Contracture Management for Traumatic Brain Injury
Original Editor - Joan Leung
Top Contributors - Kim Jackson, Joan Leung, Naomi O'Reilly, Rachael Lowe, Admin, Tarina van der Stockt, Simisola Ajeyalemi, Olajumoke Ogunleye and Stacy Schiurring
Definition of Contractures[edit | edit source]
Contractures are losses in joint range due to changes in the passive mechanical properties of soft tissues spanning joints[1][2][3].
Epidemiology[edit | edit source]
Contractures are common following acquired brain injury[4]. Up to 84% of patients with craniocerebral trauma and about 50% of patients with cerebrovascular accident develop contractures[5][6]. A study published in 2022 reports up to 84% of persons with critical TBI may develop contractures[7]. However, reports of incidence vary substantially. Contractures are undesirable because of their potentially serious implications on motor recovery, functional outcomes and care needs.
Aetiology[edit | edit source]
Movement is essential for maintaining the passive mechanical properties of soft tissues. Animal models show that soft tissues undergo a remodelling process when they are subjected to prolonged immobilisation in a shortened position, leading to a loss of extensibility and length, and subsequently restrictions in joint range[8][9][10][11][12][13][14][15][16]. A study using ultrasound imaging has demonstrated that muscle tendon units and muscle fascicles are shorter at high tension in muscles with contractures in people with brain injury[17].
Contributors to Contractures[edit | edit source]
Muscle Weakness and Associated Loss of Motor Function[edit | edit source]
People without early signs of motor recovery are prone to contractures[18][19][20]. Muscle weakness following acquired brain injury is due to altered neural input secondary to damage to the brain which compromises neuronal recruitment and volitional muscle activation. Muscles can be further weakened through disuse. Severe weakness or lack of muscle activation immobilises a limb and deprives soft tissues of stretch. This predisposes muscles placed in shortened positions to contractures. Similarly, muscle imbalance (resulting from a muscle group being much weaker than the antagonistic group) can place joints in fixed positions and increase the risk of contractures. A number of studies have demonstrated the close relationship between contractures and muscle weakness or related motor deficits such as reduced dexterity, functional ability and mobility. [18][21][22][23][24]
Spasticity[edit | edit source]
Spasticity, like muscle weakness, can induce contractures by immobilising a limb in a fixed position[1] but the remodelling process is likely accelerated by the chronic tonic contractile activity associated with spasticity[11][25]. Spasticity may further contribute to contractures through an alteration of muscle fibre composition[26][27][28][29][30] and increased cross-bridge attachments[31]. The association of spasticity with contractures has been well documented[3][27][32][33][34][35][36][37][38][39][40]. Soft tissues of hemiplegic patients with spasticity have been found to be nearly three times stiffer than that of the age matched controls[27].
Combined Factors[edit | edit source]
Spasticity and muscle weakness are both common primary motor impairments and often co-exist after an acquired brain injury. One study identifies spasticity as the main contributor to contractures in the first 4 months after stroke and thereafter muscle weakness[22]. Another study reports that up to 20% of variation in contracture development can be explained by strength, spasticity, age, pre-morbid function and pain; of all these factors muscle weakness is the most significant predictor for contractures after stroke[5].
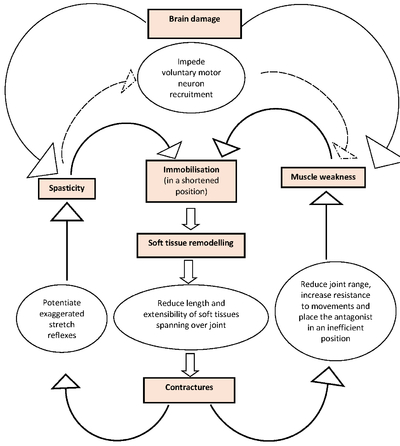
Importantly, it is the interactions amongst spasticity, weakness and contractures that may add complexity to this problem. The theoretical chains of events are illustrated in Figure 1[41].
Figure 1. A theoretical model of events contributing to the development of contractures after acquired brain injury.
