Soft Tissue Healing
Original Editor - Bo Hellinckx
Top Contributors - Andeela Hafeez, Wanda van Niekerk, Kim Jackson, Lucinda hampton, Leana Louw, Bo Hellinckx, George Prudden, WikiSysop, Claire Knott, Admin, Jess Bell, 127.0.0.1 and Tania Appelmans
Introduction[edit | edit source]
After an injury, soft tissue structures in the body undergo a natural healing process through specific phases of healing.
- The timeline for healing depends on: the individual; the extent of the injury; age; overall health status.
- Physiotherapy helps facilitate healthier healing - resulting in a smaller risk of re-injury, chronic pain and dysfunction.
- One of the main risks of future injury is how the soft tissue was rehabilitated or recovered, from previous injury/ surgery.
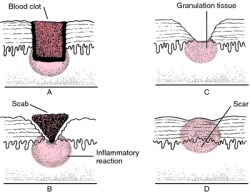
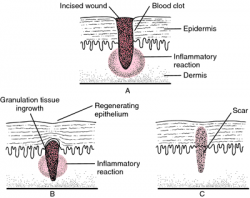
Soft tissue healing is defined as the replacement of destroyed tissue by living tissue in the body.[1] This process consists of two parts - regeneration and repair.[2] Note - There are no defined boundaries between stages as the wound healing response “transitions” into the next stage of healing[3].
- During the regeneration component, specialized tissue is replaced by the proliferation of surrounding undamaged specialized cells.
- In the repair component, lost tissue is replaced by granulation tissue which matures into scar tissue.[2]
The cellular reaction after injury depends on the tissue type as well as the extent of the wound.
- In injury to CNS tissue that damages neurons and the supporting glial cells, the body’s response is unforgiving, as regeneration of lost neurons is not possible. Activated astrocytes wall off the lesion, creating a glial scar.
- In contrast, in non-CNS tissue, a single tissue type can have multiple responses depending on the magnitude of injury.[3]
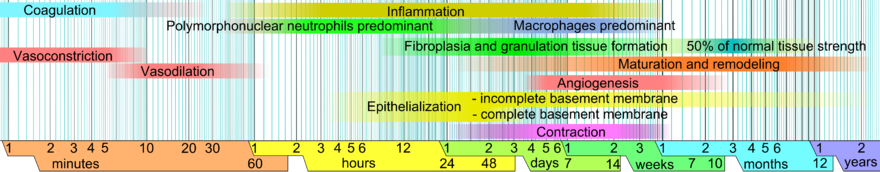
Phases of Healing[edit | edit source]
The different healing phases are not mutually exclusive and tend to overlap quite a lot.[2]
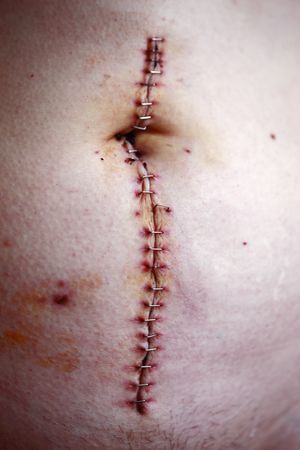
Bleeding Phase[edit | edit source]
Short phase immediately after injury - lasting about 6-8 hours, up to 24 hours after a crush injury.
- Time of the bleeding will depend on the extent of the soft tissue injury and the management thereof.
- The more vascular the injured structures, the more bleeding will occur.[2]
Inflammation Phase[edit | edit source]
The goal of the inflammation phase is to stop the bleeding phase.
- This phase starts rapidly within a 6-8 hours after the soft tissue injury, reaches the maximal reaction between 1-3 days and gradually resolves in a few weeks.[2]
- Achieved by vasoconstriction, retraction of injured blood vessels, fibrin deposition and clotting.
- The blood supply to the area increases in this time, causing oedema and redness.
- Phagocytosis ("the engulfing and usually the destruction of particulate matter by phagocytes that serves as an important bodily defence mechanism against infection by microorganisms and against occlusion of mucous surfaces or tissues by foreign particles and tissue debris[4]") happens during this phase.[2]
- The acute inflammatory response involves activities that generate exudates - plasma-like fluid that exudes out of tissue or its capillaries and is composed of protein and granular leukocytes (white blood cells).[5]
- The chronic inflammatory response is of prolonged duration and involves the presence of nongranular leukocytes and the production of scar tissue.
The acute phase involves three mechanisms that act to stop blood loss from the wound:[5]
- Local vasoconstriction occurs, lasting a few seconds to as long as 10 minutes. Larger vessels constrict due to neurotransmitters and capillaries and smaller arterioles and venules constrict due to the influence of serotonin and catecholamines released from platelets. The resulting reduction in the volume of blood flow in the region promotes increased blood viscosity or resistance to the flow, which further reduces blood loss at the injury site.
