Amyotrophic Lateral Sclerosis: A Spinal-Onset Case Study
Original Editor - Hannah Perreault, Katie Wolters, Jessie Fleming, Jill Hubbard, Rochelle McCoy, Aamira Lokhandwala
Top Contributors - Hannah Perreault, Katie Wolters, Jessie Fleming, Jill Hubbard, Rochelle Ann-Marie McCoy, Aamira Lokhandwala
Abstract[edit | edit source]
Amyotrophic Lateral Sclerosis (ALS), also known as Lou Gehrig's disease, is a progressive neurological disorder that affects voluntary muscle movement[1]. Specifically, upper and lower motor neurons are destroyed which results in various devastating symptoms such as muscle weakness, speech impairments, and swallowing difficulties[1]. Patients die on average 3-5 years after symptoms onset due to respiratory failure[1]. The current cause and cure remain unknown. In this fictional case study, a 70-year-old male has been diagnosed with the limb/spinal-onset subtype of ALS six months prior. Client characteristics, subjective and objective examination findings, and a clinical hypothesis are documented throughout the case study. Additionally, a potential physiotherapy intervention is outlined in detail using the latest evidence based research, including verified outcome measures for this population. Primary patient-centred goals include the maintenance of mobility, strength, and participation in meaningful activities despite living with a progressive disease. Throughout this case study, the role of physiotherapists and the importance of a strong inter-disciplinary team is highlighted in the management of ALS.
Introduction[edit | edit source]
Pathophysiology[edit | edit source]
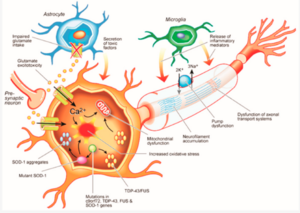
Amyotrophic Lateral Sclerosis (ALS or Lou Gerhig’s disease) is a neurodegenerative disease that is characterized by the progressive loss of upper motor neurons in the motor cortex and lower motor neurons in anterior horn of the spinal cord[2]. In limb-onset ALS, the disease begins by affecting the distal limbs and spreads through the motor system, leading to a lack of control over the musculoskeletal system[2]. Re-innervation can occur and sprouting of surrounding axons help to compensate for affected areas to preserve strength and function [2]. However, in the later stages of this disease, re-innervation can no longer compensate for the increased rate of degeneration.
ALS is also regarded as a multi-system disease that affect various areas in the body[2]. Regions beyond the motor system that may also be affected is the nervous system, cerebellum, and basal ganglion[2]. The sensory system can also be involved but is usually unaffected[2].
Etiology[edit | edit source]
The cause of ALS is still not completely known, however a number of genes and chromosomal regions have been linked to the disease[2]. An estimated 5-10% of individuals with ALS have a family history of ALS (termed familial ALS), however the majority with ALS have no family history (termed sporadic ALS)[2]. For the sporadic form, it is thought that a number of mechanisms can lead to the development of ALS, including “oxidative stress, aberrant RNA processing, exogenous neurotoxicity, excitotoxicity, impaired axonal transportation, axonal dysfunction, mitochondrial disruption, protein misfolding, protein aggregation, apoptosis (programmed cell death), and lifestyle factors”[2]. Some established environmental risk factors include male gender and smoking, while probable risk factors include US military service, lead, and pesticide exposure, and other explored risk factors include head trauma, excessive physical activity, electromagnetic radiation, and a low body mass index (BMI)[3].
Epidemiology[edit | edit source]
According to ALS Canada, the prevalence of ALS globally is around 200,000 people[4]. In Canada specifically, it is estimated that 3,000 people are currently living with ALS, and each year approximately 1,000 will die from it[4]. Further, within 2-5 years of diagnosis, roughly 80% of people with ALS die[4]. While ALS can occur at any age, its onset is most commonly seen in the mid-to-late 50s, with men slightly more affected than women (1.7:1 ratio)[2]. The incidence of ALS also appears to increase with each decade of life, where the overall incidence ranges from 0.4-2.4 cases per 100,000[2].
Diagnosis[edit | edit source]
ALS is not easily diagnosed as there is not one single test or biological marker that can be used. ALS is often a diagnosis of exclusion, meaning that individuals with its clinical presentation undergo a series of diagnostic tests, with the aim of gathering information to support the ALS diagnosis and ruling out other conditions[2]. This process of obtaining a confirmed diagnosis often takes 8-15 months[2].
Various muscle and imaging tests for diagnosis are used, such as electromyography (detects electrical activity of muscle fibres), nerve conduction velocity studies (assesses nerves ability to send a signal), muscle and nerve biopsies, and neuroimaging studies[5]. In order to reach an ALS diagnosis, certain criterion must be met. This criteria is known as the Airlie House criteria (a revised version of El Escorial criteria) using Awaji-shima recommendations. They are as follows[6]:
- the presence of evidence of LMN degeneration by clinical, electrophysiological or neuropathological examination
- the presence of UMN degeneration by clinical examination
- the presence of progressive spread of symptoms or signs within a region or other regions, as determined by history, clinical examination or electrophysiological tests, and
- an absence of electrophysiological or pathological evidence of other disease processes that might explain the observed clinical and electrophysiological signs.
There are 3 diagnostic categories: definite ALS, probable ALS, and possible ALS. These categories including the following criterion[6]:
- Definite ALS: clinical or electrophysiological evidence by the presence of LMN as well as UMN signs in the bulbar region and at least two spinal regions or the presence of LMN and UMN signs in three spinal regions
- Probable ALS: clinical or electrophysiological evidence by LMN and UMN signs in at least two regions with some UMN signs necessarily rostral to the LMN signs
- Possible ALS: clinical or electrophysiological signs of UMN or LMN dysfunction in only one region or UMN signs alone in two or more regions or LMN rostral to UMN signs
Symptoms[edit | edit source]
The symptoms of ALS vary considerably between patients, but the most prevalent and frequently observed early symptom is focal, asymmetrical muscle weakness, which eventually spreads bilaterally and can impact any limb[2]. The heterogeneity in clinical presentation can be attributed to the fact that there are different phenotypes of the disease, including the more common limb-onset, and less common bulbar onset, with the former presenting more commonly with issues in skeletal muscles and the latter with complications related to speech and swallowing[2]. Below is a table depicting commonly observed impairments (and signs) in ALS, in relation to the impacted body system[2].
*Table is modified from resource: O’Sullivan B. S, Schmitz J. T, & Fulk D. G, editors. Physical Rehabilitation. 7th-ed. Philadelphia: F. A. Davis Company, 2019 [2].
| Affected System | Clinical Manifestations and Impairments |
| LMN | Muscle weakness, hyporeflexia, hypotonicity, atrophy, muscle cramps and fasciculations |
| UMN | Spasticity, hyperreflexia, muscle weakness |
| Bulbar | Head and neck muscle weakness, dysphagia, dysarthria, excessive saliva flow, uncontrolled laughing or crying |
| Respiratory | Weakness in inspiratory and expiratory musculature, resting and exertional dyspnea, orthopnea, hypoventilation, secretion retention, weak cough |
| Other (rare and/or indirect associated impairments) | Dementia-related signs and symptoms, cognitive and behavioural deficits (ex. attention, irritability), sensory impairments, bowel and bladder issues, fatigue, weight loss, pain, depression, anxiety, balance and postural control deficits |
Medications[edit | edit source]
One of the first approved and most common medications for treatment in the ALS population is Riluzole[7]. Though there is currently no known cure for ALS, Riluzole can increase lifespan by several months, depending on the severity of the individual[7]. The medication is a glutamate blocker, meaning that it decreases the release of the neurotransmitter glutamate from neurons which stimulate the motor neurons in the body[7]. The theory behind this is the decrease of excessive glutamate release, which can cause over-stimulation known as excitotoxicity and degrades the receiving motor neurons[7]. Adverse effects of Riluzole are generally minor, including nausea and fatigue, but can affect liver function in those with pre-existing problems in the area[7]. During physiotherapy, patients who are using this medication may appear to have increased fatigue levels and require more frequent breaks.