Measurements[edit | edit source]
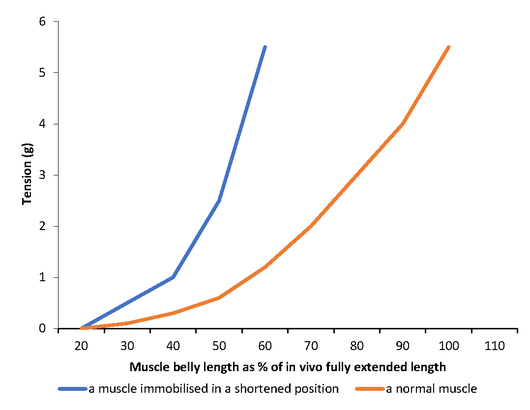
Passive stiffness is a measure of the mechanical properties of a muscle. It can be shown by the passive length-tension property of a muscle, that is, the tension encountered in a muscle at different lengths without any muscle activity[12][13]. Essentially it is the ratio of change in tension to change in length. Stiffness can be presented as the slope of a length-tension curve. Increases in passive stiffness are demonstrated by increases in the steepness of the length-tension curves (Figure 2).
Figure 2. Length/tension curves for a muscle immobilised in a shortened position and comparison is made with that of a normal muscle (control) [12]
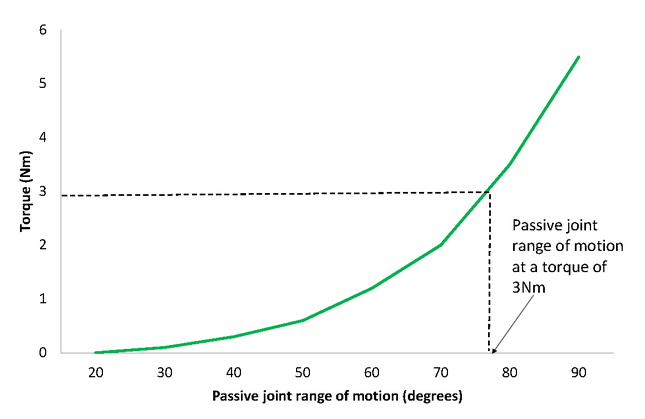
Passive stiffness can also be represented by the slope of the torque-displacement curve of a joint (Figure 3). However, the measurement procedure for this is complex and time-consuming as it involves repeated measurements of passive joint range of motion at various torques or continuous data sampling of both displacement and torque. For practical reasons, clinicians often choose to measure one point on the torque/displacement curve instead, that is, measure passive joint range with a single known torque as illustrated in Figure 3[42].
Figure 3. Torque/Displacement Curve for the Ankle Joint with the Knee Extended
Standardisation of torque is an important consideration when using passive joint range of motion to reflect soft tissue extensibility. It minimises measurement errors associated with uncontrolled torques and altered stretch tolerance of an individual[43][44][45].
Methods to apply controlled torque using a standardised procedure to reliably measure passive joint range to reflect muscle extensibility have been described in a few papers[46][47][48].
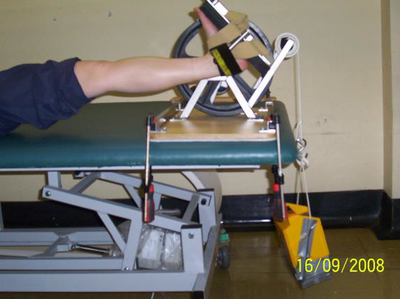
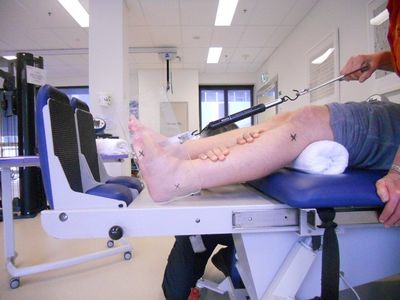
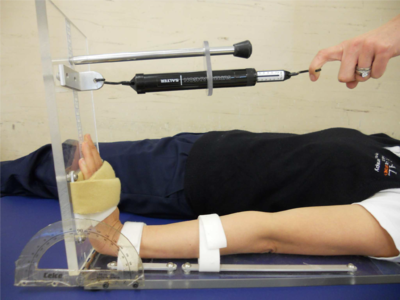
The following photographs show some of the equipment and limb position for measuring passive joint range to reflect muscle extensibility:
| Measurement Tool for Passive Ankle Range | Measurement Tool for Passive Ankle Range | Measurement for Passive Wrist Range |
|
Management / Interventions[edit | edit source]
Passive Stretch[edit | edit source]
Passive stretch has been the most widely used method to prevent and correct contractures. The observations that soft tissues undergo structural remodelling and elongate when subject to prolonged stretch underpin the rationale for using passive stretch for contracture management.
