):967-75.
- ↑ Cacchio A, De Blasis E, Necozione S, di Orio F, Santilli V. Mirror therapy for chronic complex regional pain syndrome type 1 and stroke. N Engl J Med. 2009 Aug 6;361(6):634-6.
- ↑ 17.0 17.1 17.2 Smart KM, Wand BM, O'Connell NE. Physiotherapy for pain and disability in adults with complex regional pain syndrome (CRPS) types I and II. Cochrane Database Syst Rev. 2016 Feb 24;2(2):CD010853.
- ↑ APTEI: CRPS Mirror Therapy. 2018 Available from: https://www.youtube.com/watch?v=7cwwTgMuJIw [last accessed 24/09/2022]
- ↑ Reflections, imagery, and illusions: the past, present and future of training the brain in CRPS. Available from https://www.iasp-pain.org/publications/relief-news/article/reflections-imagery-and-illusions-the-past-present-and-future-of-training-the-brain-in-crps/ [last access 21.09.2022]
- ↑ Strauss S, Barby S, Härtner J, Pfannmöller JP, Neumann N, Moseley GL, Lotze M. Graded motor imagery modifies movement pain, cortical excitability and sensorimotor function in complex regional pain syndrome. Brain Commun. 2021 Sep 25;3(4):fcab216.
- ↑ 21.0 21.1 Moseley GL. Graded motor imagery is a randomised controlled trial effective for long-standing complex regional pain syndrome. Pain. 2004 Mar;108(1-2):192-8.
- ↑ Neuro Orthopaedic Institute NOI: What is Graded Motor Imagery. 2014. Available from: https://www.youtube.com/watch?v=fWYUJscRBRw [last accessed 24/09/2022]
- ↑ Dimitrijevic IM, Lazovic MP, Kocic MN, Dimitrijevic LR, Mancic DD, Stankovic AM. [https://www.ftrdergisi.com/uploads/sayilar/286/buyuk/98-105.pdf Effects of low-level laser therapy and interferential current therapy in treating complex regional pain syndrome. Turkish Journal of Physical Medicine and Rehabilitation. 2014 Jun 1;60(2):98-106.
- ↑ Uher EM, Vacariu G, Schneider B, Fialka V. Manuelle Lymph drainage im Vergleich zur Physiotherapie bei Complex Regional Pain Syndrom Typ I. Randomisierte kontrollierte Therapievergleichsstudie [Comparison of manual lymph drainage with physical therapy in complex regional pain syndrome, type I. A comparative randomized controlled therapy study]. Wien Klin Wochenschr. 2000 Feb 11;112(3):133-7. German.
- ↑ 25.0 25.1 Halicka M, Vittersø AD, Proulx MJ, Bultitude JH. Neuropsychological Changes in Complex Regional Pain Syndrome (CRPS). Behav Neurol. 2020 Jan 14;2020:4561831.