Client Characteristics[edit | edit source]
The patient is a 70 year old, right hand dominant, caucasian male named Lou Gherig. He retired twenty years ago after a successful career in professional baseball and now lives with his wife and adult children in a two storey home on the west side of Kingston. Lou was diagnosed six months ago with ALS by a neurologist at KGH and presented with both upper and lower motor neuron deficits. Specifically, Lou suffers from the limb / spinal onset phenotype of ALS. He was prescribed Riluzole medication which has the potential to increase his lifespan by several months[7]. He does not present with any comorbidities other than hypertension. During the initial physiotherapy assessment, it was noted that Lou has progressively worsening weakness in his upper and lower extremities. Six main problems that have been identified include: muscle cramping, mild spasticity in the upper extremities, decreased balance, inability to manage stairs independently, struggling to walk his large dog and not being able to partake in painting like he used to, due to fatigue and dyspnea when engaging in activity.
Examination Findings[edit | edit source]
While there were several problems identified through assessment, the three primary concerns identified by the patient are as follows:
- Muscle weakness in the musculature of bilateral distal upper and lower extremities
- Inability to ambulate independently
- Restriction from painting due to loss of manual dexterity
A common comorbidity that should be screened in all ALS patients is hypertension. Literature shows a strong association between hypertension and ALS, with and increased risk of death occurring if both are present[8]. Therefore, Lou was screened for hypertension, and it was revealed in his past medical history that he does have this co-morbidity.
Clinical Hypothesis[edit | edit source]
A physiotherapy diagnosis notes decreased overall function due to generalized muscle weakness in the distal extremities, and impaired balance. Body structure, activities, and participation have all been impaired. Given that Lou was previously active, has a supportive family at home, and is financially stable, it is expected that physiotherapy will be able to minimize the impairment from ALS and help to maintain as much function as possible. However, Lou has been experiencing some depressive feelings after his diagnosis, is not motivated to complete rehabilitation, and has a fear of falling. These factors will be barriers in terms of prognosis.
Outcome[edit | edit source]
Body Structure and Function:[edit | edit source]
Modified Ashworth Scale (MAS)[edit | edit source]
The MAS is an assessment tool used to measure increases in muscle tone or spasticity, where the examiner applies a high velocity stretch to a joint or muscle and then grades the spasticity on an ordinal scale of 0-4[9].
Overall, measurements obtained with the Modified Ashworth Scale (MAS) have varying inter- and intra-rater reliability depending upon which limbs is being tested[10]. As a result of this varying reliability, the validity of the MAS must be questioned[10][11]. There is also a lack of research surrounding the validity and reliability of the MAS in the ALS population specifically[12]. Nonetheless, it is still the standard for clinical assessment of extreme spasticity[11], and can be considered a supplemental outcome measure for ALS[9].
Muscle Strength Testing (MMT and MVIC)[edit | edit source]
Manual muscle testing (MMT) is a technique used to assess the strength of a given muscle that is being testing, where the assessor manually resists the action of that muscle and then scores the strength on a scale ranging from 0-5 (no strength being 0/5 and normal strength being 5/5). Maximal voluntary isometric contraction (MVIC) is another method to assess strength, however it requires specialized equipment and training[2]. In neuromuscular diseases, it is considered a standardized method to assess muscle strength and has been used extensively in individuals with ALS[13].
Compared to MMT, MVIC eliminates muscle length and velocity as factors in testing, allowing for the production of more reliable and valid data[2]. Much of the research surrounding MMT is varied, with reliability and validity dependent on which muscle is being tested[14]. MMT is also said to lack sensitivity[15]. However, a study examining MMT in comparison to MVIC stated that MMT was better able to detect progressive weakness in ALS, making it the favoured technique for this population[14]. It is important to note however, that in this study there were significantly more muscles tested using MMT, and therefore difference in detecting change was likely accounted for by the number of muscles sampled[14]. Nonetheless, MMT is still considered a core assessment tool[16]. Either technique can be used for strength testing, however MVIC requires specialized equipment that not all physiotherapists have access to.
Activity:[edit | edit source]
Berg Balance Scale (BBS) [edit | edit source]
The BBS is used to assess balance and falls risk in older adults through a series of 14 items that include varying static and dynamic balance activities[17]. Each item is scored on a scale of 0-4, determined by the ability to perform a given activity[17].
The use of the BBS in the ALS population is not well cited in the literature, however its use in other neurological populations is better documented[18]. One study shares that the BBS demonstrates excellent internal consistency and reliability when used with individuals with chronic progressive neurological conditions, and is strongly recommended for that general group[19]. Its use in many older adult populations also demonstrate excellent inter and intra-rater reliability, and high specificity[17].
6 Minute Walk Test (6MWT)[edit | edit source]
The 6MWT evaluates sub-maximal aerobic capacity or endurance by assessing the distance walked over the course of 6 minutes[20]. One study determined that the 6MWT is a valid measure of aerobic capacity in ambulatory patients with ALS, while another shares that the test demonstrates good responsiveness and reliability in a longitudinal setting with ALS[21][22]. Its use in chronic progressive neurological conditions as a whole is also strongly recommended, with evidence of excellent reliability[19].
Nine Hole Peg Test (NHPT)[edit | edit source]
The NHPT is scored based on the time it takes to remove the nine pegs, one by one, from the holes on the board, place them into a container, and then one by one replace them into the holes on the board[23]. This test is used to examine hand dexterity and can be a good tool to follow dexterity progression in ALS[23].
While research is limited, this test has demonstrated high inter-rater reliability and is sensitive to patients with neuromuscular or musculoskeletal conditions[24]. In other populations such as multiple sclerosis, stroke and Parkinson's disease, this test has demonstrated excellent test-retest reliability, excellent inter-rater reliability, and validity varying between the different neurological conditions[25].
Participation:[edit | edit source]
Amyotrophic Lateral Sclerosis Functional Rating Scale revised (ALSFRS-R)[edit | edit source]
The ALSFRS-R is a 12-item scale addressing activities of daily living and global function in patients with ALS[26]. There are four specific domains that it addresses, including fine motor, gross motor, bulbar, and respiratory function[27]. For each item on the scale, patients can rate their abilities from 0 = unable to perform the task, to 4 = normal functioning[27]. This scale helps estimate a patient’s degree of functional impairment[26].
