Central Sensitisation
Original Editor - Nathan Benson as part of the PPA Pain Project.
Top Contributors - Alberto Bertaggia, Nathan Benson, Laura Ritchie, Jo Etherton, Shaimaa Eldib, Jess Bell, Bruno Serra, Uchechukwu Chukwuemeka, Kim Jackson, Lucinda hampton, Michelle Lee, Admin, Evan Thomas, WikiSysop, Claire Knott and Melissa Coetsee
Central Sensitisation[edit | edit source]
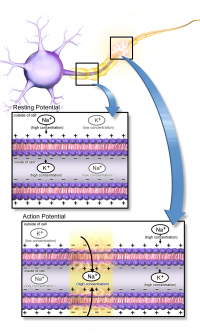
Nociception is described by IASP as the neural process of encoding noxious stimuli.
Central sensitisation is defined as an increased responsiveness of nociceptors in the central nervous system to either normal or sub-threshold afferent input[1] resulting in:
- Hypersensitivity to stimuli.[2]
- Responsiveness to non-noxious stimuli.[3]
- Increased pain response evoked by stimuli outside the area of injury, an expanded receptive field.[4].
Watch the 2 minute video below on central sensitisation.
The International Association for the Study of Pain (IASP) describes central sensitisation as
“Increased responsiveness of nociceptive neurons in the central nervous system to their normal or subthreshold afferent input”[5]
Neural plasticity plays a role in cellular changes with a demonstrable increase in both membrane excitability and synaptic efficacy.
The effect of this process is:
- Recruitment of additional, sub-threshold synaptic inputs to nociception resulting in a greater field of receptivity
- Increased output of nociception.
- The effects of this process may persist beyond the duration of the initial noxious input resulting in pain hypersensitivity to normally innocuous stimuli.
- Thought to play a role in affecting: pain facilitation and inhibition; inhibiting descending pathways[6]; over-activation of the ascending, and, pain facilitatory pathways[7].
- Simplified, it means too many messages going in and not enough coming out.
The use of the term "central sensitisation" varies referring
- Sometimes solely to the neural plastic changes
- Sometimes to the the complex and multiple processes and systems which contribute to changes in the elicitation and perception of pain[8][9][10].
- Term was first used in a study of pain hypersensitivity in rats following repeated noxious stimuli to describe the use dependent neural plasticity demonstrated[11][8]. These central changes due to peripheral noxious stimuli have been referred to as "activity-dependent central sensitisation"[2].
A discussion about the various descriptions and definitions can be found on the Body in MInd website.
Activity Dependent Central Sensitisation[edit | edit source]
Latremoliere and Woolf describe the changes demonstrated in their group's 1983 study as "activity dependent central sensitisation".
- Describes the mechanism of functional synaptic plasticity evoked in the dorsal horn neurons by input from nociceptors [2].
- Found that to invoke sensitisation, the noxious stimuli had to be sustained, intense and repeated.
- The changes can be divided into two, time dependent phases: an early, short duration phase which is phosphorylation-dependent/transcription-independent; a longer lasting transcription-dependent phase[12] (transcription is the process in which a gene's DNA sequence is copied (transcribed) to make an RNA molecule).[13]
Activation of the NMDA receptor is an essential step in initiating and maintaining the sensitisation (N-Methyl-D-Aspartate is a glutamate receptor. Glutamate is a widespread excitatory neurotransmitter in the nervous system).
- Under normal circumstances this receptor channel is blocked by Mg2+ ions[14].
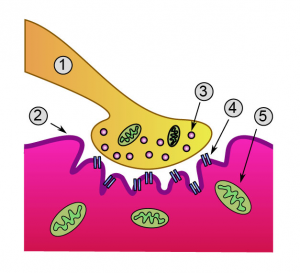
- Sustained release by nociceptors of glutamate, substance P and CGRP leads to membrane de-polarization, forcing the Mg2+ from the NMDA receptor[14].
- This rapidly boosts synaptic efficacy and allows Ca2+ in to the neuron, activating intracellular pathways and maintaining central sensitisation[2].
Central vs Peripheral Sensitisation[edit | edit source]
While descriptively central sensitisation and Peripheral Sensitisation may appear to be comparable processes, they represent quite distinct and processes and clinical features[2].
