Au-Kline Syndrome
Top Contributors - Rucha Gadgil, Reem Ramadan and Uchechukwu Chukwuemeka
Introduction[edit | edit source]
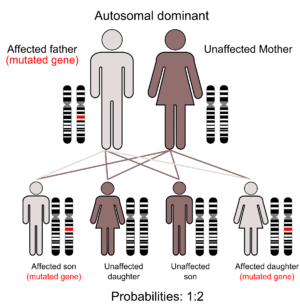
Au–Kline syndrome (AKS) which is synonymous with Okamoto syndrome, is a multiple congenital malformation syndrome mainly associated with intellectual disability. It can be categorized as a very rare autosomal dominant genetic condition.
Okamoto syndrome was first described in 1997 by Nobuhiko Okamoto et al. in Japan after observing similar symptoms and physical features in two unrelated Japanese infants[1]. Au–Kline syndrome was first described in 2015 by Ping-Yee Billie Au, Antonie D. Kline et al. after mutations in HNRNPK were found in two individuals with similar symptoms at their respective practices in Calgary, Alberta, Canada, and Baltimore, Maryland amd the United States. Both submitted the gene as a candidate to the online service GeneMatcher, which matched them together and allowed them to confirm the syndrome[2].
Clinically Relevant Anatomy[edit | edit source]
Genes are responsible for encoding proteins that in return dictate cell function and coordinate our biological functions. One of these proteins is called the Heterogeneous Nuclear Ribonucleoprotein K (HnRNP K) which is encoded by the Heterogeneous Nuclear Ribonucleoprotein K (HNRNPK), binds DNA or RNA and helps control the activity of genes and the production of proteins, helps coordinate transcriptional responses to DNA damage and plays an important role in the normal development and function of several body systems, including the brain[3].
Etiology[edit | edit source]
AKS is caused by mutations in the HNRNPK gene located on chromosome 9 at position q21.32, which leads to little or no production of HnRNP K protein. This altered gene activity and protein production disrupt the normal development and functioning of several body systems including the brain leading to intellectual disability, delayed development, and other neurological problems in people with the condition.
It is genetically inherited in an autosomal dominant pattern. Most cases of this condition result from new (de novo) mutations in the gene that occurs during the formation of reproductive cells (eggs or sperm) in an affected individual’s parent or in early embryonic development. These cases can occur in people with no history of the disorder in their family[4].
Clinical Presentation[edit | edit source]
This condition has been characterized by:
- Congenital hydronephrosis,
- Low muscle tone and reduced reflexes
- Heart defects: like aortic valve stenosis, atrial or ventricular septal defect, bicuspid aortic valve or patent ductus arteriosus.
- Intellectual disability
- Characteristic facial features: prominent, downturned ears, an open, downturned mouth, and drooping eyelids (ptosis)
- Neurological and skeletal abnormalities
- Urinary tract infections
- Language and walking
- Reduced growth: low weight and size[2]
Diagnostic Procedures[edit | edit source]
Formal diagnostic criteria for AKS have not been established however it can be suspected based on clinical and medical imaging findings. It should be suspected upon clinical observation where the patient demonstrates characteristic facial features such as ptosis, shallow orbits, broad nose, deeply grooved tongue and downturned mouth. In addition to that, presence of skeletal anomalies, presence of palate abnormalities and genitourinary anomalies[5]. Furthermore, medical imaging such as MRI aids in the diagnosis by helping identify anomalies in the brain and various radiographs of the spine to identify vertebral defects and spinal syrinx especially if scoliosis is present[6]. Genetic testing can be done to confirm the diagnosis by whole exome sequencing, and comparative genomic hybridization (for microdeletions). Sanger sequencing can confirm the nature of the mutation [7].
Management[edit | edit source]
The management depends on the symptoms of each patient. For example, to help with hypotonia and functional growth problems, physical therapy sessions may be helpful. In addition to that, if the patient is suffering from dysautonomia, the patient is referred to a neurologist to help with the autonomic dysfunction who at the same time offers various strategies to help with uncontrollable sweating and the patient's heat intolerance. Furthermore, due to the nature of the syndrome and it affecting the patient's intellectual abilities occupational therapists, speech therapists and special educators are needed for the implementation of special education programs using sign language, assistive devices and hearing aids. Genetic counselling is an important aspect of management for this condition. Prenatal testing for pregnancies at increased risk is possible if the HNRNPK pathogenic variant in the family is known. The prognosis of the condition is yet vague due to a handful of diagnosed individuals[7].