This test has excellent test-retest reliability, excellent inter and intra-rater reliability, excellent internal consistency, good construct validity, and is sensitive to change[27][26]. However, use of a total score from the scale (sum of each domain) shows weak factorial validity and poor unidimensionality, meaning the focus should be on reporting subscores from each domain instead[27]. Overall, the ALSFRS-R has also been shown to be an independent predictor of survival and a sensitive prognostic biomarker (specifically when examining the rate of change in the ALSFRS-R score over time)[27].
Goals and Interventions[edit | edit source]
There is no cure or form of treatment that can reverse the progressive effects of ALS, so the focus of this intervention program is on supportive treatments that maintains Lou’s current function and quality of life[2]. Lou expressed prior to starting his rehabilitation program that he was unmotivated and has a fear of falling. Research has shown that when health care professionals are able to understand a patient’s educational needs, behaviours such as motivation and fearfulness can be addressed leading to a more positive experience[28]. To address Lou's needs, he was provided education on ALS, possible symptoms, reduced function, and the benefits of exercise before beginning his rehabilitation program.
Short Term Goals:[edit | edit source]
| Short Term Goal 1 | Intervention: Type | Intervention: Frequency, Intensity, Timing[29] | Modifications for Intervention |
| By week 4, patient will ambulate independently using a rollator for 15 minutes with minimal breaks. 30 Second Sit to Stand and Berg Balance Test will be used to assess lower extremity endurance | Treadmill training with body weight supported | Low to Moderate intensity (RPE: 4-6)
20-30-minute duration |
Balance: patient can use a recumbent bike or go swimming
Fear of falling/balance: educate and auditory cueing Muscle weakness: pacing or breaking activity up into 10-minute intervals Fatigue: decrease intensity, have patient use AFO to conserve energy |
Goal Discussion[edit | edit source]
After the first week, Lou mentioned that he enjoyed participating in his rehabilitation program. Lou’s gait had slightly improved but was still walking with his bodyweight supported on a treadmill. This intervention for aerobic exercise follows the AMSC guidelines for neurological disorders, recommending 2 to 3 days a week of moderate intensity exercise be performed for 20-30 minutes to improve quality of life, balance, and gait[29]. Prior research in this area also supports treadmill training with bodyweight support for those with ALS. Findings from a systematic review showed an improvement in gait kinematics, velocity, symmetry, and overall endurance[30]. In addition to these findings, treadmill training with sensory feedback (such as sensory cues, visual cures, rhythmic cues) was also looked at with ALS participants[30]. It was found that both verbal and musical auditory cues had the greatest effect on improving gait, specifically step length and width[30]. When making adjustments to Lou’s intervention plan, a good modification would be to add in verbal or rhythmic cueing to help him achieve his short term goal. At the end of the intervention, outcome measures were re-taken. Lou was able to perform 10 sit to stands (below the average for his age, 14) in 30 seconds without the use of his upper extremities for support, and was able to achieve a score of 51 on the Berg Balance Scale[31][32]. These outcome measures, although maintained, indicate that he is still at risk of a fall. Therefore, a rollator would be best suited for Lou when ambulating for 15 minutes or longer[32]. By the end of the 4 weeks, although there was minimal improvement with Lou’s gait, he was still able to complete his short term goal. He is also showing improvements with his posture, as he is now able to maintain an upright position while walking.
| Short Term Goal 2 | Intervention: Type | Intervention: Frequency, Intensity, Timing[29] | Modifications for Intervention |
| Patient will perform all exercises in their individualized exercise program at a moderate intensity to maintain muscle strength by week 4. RPE and manual muscle tests will be used to measure patient intensity and strength | Isometric exercises:
1. Quad over rolls 2. Glute squeezes 4. Dorsiflexion against gravity 5. Shoulder raises 6. Bicep curls 7. Ball sqeezes |
15 reps
2 sets
2-3x a week
5-10 second holds Using a (light/medium) resistance band |
Muscle cramping during exercise: add in rest periods between sets and apply a heat pack to muscles at the end of the session
Spasticity: have the patient practice movement compositions/synergies to help smooth/coordinate movements. Motivation: Educate patient |
Goal Discussion[edit | edit source]
Lou also engaged in an individualized resistance training program to help maintain muscle strength in both his upper and lower extremities. Moderate exercise is commonly recommended to help manage many neurological diseases to help enhance muscle strength and avoid further complications[33]. However, those with ALS do not get these same benefits due to the rate of muscle deterioration that occurs over time[33]. There is a lack of evidence to support the efficacy of exercise in maintaining physical function, quality of life, and increasing life expectancy of those with ALS[34]. This has caused much controversy over the years, suggesting that regular exercise should be avoided to preserve muscle strength[34]. Recently, growing numbers of data have started to arise, showing that resistance exercise is safe and does have some benefit for those with ALS[35]. A recent randomized, controlled study looked at ALS participants and the combined effects of moderate aerobic and isometric exercises programs[36]. Positive effects associated with exercise were found in terms of overall independence, oxygen consumption, and fatigue[36]. This study then concluded that moderate intensity exercise can be beneficial in preventing muscle atrophy and deconditioning due to inactivity, especially at earlier stages of ALS[36]. Using this research and the AMSC guidelines, a resistance exercise program that was individualized to Lou was developed to help him achieve his short term goals. At the end of the 4 weeks, using the RPE scale and MMT’s outcome measures, Lou’s strength was very similar to his initial assessment. Lou achieved: -4/5 - quads, 4/5 - hamstrings, -4/5 - gastrocnemius, -4/5 - biceps, -4/5 - triceps, 4/5 - wrist flexors/extensors with an RPE of 4-6. Lou was able to complete his short term goal and was feeling very motivated to continue his intervention program to further achieve his long-term goals.
| Short Term Goal 3 | Intervention: Type | Intervention: Frequency, Intensity, Timing[37] | Modifications for Intervention |
| In 4 weeks, Patient will independently engage in painting activities using compensatory strategies. ALS Functional Rating Scale (ALSFRS) will be used to measure patients function | Dexterity Training:
- Reaching and grasping different sized paint brushes and putting them into a jar |
15 minutes (or till mental/physical fatigue)
3 days minimum but recommended daily |
Coordination: using imagery to help visualize themselves performing the task.
Increased Irritability/difficulty: breaking up and practicing different aspects of the task Motivation: Educate patient |
Goal Discussion[edit | edit source]
Lou explained during the subjective assessment that he had a passion for painting but was struggling to pick up and hold the paint brush. Before starting his rehabilitation journey Lou made it a goal of his to be able to paint again. The research provides little evidence on physical therapy and the outcome of dexterity exercises for those with ALS. However, a recent systematic review showed the importance of taking a interdisciplinary approach when caring for these patients[38]. The involvement of both occupational therapy, physical therapy and many other health care providers can maximize independence, quality of life and function for those diagnosed with ALS[38]. Evidence shows that occupational therapy (OT) can help maintain hand dexterity through assistive devices and robotic assisted gloves[37]. Thus, if Lou’s dexterity training was ineffective in maintaining his griping and grasping abilities, it would be beneficial to refer him to an OT. Dexterity training commonly had participations perform reaching and grasping tasks for 15 minutes daily, which supports the intervention parameters that were provided for Lou[37]. At the end of the 4 weeks, Lou was able to maintain a similar ALSFRS score of 40 and showed more control and confidence in himself when engaged in his painting activities using compensatory strategies. These compensatory strategies, such as using foam tubing to increase the size of the paint brush handle, allowed him to accomplish his last short-term goal.