1. Peripheral sensitisation is described by the IASP as
"Increased responsiveness and reduced threshold of nociceptive neurons in the periphery to
the stimulation of their receptive fields."[15]
- It is initiated when the peripheral terminus of nociceptors are exposed to noxious stimulus, for example inflammatory mediators in damaged tissue. On going stimulation results in a lowering of the activation threshold and thus an increase in responsiveness of nociceptors [16].
- Generally requires on going peripheral pathology for the sensitisation to be maintained
- Generally localised to the site of injury[16].
- Has a role in altered heat sensation, but not in mechanical sensitivity[2].
2. Central sensitisation
- Novel inputs are recruited to the nociceptive pathways such as large, low-threshold mechanoreceptors classified as Aß fibres.
- Resulting in hypersensitivity in non-inflamed tissue and to touch.
| [17] |
Features of Central sensitisation[edit | edit source]
A survey of expert clinicians in a Delphi-derived survey found the following characteristics to describe central sensitisation in the clinical setting [18].
Subjective features[edit | edit source]
- Disproportionate, non-mechanical, unpredictable pattern of pain provocation in response to multiple/non-specific aggravating/easing factors.
- Pain persisting beyond expected tissue healing/pathological recovery times.
- Pain disproportionate to the nature and extent of injury or pathology.
- Widespread, non-anatomical distribution of pain.
- History of failed interventions (medical/surgical/therapeutic).
- Strong association with maladaptive psychosocial factors (e.g. negative emotions, poor self-efficacy, maladaptive beliefs, and pain behaviours, altered family/work/social life, medical conflict).
- Unresponsive to NSAIDs and/or more responsive to anti-epileptic (e.g. Lyrica) /anti-depressants (e.g. Amitriptyline) medication.
- Reports of spontaneous (i.e. stimulus independent) pain and/or paroxysmal pain (i.e. sudden recurrences and intensification of pain).
- Pain in association with high levels of functional disability.
- More constant/unremitting pain.
- Night pain/disturbed sleep.
- Pain in association with other dysesthesias (e.g. burning, coldness, crawling).
- Hyperpathia or pain of high severity and irritability (i.e. easily provoked, taking a long time to settle).
Clinical features[edit | edit source]
- Disproportionate, inconsistent, non-mechanical/non-anatomical pattern of pain provocation in response to movement/mechanical testing.
- Positive findings of hyperalgesia (primary, secondary) and/or allodynia and/or hyperpathia within the distribution of pain.
- Diffuse/non-anatomical areas of pain/tenderness on palpation.
- Positive identification of various psychosocial factors (e.g. catastrophisation, fear-avoidance behaviour, distress).
Identification in the Clinical Setting[edit | edit source]
In 2009 Schäfer et al.[19] proposed a classification of low back-related leg pain using an examination protocol which incorporates first the subjective assessment, including the Leeds Assessment of Neuropathic Symptoms and Sign (LANSS) scale [20], and second the physical examination (neurological examination, assessment of active movements, neural tissue provocation tests). Based on this comprehensive assessment, a LANSS score ≥ 12 is indicative of central sensitisation in their classification algorithm.
In 2010 Nijs et al.[21] provided guidelines to aid the recognition of central sensitisation on musculoskeletal patients.
In their paper, they suggest that a patient's medical diagnosis can offer insight into the likelihood of the presence of central sensitisation (fig 1) and this in conjunction with observable features (fig 2) can inform the therapist as to the presence of central sensitisation.