A Cochrane review and an abridged republication of a Cochrane Systemic Review indicate that commonly used clinical passive stretch interventions do not produce clinically worthwhile or lasting effects. [49][50] Application duration of passive stretch, however, was short in most of the randomised controlled trials included in the review, with the median duration being 6.5 hours a day over 30 days.
Of the clinically used passive stretch interventions, serial casting provides the highest dose of stretch. Two randomised controlled trials have found serial casting effective in the correction of contractures after traumatic brain injury. [51][52] The effect of serial casting, however, was observed to dissipate within a day and completely disappear after 4 weeks. [51]
Existing evidence does not find passive stretch, as typically applied in clinical settings, produces large or sustained effects for contractures.
Note: The use of stretch over an extended period (such as,the incorporation of 24 hours a day positioning intoa daily routine or splinting for 24 hours a day over months) have not been investigated.
Electrical Stimulation[edit | edit source]
Electrical stimulation, through its effects on improving muscle strength and spasticity [53][54][55][56][57][58][59], is potentially useful for addressing the contributors to contractures.
A randomised controlled trial investigated the use of electrical stimulation (60 minutes a day for 4 weeks) on wrist range of motion as an additional treatment for contracture management in people with traumatic brain injury and stroke. It found no significant treatment effect in joint range. [60]
Another randomised controlled trial found that tilt table standing combined with electrical stimulation (30 minutes a day) and ankle splinting (12 hours a day) over 6 weeks was not more effective than tilt table standing alone on joint range of motion in people with traumatic brain injury. [61]
The addition of electrical stimulation, 30-60 minutes a day over 4 to 6 weeks, to a stretch program does not provide extra benefit to joint range.
Botulinum Toxin[edit | edit source]
Spasticity is one of the known contributors to contractures. Interventions that reduce spasticity is seemingly useful in the prevention or correction of contractures.
A double-blind placebo-controlled study investigated botulinum toxin of various doses for managing upper limb spasticity after stroke or head injury. It found no change in passive joint range of elbow, wrist and fingers. A small increase in range was observed in the elbow at the highest dose (mean 984Mu) but this effect was lost by 12 weeks. [62]
There is no evidence that botulinum toxin alone produces clinically worthwhile or sustained treatment effect on contractures.
Multi-modal Therapy[edit | edit source]
Contractures are a complex problem involving multiple contributors, such as spasticity and muscle weakness. Existing evidence has suggested that sole interventions focusing on either reversing soft tissue shortening or addressing the underlying causes of contractures have limited effectiveness. Combining passive stretch with treatments that address the contributors appears a logical and worthwhile approach to consider for contracture management.
Two case studies on severe contractures following traumatic brain injury provided justification for examining a combined approach. One reported no change in joint range using splinting alone but then a long-term resolution of the contractures using serial casting, followed by splinting and motor training. [63] Another case study also reported a large and sustained effect on t range using the same program but adding botulinum toxin prior to serial casting. [64]
A randomised controlled trial investigated a multi-modal program combining serial casting with botulinum toxin, followed by splinting and motor training, on people with severe ankle contractures after traumatic/acquired brain injury. It found a significant treatment effect on joint range immediately following casting, and the improvement was maintained at Week 2 and Week 8. [65] A follow-up study of this trial found that the gain in joint range was maintained at discharge (median follow-up duration: 198 days). This follow-up study also found an association between functional outcome and the long-term improvement in joint range resulting from this program.