- The platelet reaction provokes clotting as individual cells irreversibly combine with each other and with fibrin to form a mechanical plug that occludes the end of a ruptured blood vessel. The platelets also produce an array of chemical mediators in the inflammatory phase: serotonin, adrenaline, noradrenaline, and histamine. Also, ATP is used for energy in the healing process.
- Fibrinogen molecules are converted into fibrin for clot formation.
Approximately 1-hour post-injury, swelling or oedema, occurs as the vascular walls become more permeable and increased pressure within the vessels forces a plasma exudate out into the interstitial tissues. This happens in:
- Mild trauma - for a few minutes with a return to normal permeability in 20-30 minutes.[5]
- More severe traumas - can result in a prolonged state of increased permeability, and sometimes result in delayed onset of increased permeability, with swelling not apparent until some time has elapsed since the original injury.
Bradykinin, a major plasma protease present during inflammation, increases vessel permeability and stimulates nerve endings to cause pain[5].
Proliferation Phase[edit | edit source]
This phase starts between 24-48 hours after injury, lasts up to 2-3 weeks when the bulk of the scar tissue is formed.[2]
Fibroplasia and Granulation Tissue Formation[edit | edit source]
- The central event during the proliferative phase.
- Occurs 3-5 days following an injury and overlaps with the preceding inflammatory phase.
- Granulation tissue includes inflammatory cells, fibroblasts, and neovasculature in a matrix of fibronectin, collagen, glycosaminoglycans, and proteoglycans.[6]
Epithelialization[edit | edit source]
Formation of epithelium over a denuded surface. The process begins within hours of tissue injury.
- Involves the migration of cells at the wound edges over a distance of less than 1 mm, from one side of the incision to the other. Incisional wounds are epithelialized within 24-48 hours after injury. This epithelial layer provides a seal between the underlying wound and the environment.[6]
- Epidermal cells at the wound edges undergo structural changes, allowing them to detach from their connections to other epidermal cells and to their basement membrane. Intracellular actin microfilaments are formed, allowing the epidermal cells to creep across the wound surface.
- Occlusive and semiocclusive dressings applied in the first 48 hours after injury may maintain tissue humidity and optimize epithelialization.[6]
- When epithelialization is complete, the epidermal cell assumes its original form,[6]
Fibroplasia[edit | edit source]
Fibroplasia begins 3-5 days after injury and may last as long as 14 days.
- Skin fibroblasts and mesenchymal cells differentiate to perform migratory and contractile capabilities.
- Fibroblasts are responsible for the production of collagen, elastin, fibronectin, glycosaminoglycans, and proteases.
- Fibroblasts fill the defect left by an open wound as the number of inflammation cells decrease.[6]
- As granulation tissue matures, the fibroblasts produce less type III collagen and become more spindly in appearance. They begin to produce the much stronger type I collagen
Angiogenesis[edit | edit source]
Angiogenesis results in greater blood flow to the wound and, consequently, increased perfusion of healing factors. Angiogenesis ceases as the demand for new blood vessels ceases. New blood vessels that become unnecessary disappear by apoptosis.[6]
- A rich blood supply is vital to sustaining newly formed tissue (as is appreciated in the erythema of a newly formed scar).
- The macrophage is essential to the stimulation of angiogenesis and produces macrophage-derived angiogenic factor in response to low tissue oxygenation.[6]
Contraction[edit | edit source]
- Contraction results in a decrease in wound size eg a 2-cm incision may measure 1.8cm after contraction.
- Loose tissues contract more than tissues with poor laxity, and square wounds tend to contract more than circular wounds.
- Wound contraction depends on the myofibroblast located at the periphery of the wound, its connection to components of the extracellular matrix, and myofibroblast proliferation.[6]
- Radiation and drugs, which inhibit cell division, have been noted to delay wound contraction.
Remodelling Phase[edit | edit source]
This phase starts around the peak of the proliferation phase. The result of this phase is an organised, quality and functional scar similar to the tissue it is busy repairing.[2]
The ultimate endpoint following remodelling depends on the tissue type.
- Non-central nervous system (CNS) tissue that undergoes primary healing, very little remodelling occurs because of the lack of extracellular matrix produced during the repair. Secondary healing, in contrast, involves fibre alignment and contraction to reduce the wound size and to reestablish tissue strength. Complete recovery of original tissue strength is rarely obtained in secondary healing because repaired tissue remains less organized than non-injured tissue, which results in scar formation.[6] Collagen-rich scars are characterized morphologically by a lack of specific organization of cellular and matrix elements that comprise the surrounding uninjured tissue.