| Medical Diagnosis | Central sensitisation is a characteristic of this disorder | Central Sensitisation is present as a subgroup |
|---|---|---|
| Chronic lower back pain | X | |
| Chronic Whiplash associated disorders | X | |
| (Sub)acute whiplash associated disorders | X | |
| Tempromandibular disorders | X | |
| Myofascial pain syndrome | X | |
| Osteoarthritis | X | |
| Rheumatoid arthritis | X | |
| Fibromyalgia | X | |
| Chronic fatigue syndrome | X | |
| Chronic headache | X | |
| Irritable bowel syndrome | X |
| Symptom | Characteristic of central sensitisation | Might be related to Central sensitisation |
|---|---|---|
| Hypersensitivity to bright light | X | |
| Hypersensitivity to touch | X | |
| Hypersensitivity to noise | X | |
| Hypersensitivity to pesticides | X | |
| Hypersensitivity to mechanical pressure | X | |
| Hypersensitivity to medication | X | |
| Hypersensitivity to temperature (high or low) | X | |
| Fatigue | X | |
| Sleep disturbances | X | |
| Unrefreshing sleep | X | |
| Concentration difficulties | X | |
| Swollen feeling (e.g. in limbs) | X | |
| Tingling | X | |
| Numbness | X |
In 2012 Mayer et al.[22] proposed the Central Sensitisation Inventory (CSI). The clinical goal of this screening instrument is to help better assess symptoms thought to be associated with CS in order to aid physicians and other clinicians in syndrome categorzsation, sensitivity, severity identification, and treatment planning, to help minimize, or possibly avoid, unnecessary diagnostics and treatment procedures. CSI has showed good psychometric strength, the clinical utility, and the initial construct validity.
Management of Central Sensitisation[edit | edit source]
Central sensitisation is characterized by the absence of peripheral sources of nociceptive input, therefore it seems more appropriate to use a treatment with a top-down mechanism, activating descending nociceptive processing together with decreasing descending nociceptive facilitation[23].
In the video below, Prof Peter O'Sullivan discusses some of the myths about back pain which are widely held and negatively impact on the perception and treatment of back pain.
| [24] |
Non-Pharmacological Approaches[edit | edit source]
1.Patient Education
In cases of central sensitisation it is important to:
- change maladaptive illness perceptions,
- alter maladaptive pain cognitions,
- re-conceptualise pain.
This can be accomplished with pain physiology education, which is indicated when:
- the clinical picture is characterized and dominated by central sensitisation;
- maladaptive illness perceptions are present.
Face-to-face sessions of pain physiology education, in conjunction with written educational material, are effective for changing pain cognitions and improving health status in patients with various chronic musculoskeletal pain disorders (i.e. chronic low back pain, chronic whiplash, fibromyalgia and chronic fatigue syndrome)[25].
Check out the following video from Lorimer Moseley to see his approach to educating patients about managing pain.[26] [27]
2. Manual Therapy
Usually Manual Therapy is used for its peripheral effects, however it also produces central analgesic effects[28][29][30] activating descending anti-nociceptive pathways for a short period of time (30 - 35 mins.)[31][32]. This limits its clinical use in the management of central sensitisation.
- Some speculate that repeated sessions of Manual Therapy may result in a long term activation of descending anti-nociceptive pathways, there is no evidence of this mechanism available yet[23].
- On the contrary, Manual Therapy could also add peripheral nociceptive inputs, thus worsening the condition [23].
- Therefore, Manual Therapy should be delivered carefully.
3.Transcranial magnetic stimulation
Repetitive transcranial magnetic stimulation is more effective in suppressing centrally than peripherally originated pain states[33]. It provides short-term analgesic effects by stimulating the motor cortex or dorsolateral prefrontal cortex in various type of chronic pain patients[33][34][35]. However, the precise mechanism of action is still not clear, and the clinical utility of the technique is limited by practical obstacles (too short analgesic effects, availability of the equipment limited to few specialized centers)[23].
Pharmacological Approaches[edit | edit source]
A variety of pharmacological treatments have been trialed in patients with neuropathic pain,including conditions that are known to involve central sensitisation. However, some of these treatments are still under investigation and are not in widespread clinical use.
- Pharmacological agents such as non-steroidal anti-inflammatory drugs and coxibs have peripheral effects, and are therefore inappropriate for the treatment of central sensitisation in patients with chronic pain[36].