A multi-modal program comprising serial casting and botulinum toxin, followed by splinting and motor training can produce lasting benefits for the correction of contractures. People with lesser residual physical impairments or better prospect for functional improvements are more likely to respond well to the program.
Recommendations for Clinical Management[edit | edit source]
There is evidence that supports the principle of using high intensity passive stretch in combination with interventions to address the underlying causes of contractures. Clinicians may consider using a multi-modal program comprising serial casting and botulinum toxin, followed by splinting and motor training or other plausible treatment combinations based on the principle.
Clinical Guidelines[edit | edit source]
Rehabilitation following Acquired Brain Injury, National Clinical Guidelines, the British Society of Rehabilitation Medicine (BSRM) 2003
5.4 Positioning and Handling - Therapeutic positioning is widely advocated to discourage the development of abnormal postures, contractures, pain, skin breakdown and respiratory complications – all of which are associated with delayed discharge and poorer outcomes. Prevention of these complications in the early stages is an important element in maximising functional gains and quality of life and in reducing the long-term costs of care (NCGS2).
G61 Patients with marked spasticity and/or contractures should have a coordinated plan for interdisciplinary management including:
- elimination of simple causative or aggravating factors such as pain and infection
- the use of specific treatment modalities such as serial plaster casts or removable splints if appropriate (B)
- the use of antispasmodic drugs including botulinum toxin where appropriate.
Brain Injury Rehabilitation in Adults: A National Clinical Guideline Scottish Intercollegiate Guidelines Network (2013)
4.2.1 Splints, Casts , Stretches and Orthoses - Casts, splints and passive stretching may be considered in cases where contracture and deformity are progressive.
References[edit | edit source]
- ↑ 1.0 1.1 Botte MJ, Nickel VL, Akeson WH. Spasticity and contracture. Physiologic aspects of formation. Clin Orthop Relat Res. 1988(233):7-18.
- ↑ Dudek N, Trudel G. Joint contractures. In: Frontera W, Silver J, Rizzo T, editors. Essentials of physical medicine and rehabilitation musculoskeletal disorders, pain, and rehabilitation. 2nd ed: Philadelphia: Saunders; 2008. p. 651-5.
- ↑ 3.0 3.1 Harburn KL, Potter PJ. Spasticity and contractures. Phys Med Rehabil State of the Art Reviews. 1993;7:113-32.
- ↑ Fergusson D, Hutton B, Drodge A. The epidemiology of major joint contractures: a systematic review of the literature. Clin Orthop Relat Res. 2007(456):22-9.
- ↑ 5.0 5.1 Kwah LK, Harvey LA, Diong JH, Herbert RD. Half of the adults who present to hospital with stroke develop at least one contracture within six months: an observational study. Journal of Physiotherapy. 2012;58(1):41-7
- ↑ Yarkony GM, Sahgal V. Contractures. A major complication of craniocerebral trauma. Clin Orthop Relat Res. 1987(219):93-6.
- ↑ Mousa AS, Ali ZH, Elbably MM. Assessment of Extremities Muscle Contracture for Critically Brain Injured Patients. 2022.
- ↑ Tabary JC, Tabary C, Tardieu C, Tardieu G, Goldspink G. Physiological and structural changes in the cat's soleus muscle due to immobilization at different lengths by plaster casts. J Physiol. 1972;224(1):231-44.
- ↑ Goldspink G, Tabary C, Tabary JC, Tardieu C, Tardieu G. Effect of denervation on the adaptation of sarcomere number and muscle extensibility to the functional length of the muscle. J Physiol. 1974;236(3):733-42
- ↑ Tabary JC, Tardieu C, Tabary C, Lombard M, Gagnard L, Tardieu G. [Neural regulation and adaptation of the number of sarcomeres of the muscle fiber to the length imposed upon it]. J Physiol (Paris). 1972;65:Suppl 1:168A
- ↑ 11.0 11.1 Tabary JC, Tardieu C, Tardieu G, Tabary C. Experimental rapid sarcomere loss with concomitant hypoextensibility. Muscle Nerve. 1981;4(3):198-203.