Although there weren’t any significant changes in Lou’s results, maintaining stable scores allowed him to achieve his short term goals. In other words, the intervention plan provided may have decreasing acute progression of Lou’s ALS. Lou expressed that he is motivated to continue his therapy sessions to further pursue his long term goals.
Long Term Goals:[edit | edit source]
| Long Term Goal 1 | Intervention: Type | Intervention: Frequency, Intensity, Timing | Modifications for Intervention |
| At 3 months post-diagnosis, the patient will be able to maintain independent ambulation in a safe manner, measured as a score of 45 or greater on the Berg Balance Scale[39] | Treadmill training with Body Weight Support[30]
1. Sit-to-stand 2. Standing Marches 3. Lateral walking 4. Standing Leg abductions |
2-3x per week for each type of exercise[29]
20-30 minute duration for aerobic exercise[29] 5 reps, 1-2 sets for balance exercises[40] |
Balance / Fear of Falling: patient can use a recumbent bike instead or place hands on the guide rails of the treadmill during walking. Balance exercises can be traded for seated balance exercises[40]
Muscle Weakness: have patient use AFO or implement non-invasive brain stimulation before exercise[42] Fatigue: decrease intensity or implement pacing by breaking exercise up into 5 or 10 minute segments[43] Motivation: play the patient’s favourite type of music during exercise[30] Dysarthria: provide signage for the patient to point to in order to indicate their RPE so that it does not need to be vocalized[43] |
Goal Discussion[edit | edit source]
This goal was chosen to reflect the problem of loss of ambulation which has significant consequences on independent functioning in the ALS population. The Berg Balance Scale was chosen to measure this goal because it is a scale that has significant cut offs which indicate risk of falls[39]. The goal was set to maintain a score of 45 or greater because scores less than 45 indicate a higher risk of falls[39]. The intervention for this goal includes a mix of ambulation practice as well as balance exercises. Though there is not very much evidence for aerobic training in the specific population with ALS, there is previous research which supports the use of body-weight supported treadmill training for other neurologically impaired populations such as multiple sclerosis[30]. In these trials, it was found that auditory cues also had a significant impact on gait improvement, specifically in the form of verbal and musical cues[30]. As for the balance exercises, there is also a limited amount of research on the ALS population specifically. A review of intervention measures for this population done in 2018 recommends that balance training is effective[40], but does not provide parameters. The Cochrane review on therapeutic exercise for individuals with ALS, however, does offer a recommendation of moderate load / moderate intensity exercise to promote strengthening without overworking the muscles[34]. Modifications for these interventions were found in various reviews of both medical and exercise interventions for the ALS population, and will depend on the severity of Lou’s symptoms as he progresses.
| Long Term Goal 2 | Intervention: Type | Intervention: Frequency, Intensity, Timing | Modifications for Intervention |
| At 3 months post-diagnosis, the patient will have maintained function of muscles involved in hand and foot movement as measured by a maintained score on the Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS)[34] within 3 points[44] | Functional exercises:
1. Fine motor exercise - practicing brush strokes with paintbrush, towel scrunches with toes 2. Gross motor exercise - pushing/pulling door closed/open, standing heel and toe raises 3. Gripping exercise - paintbrush squeezes, holding cup with water 4. Walking exercises - stepping over a pylon forward, backward, and sideways, stopping and turning drills |
Moderate intensity resistance exercise parameters[34]
15 reps[29] 1-2 sets[29]
RPE: 2-4 on the Borg CR10 Scale)[41] |
Balance / Fear of Falling: patient can perform exercises in a seated, supine, or prone position instead of standing[40]
Muscle Weakness: implement non-invasive brain stimulation before exercise[42]. Note that functional electrical muscle stimulation is NOT indicated for this population[45] Fatigue: decrease intensity or implement pacing by breaking exercise up into 5 or 10 minute segments[43] Motivation: play the patient’s favourite type of music during exercise[30] Dysarthria: provide signage for the patient to point to in order to indicate their RPE so that it does not need to be vocalized[43] |
Goal Discussion[edit | edit source]
This goal was chosen to address Lou’s concerns of the progressive muscle weakness in his upper and lower distal extremities. The outcome measure that was chosen was picked with intention of focussing on the effects that this progressive weakness has on Lou’s functional tasks. Though the ALSFRS may not be as specific to muscle strength as a manual muscle test, this scale allows room for consideration of compensatory strategies such as utilizing adjacent muscles in place of those which are experiencing greater degradation during functional movements. The Cochrane review of therapeutic exercise for individuals with ALS suggests that the ALSFRS is a better indicator of maintained function over long term periods of at least 3 months as opposed to manual muscle testing, which will likely decrease despite exercise efforts due to the progressive nature of this disease[34]. The goal specifies the maintenance of score within 3 points because the Minimal Important Difference of the scale is 3.24[44], meaning that a change of less than 3.24 points does not make a significant difference in the outcome. Functional exercises were chosen and prescribed to Lou in reflection of the ALSFRS by which his goal will be measured. The same Cochrane review also suggests that moderate intensity is appropriate for the ALS population to maintain muscle activity without overworking the muscles and further degrading them[34]. Therefore, American College of Sports Medicine (ACSM) guidelines[29] were followed for moderate intensity resistance exercises, which can also be applied to functional exercises that consist of strengthening activities (like those prescribed). Intervention modifications were found throughout several reviews and meta-analyses which discuss solutions for several of the impairments which complicate ADLs that are common in the ALS population. Again, these interventions will be used depending on how Lou progresses over the 3 months following his initial visit.
| Long Term Goal 3 | Intervention: Type | Intervention: Frequency, Intensity, Timing | Modifications for Intervention |
| At 3 months post-diagnosis, the patient will be able to maintain the ability to paint using a robotic-assistive glove. Manual dexterity of the hand will be measured by performing a 9 Hole Peg Test[46], on which the patient will score under 1 minute with use of the adaptive device[23] | Painting with a robotic-assisted glove[37] | 2-3 days/week[29]
Light to Moderate intensity[34] (RPE: 2-4 on the Borg CR10 Scale)[41] 15 minutes[37] |
Fatigue: decrease intensity or implement pacing by breaking exercise up into 5 or 10 minute segments[43]
Motivation: play the patient’s favourite type of music during exercise[30] Dysarthria: provide signage for the patient to point to in order to indicate their RPE so that it does not need to be vocalized[43] |
Goal Discussion[edit | edit source]
The purpose of this goal was to address Lou’s concern about his ability to paint. This goal can be considered multi-disciplinary, as it involves an adaptive device which can also be used with an Occupational Therapist[37]. A review on the effects of rehabilitation on the ALS population highlights the benefits to having an interdisciplinary team for support and expertise in each area of life that the disease affects[38]. Aside from introducing opportunity for collaboration with occupational therapists, this intervention also allows Lou to continue painting at the skill level which he has worked up to over the years. Though the use of this device would not be recommended to replace all fine motor exercises (as shown in Long Term Goal 2), Lou can use it to compensate for some of the weakness and motor control loss that he is experiencing in his hands[37]. An appropriate outcome measure for this goal is therefore the 9 Hole Peg Test, which can be used to assess manual dexterity during use of the robotic-assisted glove in comparison to baseline[46]. The leading research on this adaptive device suggests that 15 minute periods of use are beneficial in the ALS population[37], and the Cochrane review on exercise in this population suggests that moderate intensity exercise is most appropriate[34]. As stated for the above Long Term Goals, modifications for this intervention will depend on how Lou’s specific case progresses, but could involve fatigue levels, motivation levels, or communication capabilities. Suggested modifications have been found in various reviews and meta-analyses which discuss exercise in the ALS population.