Often used drugs to treat central sensitisation include[23]:
- Acetaminophen (paracetamol) - primarily acts centrally reinforcing descending inhibitory pathways. In addition, it may exert an inhibitory action on the enzyme cyclooxygenase in the CNS
- Serotonin- and norepinephrine-reuptake inhibitors - activate noradrenergic descending pathways together with serotonergic pathways
- Opioids - activation of opioid receptors has inhibitory effects, including presynaptic inhibition of primary nociceptive afferents and postsynaptic inhibition of projecting neurons
- N-methyl-D-aspartate receptor blockers (i.e. ketamine) - blockade of excitation with NMDA-receptor antagonists may limit or reduce the spread of hyperalgesia and allodynia due to sensitisation and in consequence, NMDA-receptor antagonists may be seen preferentially as anti-hyperalgesic or anti-allodynic agents rather than as traditional analgesics
- Gabapentin/pregabalin (calcium channel alpha(2)delta ligands) - bind to the alpha(2)delta (a2d) subunit of voltage-sensitive Ca2+ channels, which sustain the enhanced release of pain transmitters at the synapses between primary afferent fibres and second-order sensory neurons under conditions of chronic pain
- Tramadol - centrally acting drug that induces anti-nociception in animals and analgesia in humans.
Resources[edit | edit source]
- "How to explain central sensitisation to patients with ‘unexplained’ chronic musculoskeletal pain: Practice guideline" - open access article can be found here.
References[edit | edit source]
- ↑ Louw A, Nijs J, Puentedura EJ. A clinical perspective on a pain neuroscience education approach to manual therapy. J Man Manip Ther. 2017; 25(3): 160-168.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Woolf CJ, Latremoliere A. Central Sensitization: A generator of pain hypersensitivity by central neural plasticity. The Journal of Pain 2009; 10(9):895-926
- ↑ Loeser JD, Treede RD. The Kyoto protocol of IASP basic pain terminology. Pain 2008;137: 473–7.
- ↑ Dhal JB, Kehlet H. Postoperative pain and its management. In:McMahon SB, Koltzenburg M, editors. Wall and Melzack's Textbook of pain. Elsevier Churchill Livingstone;2006. p635-51.
- ↑ International Association for the Study of Pain. IASP Terminology. Available from: https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1698#Sensitization. [Accessed 19 July 2020]
- ↑ Meeus M, Nijs J, Van der Wauwer N, Toeback L, Truijen S. Diffuse noxious inhibitory control is delayed in chronic fatigue syndrome: an experimental study. Pain 2008;139:439-48
- ↑ Meeus M, Nijs J. Central Sensitization: a biopyschosocial explanation for chronic widespread pain in patients with fibromyalgia and chronic fatigue syndrome. Clinical Rheumatology 2007; 26:465-73
- ↑ 8.0 8.1 Woolf CJ. What to call the amplification of nociceptive signals in the central nervous system that contribute to widespread pain? Pain 2014. Article in Press.
- ↑ Hansson P. PAIN 2014. http://dx.doi.org/10.1016/j.pain.2014.07.016. pii: S0304-3959(14)00335-2
- ↑ Body in Mind. Everything you wanted to know about CENTRAL SENSITISATION http://www.bodyinmind.org/central-sensitisation/ (accessed 10 June 2014)
- ↑ Woolf CJ.Evidence of a central component of post-injury pain hypersensitivity. Nature 1983;306;686-688.
- ↑ Woolf CJ, Saltar MW. Neuronal plasticity:increasing the gain in pain. Science 2000;288:1765-69.
- ↑ Khan academy stages of transcription Available from:https://www.khanacademy.org/science/biology/gene-expression-central-dogma/transcription-of-dna-into-rna/a/stages-of-transcription (last accessed 1.6.2020)
- ↑ 14.0 14.1 Mayer ML, Westbroke GL, Guthrie, PB. Voltage-dependant block by Mg2+ of NMDA responses in spinal cord neurones. Nature 1984;309:261-263.
- ↑ International Association for the Study of Pain. Pain Terms: A Current List with Definitions and Notes on Usage. http://iasp.files.cms-plus.com/Content/ContentFolders/Publications2/ClassificationofChronicPain/Part_III-PainTerms.pdf(accessed 12 July 2014
- ↑ 16.0 16.1 Hucho T, Levine JD. Signalling pathways in sensitization: Towards a nociceptor cell biology. Neuron 2007;55:365-376
- ↑ Danny Orchard. Peripheral and central sensitisation. Available from: http://www.youtube.com/watch?v=YwDMmSwUOOU [last accessed 22/11/15]
- ↑ Smart KM, Blake C,Staines A, Doody C. Clinical Indicators of "Nociceptive", "peripheral neuropathic", and "central sensitisation" as mechanisms based classifications of musculoskeletal pain. A Delphi survey of expert clinicians. Manual Therapy 2010;15:80-7
- ↑ Schäfer A, Hall T, Briffa K. Classification of low back-related leg pain—A proposed patho-mechanism-based approach. Manual Therapy. 2009 Apr 1;14(2):222–30.