- ↑ 12.0 12.1 12.2 Williams PE, Goldspink G. Changes in sarcomere length and physiological properties in immobilized muscle. J Anat. 1978;127(Pt 3):459-68.
- ↑ 13.0 13.1 Herbert RD, Balnave RJ. The effect of position of immobilisation on resting length, resting stiffness, and weight of the soleus muscle of the rabbit. J Orthop Res. 1993;11(3):358-66.
- ↑ Akeson WH, Woo SL, Amiel D, Matthews JV. Biomechanical and biochemical changes in the periarticular connective tissue during contracture development in the immobilized rabbit knee. Connect Tissue Res. 1974;2(4):315-23
- ↑ Akeson WH, Amiel D, Abel MF, Garfin SR, Woo SL. Effects of immobilization on joints. Clin Orthop Relat Res. 1987(219):28-37.
- ↑ Woo SL, Matthews JV, Akeson WH, Amiel D, Convery FR. Connective tissue response to immobility. Correlative study of biomechanical and biochemical measurements of normal and immobilized rabbit knees. Arthritis Rheum. 1975;18(3):257-64
- ↑ Kwah LK, Herbert RD, Harvey LA, Diong J, Clarke JL, Martin JH, et al. Passive Mechanical Properties of Gastrocnemius Muscles of People With Ankle Contracture After Stroke. Archives of Physical Medicine and Rehabilitation. 2012;93(7):1185-90
- ↑ 18.0 18.1 Pandyan AD, Cameron M, Powell J, Stott DJ, Granat MH. Contractures in the post-stroke wrist: a pilot study of its time course of development and its association with upper limb recovery. Clin Rehabil. 2003;17(1):88-95.
- ↑ Turton AJ, Britton E. A pilot randomized controlled trial of a daily muscle stretch regime to prevent contractures in the arm after stroke. Clin Rehabil. 2005;19(6):600-12.
- ↑ Malhotra S, Pandyan AD, Rosewilliam S, Roffe C, Hermens H. Spasticity and contractures at the wrist after stroke: time course of development and their association with functional recovery of the upper limb. Clinical Rehabilitation. 2011;25(2):184-91.
- ↑ Sackley C, Brittle N, Patel S, Ellins J, Scott M, Wright C, et al. The prevalence of joint contractures, pressure sores, painful shoulder, other pain, falls, and depression in the year after a severely disabling stroke. Stroke. 2008;39(12):3329-34
- ↑ 22.0 22.1 Ada L, O'Dwyer N, O'Neill E. Relation between spasticity, weakness and contracture of the elbow flexors and upper limb activity after stroke: an observational study. Disability and Rehabilitation. 2006;28(13-14):891-7
- ↑ Faghri PD, Rodgers MM, Glaser RM, Bors JG, Ho C, Akuthota P. The effects of functional electrical stimulation on shoulder subluxation, arm function recovery, and shoulder pain in hemiplegic stroke patients. Arch Phys Med Rehabil. 1994;75(1):73-9.
- ↑ Bohannon RW. Relationship between shoulder pain and selected variables in patients with hemiplegia. Clin Rehabil. 1988;2:111-17.
- ↑ Huet de la Tour E, Tardieu C, Tabary JC, Tabary C. Decrease of muscle extensibility and reduction of sarcomere number in soleus muscle following a local injection of tetanus toxin. J Neurol Sci. 1979;40(2-3):123-31.
- ↑ Edstrom L. Relation between spasticity and muscle atrophy pattern in upper motor neurone lesions. Scand J Rehabil Med. 1973;5(4):170-1.
- ↑ 27.0 27.1 27.2 Sinkjaer T, Magnussen I. Passive, intrinsic and reflex-mediated stiffness in the ankle extensors of hemiparetic patients. Brain. 1994;117 ( Pt 2):355-63.