When completed together, Lou's Long Term Goals will allow him to train in balance exercises and walking for prolonged independent ambulation, complete functional exercises for the maintained strength of his muscles during daily tasks, and utilize a robotic-assisted glove for the continuation of his painting hobby. Though these goals will not recover strength and motor control which has been lost throughout the disease progression, they will allow Lou to continue to participate in his daily activities and hobbies through maintained strength and compensatory strategies.
Technology - Mediated Tools[edit | edit source]
There are several different technology-mediated tools available for individuals with ALS that are intended to aid in treatment and/or assessment of functioning[47]. These tools include but are not limited to multidisciplinary telehealth consults, communication devices through eye tracking, brain-computer-interfaces, and different types of robotics aimed at improving mobility[47]. The use of certain devices by individuals depends on where they are in their disease progression. For example, the majority of the brain computer interface devices available are used by individuals who have lost the majority of their voluntary motor control.

For Lou, he has not progressed to advanced stages of ALS yet and has goals surrounding functioning and mobility, therefore the use of a robotic exoskeleton may be beneficial. Robotic exoskeletons are wearable units that are controlled by computer boards to power the system in order to maintain mobility and cognition[49]. There are various robotic exoskeletons available on the market for both upper and lower extremities, each providing different levels of assistance for the individual. Some of these products include the Hybrid Assistive Limb, KINARM, and ArmeoPower.


With these types of devices, patients are able to perform intensive, repetitive, goal-oriented exercises to facilitate improvement in strength and sensorimotor output[50]. A case study showed that using Armeo Power, which assists active movement across a large 3D workspace, showed significant improvement in strength related to shoulder abduction, elbow flexion and elbow extension in an individual suffering with flail arm ALS[47]. For Lou, the combination of the traditional rehabilitation approach for dexterity or locomotion training in conjunction with training with robotics can help him reach his goal of painting and being able to maintain his level of functioning. The use of robotics is also beneficial in a clinical setting as it can be used as an assessment tool as well as a treatment method, thus being able to quantify the sensory, motor, and cognitive impairments in patients which can be useful for therapeutic decision making[47].
There are various challenges associated with using robotic devices, including fatigue related to usage and the lack of global muscle activation. One study that investigated the feasibility of robotic exoskeletons (specifically the KINARM) use in the ALS population found that the most common reason that individuals were unable to complete the task was because the task was not attempted due to fatigue and/or motivation. In order to overcome this barrier, positioning of the machine should be considered[51]. With a modification of positioning, the machine was able to support more of the weight of the upper extremity and torso. Patients were also strapped into harnesses to aid in posture[51]. Another strategy that could be used is implementing a task hierarchy to reduce the number of tasks performed. Within the study there were a total of nine tasks that the participants were asked to perform, including a combination of sensorimotor and behavioural tasks[51]. If this type of treatment was to be used in a clinic setting, I think it would be important to consider tasks most relevant to patient goals and implement rest periods to avoid overexertion and keep motivation high.
Secondly, when using the device, only one body part can be trained at the same time which is not conducive to overall improvement[50]. ALS is usually a condition that affects the body bilaterally, therefore with the consideration of fatigability, it is likely that in a session only one body part would be able to be trained during a session. In order to manage this challenge, clinicians should consider the goals and needs of the patient. The priority in which areas are treated through the robotic exoskeleton should be based on function as well as patient goals.
Referrals[edit | edit source]
Multidisciplinary teams are vital in the management of ALS, due to the progressive and widespread effect of the disorder on multiple body systems[2]. Thus, it is important to view patients with a holistic lens, taking into consideration how aspects aside from physical body systems (ie. Musculoskeletal, respiratory, etc.) can be greatly impacted, especially in conditions such as ALS. Likewise, as physiotherapists who strive to deliver client-centred care, the International Classification of Functioning, Disability, and Health (ICF) model forms the basis of assessment, treatment, and management methods[2]. Accordingly, in addition to seeing a physical therapist, Lou would benefit from being referred to other health care professionals including a respiratory therapist (RT), occupational therapist (OT), and dietitian, among others, to address both physical and cognitive complications associated with the condition[2].
One of the major complications of ALS is respiratory insufficiency, including respiratory muscle weakness, ventilatory failure, and a decline in vital capacity [2]. This decline in respiratory functioning can progress to complications including truncated speech, dyspnea, fatigue, orthopnea, and inefficient breathing mechanics, and is the most frequent cause of death in ALS [2] [52]. Moreover, respiratory insufficiency is a strong predictor of quality of life due to its interference with routine and leisure daily activities and its association with complications such as the development of respiratory infections[2]. Thus, an important referral for Lou is to an RT. The importance of RTs in the management of ALS has been highlighted in various studies. For example, non-invasive ventilation (NIV), now considered the standard of care for respiratory-related complications in ALS, is a highly effective but under-utilized management strategy[52][53]. Its use has been associated with significant improvements in quality of life and survival rates and has added benefits when implemented early on, reinforcing the importance of Lou having an RT as part of his care team [54]. Even if Lou has yet to develop severe respiratory complications, it is never too early to adopt a proactive approach. Studies have found patients to be more compliant with NIV when an RT is part of the multidisciplinary clinic they attend, due to enhanced patient and caregiver education on its use and importance and the ability of the therapist to make individualized machine setting adjustments for patients, illustrating just one benefit of Lou seeing an RT[55]. In addition to implementing interventions such as NIV, RTs are important in the continual monitoring of respiratory function, such as evaluating forced vital capacity and vital capacity, to assess objective changes in status as well as patient-reported subjective symptoms, and treating them accordingly [56]. Thus, it seems reasonable to suggest that an important referral for Lou is to an RT, to minimize the lethal effects of respiratory dysfunction that are seen in ALS and maximize his quality of life for as long as possible.
Since there is currently no cure for ALS and it has a progressive course, OTs can serve as another key addition to the team involved in the care of individuals living with ALS [2]. An OT can help Lou maintain as much independence as possible for as long as possible by helping him find compensatory strategies for completing ADLs and leisure activities, modifying his home and work environments, and optimizing his use of and access to assistive devices. For example, an OT can help fit and educate Lou on using a robotic-assisted glove (RAG) device to help moderate the weakness he experiences, which is especially prevalent in his distal extremities[57]. Such a device will allow Lou to continue to use his hands in a natural manner, helping him retain functional independence and the ability to participate in hobbies such as painting[57]. Finding ways for Lou to continue to participate in activities he enjoys is highly beneficial for many reasons, one of which includes enhanced quality of life. Qualitative studies have highlighted that the struggle with integrating new and multiple assistive devices into everyday life hinders quality of life in individuals with ALS, because of issues such as accessibility and the fact that devices can change frequently and quickly as the disease progresses[58]. OTs can help patients navigate issues encountered with their new devices and help them adapt to the novel changes both physically and mentally. Having an OT within the multidisciplinary care team can also aid in setting practical and client-centered goals, which may enhance Lou's sense of autonomy and feelings of self-control, with the hopes of mitigating his feelings of depression[59]. Interestingly, RAG has also been found to induce neuroplastic changes in sensorimotor pathways and enhance subjective perceptions of performance in daily tasks in patients with ALS, lending further support for the beneficial effects of Lou seeing a professional who can manage assistive devices, such as an OT [57].