- ↑ Bennett M. The LANSS Pain Scale: the Leeds assessment of neuropathic symptoms and signs. Pain. 2001 May;92(1-2):147–57.
- ↑ 21.0 21.1 Nijs J, Van Houdenhove B, Oostendorp RAB. Recognition of central sensitization in patients with musculoskeletal pain: application of pain neurophysiology in manual therapy practice. Manual Therapy 2010;15:135-41
- ↑ Mayer TG, Neblett R, Cohen H, Howard KJ, Choi YH, Williams MJ, et al. The Development and Psychometric Validation of the Central Sensitization Inventory (CSI). Pain Pract. 2012 Apr;12(4):276–85.
- ↑ 23.0 23.1 23.2 23.3 23.4 Nijs J, Meeus M, Van Oosterwijck J, Roussel N, De Kooning M, Ickmans K, et al. Treatment of central sensitization in patients with ‘unexplained’ chronic pain: what options do we have? Expert Opin Pharmacother. 2011 May;12(7):1087–98.
- ↑ Pain-Ed.com. Prof Peter O'Sullivan - Back pain - separating fact from fiction. Available from: http://www.youtube.com/watch?v=dlSQLUE4brQ[last accessed 22/11/15]
- ↑ Nijs J, Wilgen CP van, Oosterwijck JV, Ittersum M van, Meeus M. How to explain central sensitization to patients with ‘unexplained’ chronic musculoskeletal pain: Practice guidelines. Manual Therapy. 2011 Oct 1;16(5):413–8.
- ↑ Moseley L. Tame the Beast. Available at https://www.tamethebeast.org/#tame-the-beast (accessed 2 Mar 2020).
- ↑ Lorimer Moseley. Tame the Beast. Available from https://www.youtube.com/watch?time_continue=295&v=ikUzvSph7Z4&feature=emb_logo [last accessed 09/03/2020]
- ↑ Vicenzino B, Collins D, Wright A. The initial effects of a cervical spine manipulative physiotherapy treatment on the pain and dysfunction of lateral epicondylalgia. Pain. 1996 Nov;68(1):69–74.
- ↑ Bialosky JE, Bishop MD, Robinson ME, Zeppieri G, George SZ. Spinal manipulative therapy has an immediate effect on thermal pain sensitivity in people with low back pain: a randomized controlled trial. Phys Ther. 2009 Dec;89(12):1292–303.
- ↑ Bialosky JE, Bishop MD, Robinson ME, Barabas JA, George SZ. The influence of expectation on spinal manipulation induced hypoalgesia: an experimental study in normal subjects. BMC Musculoskelet Disord. 2008;9:19.
- ↑ Moss P, Sluka K, Wright A. The initial effects of knee joint mobilization on osteoarthritic hyperalgesia. Man Ther. 2007 May;12(2):109–18.
- ↑ Sluka KA, Skyba DA, Radhakrishnan R, Leeper BJ, Wright A. Joint mobilization reduces hyperalgesia associated with chronic muscle and joint inflammation in rats. J Pain. 2006 Aug;7(8):602–7.
- ↑ 33.0 33.1 Leung A, Donohue M, Xu R, Lee R, Lefaucheur J-P, Khedr EM, et al. rTMS for suppressing neuropathic pain: a meta-analysis. J Pain. 2009 Dec;10(12):1205–16.
- ↑ Leo RJ, Latif T. Repetitive transcranial magnetic stimulation (rTMS) in experimentally induced and chronic neuropathic pain: a review. J Pain. 2007 Jun;8(6):453–9.
- ↑ Lefaucheur JP. The use of repetitive transcranial magnetic stimulation (rTMS) in chronic neuropathic pain. Neurophysiol Clin. 2006 Jun;36(3):117–24.
- ↑ Kosek E. Medical management of pain.Chapter 12. In: Sluka K. Mechanisms And management of pain for the physical therapist. IASP press, Seattle;2009. p. 231-55