Differential Diagnosis[edit | edit source]
There are four main syndromes to consider in the differential diagnosis of Au-Kline syndrome. One of which is Kabuki syndrome which is distinguished from AKS by the physical characteristics of the patient which include arched and broad eyebrows, a depressed nasal tip and a cleft lip. In addition to that, the patient would be suffering from immunodeficiency and craniosynostosis is rare[8]. Another is Shprintzen-Goldberg syndrome which is distinguished from AKS by the absence of typical AKS facial features, marfanoid body habitus and arachnodactyly[9]. Noonan syndrome is another which can be distinguished from AKS also by the absence of AKS facial features, the presence of hypertrophic cardiomyopathy, the presence of coagulation abnormalities and myelodysplasia[10]. Also, Simpson-Golabi-Behmel syndrome type 1 which is different that AKS by its prevalence in males, the absence of AKS typical facial features, the occurrence of prenatal and postnatal overgrowth and presence of macrostomia[11].
References[edit | edit source]
- ↑ Wallerstein R, Rhoads F. Natural history of a child with Okamoto syndrome. Clinical Dysmorphology. 2013 Jul 1;22(3):127-8.
- ↑ 2.0 2.1 Au, P; Innes, M; Kline,A.; Ardinger, Holly H.; Pagon, Roberta A.; Wallace, Stephanie E. (eds.), "Au-Kline Syndrome", GeneReviews®, University of Washington, Seattle, 2019, PMID 30998304
- ↑ Mikula M, Ostrowski J. HNRNP K (heterogeneous nuclear ribonucleoprotein K). Atlas Genet Cytogenet Oncol Haematol. 2010;14(2):127-9.
- ↑ Au PYB, You J, Caluseriu O, Schwartzentruber J, Majewski J, Bernier FP, et al. GeneMatcher aids in the identification of a new malformation syndrome with intellectual disability, unique facial dysmorphisms, and skeletal and connective tissue abnormalities caused by de novo variants in HNRNPK. Hum Mutat. 2015;36:1009–14. doi: 10.1002/humu.22837.
- ↑ Pua HH, Krishnamurthi S, Farrell J, Margeta M, Ursell PC, Powers M, Slavotinek AM, Jeng LJ. Novel interstitial 2.6 Mb deletion on 9q21 associated with multiple congenital anomalies. American Journal of Medical Genetics Part A. 2014 Jan;164(1):237-42.
- ↑ Lange L, Pagnamenta AT, Lise S, Clasper S, Stewart H, Akha ES, Quaghebeur G, Knight SJ, Keays DA, Taylor JC, Kini U. A de novo frameshift in HNRNPK causing a Kabuki‐like syndrome with nodular heterotopia. Clinical genetics. 2016 Sep;90(3):258-62.
- ↑ 7.0 7.1 Au, P. Y., Goedhart, C; Ferguson, M; Breckpot, J; Devriendt, K; Wierenga, K; et al. "Phenotypic spectrum of Au-Kline syndrome: a report of six new cases and review of the literature". European Journal of Human Genetics. Sept. 2018. 26 (9): 1272–1281. doi:10.1038/s41431-018-0187-2. ISSN 1476-5438. PMC 6117294. PMID 29904177
- ↑ Dugan S. Kabuki syndrome. Cassidy and Allanson's management of genetic syndromes. 2021 Feb 19:529-38.
- ↑ Bari A, Sadaqat N, Nawaz N, Bano I. Shprintzen-Goldberg Syndrome: A Rare Disorder. Journal of the College of Physicians and Surgeons--pakistan: JCPSP. 2019 Jun 1;29(6):S41-2.
- ↑ Allanson JE. Noonan syndrome. Journal of medical genetics. 1987 Jan;24(1):9.
- ↑ Sajorda BJ, Gonzalez-Gandolfi CX, Hathaway ER, Kalish JM. Simpson-golabi-behmel syndrome type 1.