Because nutritional concerns, such as malnutrition and weight loss, are also common among those with ALS, another beneficial referral for Lou is to a dietitian[60]. In the management of ALS, dietitian's can improve the nutritional status of patients by providing accurate information and education on calorically sufficient diets, valuable supplements, food modifications to address chewing/swallowing difficulties, possible tube feeding methods, and overall safety of consuming and handling food[60]. Having Lou see a dietitian will help mitigate the progressively worsening factors that contribute to poor nutrition in individuals with ALS, and prevent complications such as excessive weight loss, muscle mass and strength loss, malnutrition, dehydration, choking, and aspiration[60][52]. Likewise, due to Lou’s hypertension, having a dietitian on his care team can help him make optimal decisions about the foods he consumes to ensure he feels as healthy as possible for as long as possible, while still enjoying what he likes. It is important to note that nutrition plays a large role in many other aspects of an individual's life, such as on their mood and cognition, in addition to physical body systems such as muscle mass. While it is true that weight loss in ALS is associated with a decreased survival rate, it is also associated with a reduction in quality of life, as malnutrition often leads to outcomes such as excessive fatigue, apathy, and depression, which hinders an individual's ability to engage in an enjoyable life[61]. Moreover, it has been noted that the problem of malnutrition is often overlooked in clinical practice, highlighting the importance of addressing this issue promptly in the case of Lou even if he has yet to show any observable impact of insufficient dietary intake [61]. Considering this and keeping the ICF model in mind, having Lou see a dietitian will not only help prevent the negative impact of nutritional problems on his physical bodily functioning, but can also help address the psychological and psychosocial problems that are often associated.
Referral Letter:
*Please note: this sample referral letter is directed toward Mr. Gherig's GP, for a formal referral to a respiratory therapist (RT).
To Dr. QueensU,
After a comprehensive physiotherapy assessment of Mr. Lou Gherig, whom you diagnosed with ALS, it is highly advised that he receive a referral to a respiratory therapist as well. Mr. Gherig would greatly benefit from seeing a respiratory therapist to assess, monitor, and mitigate the adverse effects of respiratory insufficiency that are starting to appear and will continue to worsen rapidly. Having Mr. Gherig see a respiratory therapist will allow me (lead physiotherapist) and the rest of the multidisciplinary care team to focus on our specialities to ensure he receives the best possible care for every aspect of his condition.
Thank you for your time and consideration.
Sincerely,
PT858
Discussion[edit | edit source]
Each of the standardized outcome measures were chosen based on the patient’s identified problems. The MAS was selected to assess the patient’s spasticity as it is a well-known and widely used test, even though its use in ALS lacks research[12]. When examining the patient’s loss in muscle strength, two options were outlined. Both MMT and MVIC have their pros and cons, which is why we decided to include both of them and allow readers to make decisions based on this. In terms of our patients reported decline in balance, there are a number of tests options apart from the BBS, such as the Tinetti Performance Oriented Mobility Assessment (POMA) or the Functional Reach Test (FRT)[2]. The POMA could still be a great test to examine balance in a patient with ALS, however it is known to be more reliable in the early or mid-stages of ALS[62]. The FRT was not used because it only assesses one simple task (standing and reaching), while the BBS allows us to see balance in a variety of tasks which can be important when considering that ALS affects different parts of the body to different degrees and at different times. Our patient had also identified that he struggled to walk with his dog, so we decided to use the 6MWT as an objective, simple, and inexpensive measure of walking capacity for our patient[22]. To examine hand dexterity in relation to the patient’s difficulty with painting recently, we decided to use the NHPT as a quick, easy to administer test[24]. There are many other many other tests to examine hand dexterity or function, however, most also lack evidence surrounding their specific use with ALS[24]. Finally, we used the ALSFRS to gain a more global understanding of our patient and his impairments. This test is quick, requires no training, and is a great way to assess progression of disease or response to treatment over a period of time[27][26].
Overall, while keeping clinical practice guidelines and the ICF model in mind, a multidisciplinary team is a non-negotiable when it comes to the management of conditions such as ALS [52][2]. Three beneficial referrals for Lou include a respiratory therapist (RT), occupational therapist (OT), and dietitian, along with him engaging in physiotherapy and seeing his general practitioner regularly[2] [56]. An RT can help assess, monitor, and manage the lethal respiratory complications that quickly develop in individuals with ALS, and help educate Lou and his family on effective management strategies, including the use of NIV[52] [54][53][55]. An RT is at the top of the list of referrals here, as respiratory insufficiency not only reduces quality of life but is also the main cause of death in patients with ALS [2][52]. OTs play another key role in helping patients manage their ALS, as they can address access to assistive devices, proper fitting of devices, and education on how to use assistive devices in differing contexts[57] [58][59]. A major benefit of referring Lou to an OT is to facilitate greater independence and practical goal-setting in the execution of routine activities of daily living, work, and leisure activities, to help address Lou’s activity and participation restrictions[2]. Lastly, Lou was referred to a dietitian to help manage the commonly encountered problems related to nutritional insufficiency seen in individuals with ALS, such as malnutrition and consequential muscle atrophy[2]. Moreover, addressing nutrition is vital while attempting to maintain as optimal a quality of life as possible in progressive conditions, as what we eat impacts all aspects of the body and mind[61]. Dietitian's can help identify Lou’s unique nutritional needs and formulate a strategic plan, establish expectations, and provide and implement strategies to use going forward[60][61][52].
Self-Study Questions[edit | edit source]
- Which of these is a phenotype / disease subtype of ALS?
- Reticulospinal ALS
- Limb / spinal onset ALS
- Relapsing remitting ALS
- Back dominant ALS
- Which of the following outcome measures serves as a predictor of survival in ALS and a prognostic biomarker?
- Berg Balance Scale
- Modified Ashworth Scale
- Amyotrophic Lateral Sclerosis Functional Rating Scale
- 6 Minute Walk Test
- Which of the following symptoms is more commonly seen in the early stages of bulbar-onset ALS?
- Difficulty with speech and swallowing
- Muscle weakness affecting the legs
- Loss of grip strength in the hands
- Muscle twitching (fasciculations) in the arms
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Kiernan MC, Vucic S, Cheah BC, Turner MR, Eisen A, Hardiman O, et al. Amyotrophic lateral sclerosis. The Lancet. 2011;377(9769):942–55. doi:10.1016/s0140-6736(10)61156-7
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 2.17 2.18 2.19 2.20 2.21 2.22 2.23 2.24 2.25 2.26 2.27 2.28 2.29 2.30 2.31 2.32 O’Sullivan B. S, Schmitz J. T, & Fulk D. G, editors. Physical Rehabilitation. 7th-ed. Philadelphia: F. A. Davis Company, 2019.