- ↑ Edstrom L. Selective changes in the sizes of red and white muscle fibres in upper motor lesions and Parkinsonism. Journal of the Neurological Sciences. 1970;11(6):537-50
- ↑ Dattola R, Girlanda P, Vita G, Santoro M, Roberto ML, Toscano A, et al. Muscle rearrangement in patients with hemiparesis after stroke: an electrophysiological and morphological study. European Neurology. 1993;33(2):109-14.
- ↑ Dietz V, Ketelsen UP, Berger W, Quintern J. Motor unit involvement in spastic paresis. Relationship between leg muscle activation and histochemistry. Journal of the Neurological Sciences. 1986;75(1):89-103
- ↑ Carey JR, Burghardt TP. Movement dysfunction following central nervous system lesions: a problem of neurologic or muscular impairment? Phys Ther. 1993;73(8):538-47.
- ↑ Bobath B, editor. Adult hemiplegia: evaluation and treatment: London: Heinemann Medical Books; 1990.
- ↑ O'Dwyer NJ, Ada L, Neilson PD. Spasticity and muscle contracture following stroke. Brain. 1996;119 ( Pt 5):1737-49.
- ↑ Vattanasilp W, Ada L, Crosbie J. Contribution of thixotropy, spasticity, and contracture to ankle stiffness after stroke. J Neurol Neurosurg Psychiatry. 2000;69(1):34-9.
- ↑ Singer B, Dunne J, Singer KP, Allison G. Evaluation of triceps surae muscle length and resistance to passive lengthening in patients with acquired brain injury. Clin Biomech (Bristol, Avon). 2002;17(2):152-61.
- ↑ Chung SG, Van Rey E, Bai Z, Roth EJ, Zhang LQ. Biomechanic changes in passive properties of hemiplegic ankles with spastic hypertonia. Arch Phys Med Rehabil. 2004;85(10):1638-46.
- ↑ Given JD, Dewald JP, Rymer WZ. Joint dependent passive stiffness in paretic and contralateral limbs of spastic patients with hemiparetic stroke. Journal of Neurology, Neurosurgery and Psychiatry. 1995;59(3):271-9.
- ↑ Lee WA, Boughton A, Rymer WZ. Absence of stretch reflex gain enhancement in voluntarily activated spastic muscle. Experimental Neurology. 1987;98(2):317-35.
- ↑ Dietz V, Trippel M, Berger W. Reflex activity and muscle tone during elbow movements in patients with spastic paresis. Annals of Neurology. 1991;30(6):767-79.
- ↑ Perry J. Rehabilitation of spasticity. In: Feldman RG, Young RR, Koella WP, editors. Spasticity: disordered motor control. Miami: Symposia Specialists1980. p. 87-100.
- ↑ Leung JW. Physiotherapy management of contractures after acquired brain injury. University of Sydney.
- ↑ Chesworth BM, Vandervoort AA. Comparison of passive stiffness variables and range of motion in uninvolved and involved ankle joints of patients following ankle fractures. Phys Ther. 1995;75(4):253-61.
- ↑ Halbertsma JP, Goeken LN, Hof AL, Groothoff JW, Eisma WH. Extensibility and stiffness of the hamstrings in patients with nonspecific low back pain. Archives of Physical Medicine and Rehabilitation. 2001;82(2):232-8.
- ↑ Magnusson SP, Simonsen EB, Aagaard P, Sorensen H, Kjaer M. A mechanism for altered flexibility in human skeletal muscle. J Physiol. 1996;497 ( Pt 1):291-8.
- ↑ Folpp H, Deall S, Harvey LA, Gwinn T. Can apparent increases in muscle extensibility with regular stretch be explained by changes in tolerance to stretch? Aust J Physiother. 2006;52(1):45-50.
- ↑ Harvey L, Byak A, Ostrovskaya M, Glinsky J. Reliability of a device designed to measure ankle mobility. Spinal Cord. 2003;41(10):559-62.
- ↑ Moseley A, Adams R. Measurement of passive ankle dorsiflexion: Procedure and reliability. Aust J Physiother. 1991;37(3):175-81.