- CNS tissue - there is no repair or regeneration of injured neurons, relatively little reestablishment of structural integrity in the region. During CNS remodelling, activated astrocytes wall off the lesion, creating a glial scar. These activated astrocytes may prevent further tissue damage, although neuron axonal regrowth is inhibited.[3][6]
Types[edit | edit source]
Primary Intention[edit | edit source]
Restoration of continuity occurs directly by fibrous adhesion, without the formation of granulation tissue; it results in a thin scar.[7]
Secondary Intention[edit | edit source]
Wound healing occurs by union by adhesion of granulating surfaces when the edges of the wound are far apart and cannot be brought together. Granulations form from the base and sides of the wound toward the surface.[7]
Tertiary Intention[edit | edit source]
Wound healing occurs by the gradual filling of a wound cavity by granulations and a cicatrix.[7]
Physiotherapy Management[edit | edit source]
This classification is based on a treatment protocol of Clanton et al.[8], but it is similar to other classifications. It is possible that some phases overlap, dependable on the individual response to healing and the type of injury. Not every patient undergoes all phases to achieve full rehabilitation.
Phase 1: Acute Phase (1 - 7 Days)[edit | edit source]
- Goal: Minimize inflammation and pain.
- Treatment:
- RICE-method: Rest, ice, compression and elevation
- Pain-free range of motion with cryotherapy
Phase 2: Subacute Phase (Day 3 - < 3 Weeks)[edit | edit source]
This phase starts when signs of inflammation begin to reduce. Inflammation signs are heat, swelling, redness and pain.
- Goal: Prevent muscle atrophy
- Treatment:
- Pain-free full range of motion: concentric strengthening
- If any pain present: decrease the intensity of exercises
Phase 3: Remodelling Phase: ( 1 - 6 Weeks)[edit | edit source]
- Stretching to avoid a decrease in flexibility
- Eccentric strengthening
- It is important to make sure that the muscle is already regenerated, to prevent risk of re-injury
Phase 4: Functional Phase: (2 Weeks - 6 Months)[edit | edit source]
- Goal: Return to sport without re-injury.
- Treatment:
- Increase their strength, endurance, speed, agility, flexibility and proprioception
- Sport-specific activities
Phase 5: Return to Competition Phase: (3 Weeks to 6 Months)[edit | edit source]
- Goal: Avoid a re-injury
- Criteria: Full range of motion, strength, coordination and psychological readiness
- Treatment:
- Address deficits in criteria
- Progressive agility and trunk stabilization[9]
Clinical Bottom Line[edit | edit source]
Soft tissue healing is a natural process that occurs in the body after an injury. This process happens without the need for medication and therapy, but this can play an important role in cases where problems are identified in this natural process, such as repeated trauma, inhibited response or delayed reactions. The aim would then be to facilitate and stimulate the soft tissue healing process.[2] It is also important to realise that inappropriate therapy will inhibit these events. It is thus very important to be selective of the most appropriate therapy at each stage.
Reference[edit | edit source]
- ↑ Walter JB, Israel MS. General Pathology, Churchill Livingstone. Six. Ed., Edinburg. 1987:151-3.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 Electrotherapy on the web. Soft Tissue Repair and Healing Review. Available from http://www.electrotherapy.org/modality/soft-tissue-repair-and-healing-review (accessed 20/02/2019).
- ↑ 3.0 3.1 3.2 Stroncek JD, Reichert WM. Overview of wound healing in different tissue types. Indwelling neural implants: strategies for contending with the in vivo environment. 2008:3-40.Available from:https://www.ncbi.nlm.nih.gov/books/NBK3938/ (last accessed 31.5.2020)
- ↑ “Phagocytosis.” Merriam-Webster.com Dictionary, Merriam-Webster, https://www.merriam-webster.com/dictionary/phagocytosis. (Accessed 19 Mar. 2020).
- ↑ 5.0 5.1 5.2 5.3 Pasadena. Soft tissue healing. Available from www.pasadena.edu/files/syllabi/rxaguilar_11913.doc soft tissue healing (accessed 28/02/2019).
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 6.9 Medscape. Simon, PE. Skin Wound Healing. Available from: https://emedicine.medscape.com/article/884594-overview (accessed 28/02/2019).
- ↑ 7.0 7.1 7.2 Medical dictionary. Healing by first intention. Available from: http://medical-dictionary.thefreedictionary.com/healing+by+first+intention (accessed 28/02/2019).
- ↑ Arnheim DD. Essentials of athletic training. St Louis: CV Mosby Co. 1995.
- ↑ Sherry MA, Best TM. A comparison of 2 rehabilitation programs in the treatment of acute hamstring strains. Journal of Orthopaedic & Sports Physical Therapy 2004;34(3):116-25.
- ↑ Flex Physiotherapy. Soft tissue injury and the healing process. Published on 26 August 2017. Available from https://www.youtube.com/watch?v=i05wu5gI_4o&t=14s [last accessed 19 March 2020]