- ↑ Oskarsson B, Horton DK, Mitsumoto H. Potential environmental factors in amyotrophic lateral sclerosis. Neurologic Clinics. 2015;33(4):877–88. doi:10.1016/j.ncl.2015.07.009
- ↑ 4.0 4.1 4.2 About Als [Internet]. 2021 [cited 2023 May 12]. Available from: https://als.ca/what-is-als/about-als/
- ↑ National Institute of Neurological Disorders and Stroke. [Internet]. 2023 [cited 2023 May 12]. Available from: https://www.ninds.nih.gov/health-information/disorders/amyotrophic-lateral-sclerosis-als.
- ↑ 6.0 6.1 Marcu I R, Patru S, & Bighea A C. Diagnosis Particularities of Amyotrophic Lateral Sclerosis in an Elderly Patient. Current health sciences journal. 2018; 44(1): 92–96. https://doi.org/10.12865/CHSJ.44.01.16
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 Fang T, Al Khleifat A, Meurgey JH, Jones A, Leigh PN, Bensimon G, Al-Chalabi A. Stage at which riluzole treatment prolongs survival in patients with amyotrophic lateral sclerosis: a retrospective analysis of data from a dose-ranging study. Lancet Neurol. 2018 May;17(5):416-422. doi: 10.1016/S1474-4422(18)30054-1. Epub 2018 Mar 7. PMID: 29525492; PMCID: PMC5899963.
- ↑ Hu, N., Ji, H. Medications on hypertension, hyperlipidemia, diabetes, and risk of amyotrophic lateral sclerosis: a systematic review and meta-analysis. Neurol Sci 43, 5189–5199 (2022). https://doi.org/10.1007/s10072-022-06131-7
- ↑ 9.0 9.1 Ashworth scale / modified Ashworth Scale [Internet]. 2021 [cited 2023 May 12]. Available from: https://www.sralab.org/rehabilitation-measures/ashworth-scale-modified-ashworth-scale
- ↑ 10.0 10.1 Ansari NN, Naghdi S, Arab TK, Jalaie S. The interrater and intrarater reliability of the modified Ashworth scale in the assessment of muscle spasticity: Limb and Muscle Group effect. NeuroRehabilitation. 2008;23(3):231–7. doi:10.3233/nre-2008-23304
- ↑ 11.0 11.1 Harb A, Kishner S. Modified Ashworth Scale. In: StatsPearls [Internet]. StatsPearls Publishing; 2022 [cited 2023 May 12]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK554572/
- ↑ 12.0 12.1 Paganoni S, Cudkowicz M, Berry JD. Outcome measures in amyotrophic lateral sclerosis clinical trials. Clinical Investigation. 2014;4(7):605–18. doi:10.4155/cli.14.52
- ↑ Meldrum D, Cahalane E, Conroy R, Fitzgerald D, Hardiman O. Maximum voluntary isometric contraction: Reference values and clinical application. Amyotrophic Lateral Sclerosis. 2007 Feb 8;8(1):47–55. doi:10.1080/17482960601012491
- ↑ 14.0 14.1 14.2 Great Lakes ALS Study Group. A comparison of muscle strength testing techniques in amyotrophic lateral sclerosis. Neurology. 2003;61(11):1503–7. doi:10.1212/01.wnl.0000095961.66830.03
- ↑ Colombo R, Mazzini L, Mora G, Parenzan R, Creola G, Pirali I, et al. Measurement of isometric muscle strength: A reproducibility study of maximal voluntary contraction in normal subjects and amyotrophic lateral sclerosis patients. Medical Engineering & Physics. 2000;22(3):167–74. doi:10.1016/s1350-4533(00)00024-2
- ↑ Manual Muscle Test [Internet]. 2020 [cited 2023 May 12]. Available from: https://www.sralab.org/rehabilitation-measures/manual-muscle-test
- ↑ 17.0 17.1 17.2 Berg balance scale [Internet]. 2020 [cited 2023 May 12]. Available from: https://www.sralab.org/rehabilitation-measures/berg-balance-scale
- ↑ Schell WE, Mar VS, Da Silva CP. Correlation of falls in patients with amyotrophic lateral sclerosis with objective measures of balance, strength, and spasticity. NeuroRehabilitation. 2019;44(1):85–93. doi:10.3233/nre-182531
- ↑ 19.0 19.1 Moore JL, Potter K, Blankshain K, Kaplan SL, O’Dwyer LC, Sullivan JE. A core set of outcome measures for adults with neurologic conditions undergoing rehabilitation. Journal of Neurologic Physical Therapy. 2018;42(3):174–220. doi:10.1097/npt.0000000000000229
- ↑ 6 minute walk test [Internet]. 2013 [cited 2023 May 12]. Available from: https://www.sralab.org/rehabilitation-measures/6-minute-walk-test
- ↑ Sanjak M, Langford V, Holsten S, Rozario N, Patterson CG, Bravver E, et al. Six-minute walk test as a measure of walking capacity in ambulatory individuals with amyotrophic lateral sclerosis. Archives of Physical Medicine and Rehabilitation. 2017;98(11):2301–7. doi:10.1016/j.apmr.2017.04.004
- ↑ 22.0 22.1 Russo M, Lunetta C, Zuccarino R, Vita GL, Sframeli M, Lizio A, et al. The 6-min walk test as a new outcome measure in amyotrophic lateral sclerosis. Scientific Reports. 2020;10(1). doi:10.1038/s41598-020-72578-3
- ↑ 23.0 23.1 23.2 Czell D, Neuwirth C, Weber M, Sartoretti-Schefer S, Gutzeit A, Reischauer C. Nine Hole Peg test and transcranial magnetic stimulation: Useful to evaluate dexterity of the hand and disease progression in amyotrophic lateral sclerosis. Neurology Research International. 2019;2019:1–5. doi:10.1155/2019/7397491
- ↑ 24.0 24.1 24.2 Hayden CD, Murphy BP, Hardiman O, Murray D. Measurement of upper limb function in ALS: A structured review of current methods and Future Directions. Journal of Neurology. 2022;269(8):4089–101. doi:10.1007/s00415-022-11179-8
- ↑ Nine-hole peg test [Internet]. 2021 [cited 2023 May 12]. Available from: https://www.sralab.org/rehabilitation-measures/nine-hole-peg-test#multiple-sclerosis
- ↑ 26.0 26.1 26.2 26.3 Amyotrophic lateral sclerosis functional rating scale [Internet]. 2014 [cited 2023 May 12]. Available from: https://www.sralab.org/rehabilitation-measures/amyotrophic-lateral-sclerosis-functional-rating-scale
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 Lee M, McCambridge A. Clinimetrics: Amyotrophic lateral sclerosis functional rating scale-revised (ALSFRS-R). Journal of Physiotherapy. 2018;64(4):269–70. doi:10.1016/j.jphys.2018.07.005
- ↑ Madsen, L. S., Jeppesen, J., & Handberg, C. “Understanding my ALS”. Experiences and reflections of persons with amyotrophic lateral sclerosis and relatives on participation in peer group rehabilitation. Disability and Rehabilitation, 2019, 41(12), 1410–1418.