- ↑ Harvey L, King M, Herbert R. Test-retest reliability of a procedure for measuring extensibility of the extrinisic finger flexor muscles. J Hand Ther. 1994;7:251-4
- ↑ Katalinic OM, Harvey LA, Herbert RD. Effectiveness of stretch for the treatment and prevention of contractures in people with neurological conditions: a systematic review. Physical Therapy. 2011;91(1):1-14.
- ↑ Harvey LA, Katalinic OM, Herbert RD, Moseley AM, Lannin NA, Schurr K. Stretch for the treatment and prevention of contracture: an abridged republication of a Cochrane Systematic Review. J Physiother. 2017;63(2):67-75.
- ↑ 51.0 51.1 Moseley AM, Hassett LM, Leung J, Clare JS, Herbert RD, Harvey LA. Serial casting versus positioning for the treatment of elbow contractures in adults with traumatic brain injury: a randomized controlled trial. Clin Rehabil. 2008;22(5):406-17.
- ↑ Moseley AM. The effect of casting combined with stretching on passive ankle dorsiflexion in adults with traumatic head injuries. Phys Ther. 1997;77(3):240-7; discussion 8-59.
- ↑ Glinsky J, Harvey L, Van Es P. Efficacy of electrical stimulation to increase muscle strength in people with neurological conditions: a systematic review. Physiother Res Int. 2007;12(3):175-94
- ↑ Yan T, Hui-Chan CW, Li LS. Functional electrical stimulation improves motor recovery of the lower extremity and walking ability of subjects with first acute stroke: a randomized placebo-controlled trial. Stroke. 2005;36(1):80-5.
- ↑ Bakhtiary AH, Fatemy E. Does electrical stimulation reduce spasticity after stroke? A randomized controlled study. Clin Rehabil. 2008;22(5):418-25.
- ↑ Sabut SK, Sikdar C, Kumar R, Mahadevappa M. Functional electrical stimulation of dorsiflexor muscle: effects on dorsiflexor strength, plantarflexor spasticity, and motor recovery in stroke patients. NeuroRehabilitation. 2011;29(4):393-400.
- ↑ Mangold S, Schuster C, Keller T, Zimmermann-Schlatter A, Ettlin T. Motor training of upper extremity with functional electrical stimulation in early stroke rehabilitation. Neurorehabil Neural Repair. 2009;23(2):184-90.
- ↑ Sahin N, Ugurlu H, Albayrak I. The efficacy of electrical stimulation in reducing the post-stroke spasticity: a randomized controlled study. Disability and Rehabilitation. 2012;34(2):151-6.
- ↑ Lin Z, Yan T. Long-term effectiveness of neuromuscular electrical stimulation for promoting motor recovery of the upper extremity after stroke. J Rehabil Med. 2011;43(6):506-10.
- ↑ Leung J, Harvey LA, Moseley AM, Tse C, Bryant J, Wyndham S, et al. Electrical stimulation and splinting were not clearly more effective than splinting alone for contracture management after acquired brain injury: a randomised trial. J Physiother. 2012;58(4):231-40.
- ↑ Leung J, Harvey LA, Moseley AM, Whiteside B, Simpson M, Stroud K. Standing with electrical stimulation and splinting is no better than standing alone for management of ankle plantarflexion contractures in people with traumatic brain injury: a randomised trial. J Physiother. 2014;60(4):201-8.
- ↑ Smith SJ, Ellis E, White S, Moore AP. A double-blind placebo-controlled study of botulinum toxin in upper limb spasticity after stroke or head injury. Clin Rehabil. 2000;14(1):5-13.
- ↑ Leung J, Harvey LA, Moseley AM. An intensive programme of passive stretch and motor training to manage severe knee contractures after traumatic brain injury: a case report. Physiotherapy Canada. 2013;65(3):223-8.
- ↑ Leung J, Stroud K. Long-Term Resolution of Severe Ankle Contractures Using Botulinum Toxin, Serial Casting, Splinting, and Motor Retraining. Physiother Can. 2018;70(2):152-9.
- ↑ Leung J, King C, Fereday S. Effectiveness of a programme comprising serial casting, botulinum toxin, splinting and motor training for contracture management: a randomized controlled trial. Clin Rehabil. 2019;33(6):1035-44.