- ↑ 29.00 29.01 29.02 29.03 29.04 29.05 29.06 29.07 29.08 29.09 29.10 American College of Sports Medicine. ACSM's guidelines for exercise testing and prescription. Lippincott Williams & Wilkins; 2013 Mar 4
- ↑ 30.0 30.1 30.2 30.3 30.4 30.5 30.6 30.7 30.8 Bishnoi, A., Lee, R., Hu, Y., Mahoney, J. R., & Hernandez, M. E. Effect of Treadmill Training Interventions on Spatiotemporal Gait Parameters in Older Adults with Neurological Disorders: Systematic Review and Meta-Analysis of Randomized Controlled Trials. International Journal of Environmental Research and Public Health, 2022, 19(5), 2824.
- ↑ Tsekoura, M., Anastasopoulos, K., Kastrinis, A., & Dimitriadis, Z. What is most appropriate number of repetitions of the sit-to-stand test in older adults: a reliability study. Journal of Frailty, Sarcopenia and Falls, 2020, 5(4), 109–113.
- ↑ 32.0 32.1 Downs S, Marquez J, Chiarelli P. Normative scores on the Berg Balance Scale decline after age 70 years in healthy community-dwelling people: a systematic review. J Physiother. 2014 Jun;60(2):85-9.
- ↑ 33.0 33.1 Drory, V. E., Goltsman, E., Goldman Reznik, J., Mosek, A., & Korczyn, A. D. The value of muscle exercise in patients with amyotrophic lateral sclerosis. Journal of the Neurological Sciences, 2001, 191(1), 133–137.
- ↑ 34.0 34.1 34.2 34.3 34.4 34.5 34.6 34.7 34.8 34.9 Dal Bello-Haas, V., & Florence, J. M. Therapeutic exercise for people with amyotrophic lateral sclerosis or motor neuron disease. Cochrane Database of Systematic Reviews, 2013, 2013(5), CD005229–CD005229.
- ↑ Lui AJ, PT Byl NN. A systematic review of the effect of moderate intensity exercise on function and disease progression in amyotrophic lateral sclerosis. J Neurol Phys Ther 2009, 33, 68-87.
- ↑ 36.0 36.1 36.2 Merico, A., Cavinato, M., Gregorio, C., Lacatena, A., Gioia, E., Piccione, F., & Angelini, C. Effects of combined endurance and resistance training in Amyotrophic Lateral Sclerosis: A pilot, randomized, controlled study. European Journal of Translational Myology, 2018, 28(1).
- ↑ 37.0 37.1 37.2 37.3 37.4 37.5 37.6 37.7 Yamakawa, I., Yamada, A., Sonoda, Y., Wakita, K., Nishioka, T., Harada, Y., Ogawa, N., Kitamura, A., Sanada, M., Tani, T., Imai, S., & Urushitani, M. Occupational therapy using a robotic-assisted glove ameliorates finger dexterity and modulates functional connectivity in amyotrophic lateral sclerosis. Journal of Clinical Neuroscience, 2023, 107, 144–149.
- ↑ 38.0 38.1 38.2 Majmudar, S., Wu, J., & Paganoni, S. Rehabilitation in amyotrophic lateral sclerosis: Why it matters: ALS Rehabilitation. Muscle & Nerve, 2014, 50(1), 4–13.
- ↑ 39.0 39.1 39.2 Lowe R, Upadhyay A, Jackson K, Walker W, Khadir SA, Coh C, et al. Berg Balance Scale [Internet]. Physiopedia. [cited 2023 May 10]. Available from: https://www.physio-pedia.com/Berg_Balance_Scale
- ↑ 40.0 40.1 40.2 40.3 40.4 Bello-Haas VD. Physical therapy for individuals with amyotrophic lateral sclerosis: current insights. Degenerative Neurology. 2018 Jul 16;8:45-54. doi: 10.2147/DNND.S146949. PMID: 30890895; PMCID: PMC6065609.
- ↑ 41.0 41.1 41.2 Chukwuemeka U. Borg Rating of Perceived Exertion [Internet]. Physiopedia. 2023 [cited 2023 May 11]. Available from: https://www.physio-pedia.com/Borg_Rating_Of_Perceived_Exertion
- ↑ 42.0 42.1 Liscic RM, Alberici A, Cairns NJ, Romano M, Buratti E. From basic research to the clinic: innovative therapies for ALS and FTD in the pipeline. Mol Neurodegener. 2020 Jun 1;15(1):31. doi: 10.1186/s13024-020-00373-9. PMID: 32487123; PMCID: PMC7268618.
- ↑ 43.0 43.1 43.2 43.3 43.4 43.5 Shoesmith C, Abrahao A, Benstead T, Chum M, Dupre N, Izenberg A, et al. Canadian best practice recommendations for the management of amyotrophic lateral sclerosis. Canadian Medical Association Journal. 2020 Nov 16;192(46):1453–68.
- ↑ 44.0 44.1 Fournier CN, James V, Glass JD. Clinically meaningful change: evaluation of the Rasch-built Overall Amyotrophic Lateral Sclerosis Disability Scale (ROADS) and the ALSFRS-R. Amyotroph Lateral Scler Frontotemporal Degener. 2023 May;24(3-4):311-316. doi: 10.1080/21678421.2022.2153607. Epub 2022 Dec 7. PMID: 36476010.
- ↑ Sun XS, Liu WX, Chen ZH, Ling L, Yang F, Wang HF, Cui F, Huang XS. Repetitive Nerve Stimulation in Amyotrophic Lateral Sclerosis. Chin Med J (Engl). 2018 Sep 20;131(18):2146-2151. doi: 10.4103/0366-6999.240798. PMID: 30203787; PMCID: PMC6144859.
- ↑ 46.0 46.1 Wang YC, Bohannon RW, Kapellusch J, Garg A, Gershon RC. Dexterity as measured with the 9-Hole Peg Test (9-HPT) across the age span. Journal of Hand Therapy. 2015; 28(1):53-60
- ↑ 47.0 47.1 47.2 47.3 Pugliese R, Sala R, Regondi S, Beltrami B, & Lunetta C. Emerging technologies for management of patients with amyotrophic lateral sclerosis: from telehealth to assistive robotics and neural interfaces. Journal of neurology. 2022; 269(6):2910–2921. https://doi.org/10.1007/s00415-022-10971-w
- ↑ Morioka H, Hirayama T, Sugisawa T, et al. Robot-assisted training using hybrid assistive limb ameliorates gait ability in patients with amyotrophic lateral sclerosis. Journal of clinical neuroscience. 2022;99:158-163. doi:10.1016/j.jocn.2022.02.032
- ↑ Gorgey A S. Robotic exoskeletons: The current pros and cons. World journal of orthopedics. 2018; 9(9): 112–119. https://doi.org/10.5312/wjo.v9.i9.112
- ↑ 50.0 50.1 50.2 Calabrò R, Russo M, Naro A, Milardi D, Balletta T, Leo A, et al. Who May Benefit From Armeo Power Treatment? A Neurophysiological Approach to Predict Neurorehabilitation Outcomes. PM & R. 2016; 8(10): 971–978. https://doi.org/10.1016/j.pmrj.2016.02.004
- ↑ 51.0 51.1 51.2 51.3 Simmatis L, Atallah G, Scott S H, & Taylor S. The feasibility of using robotic technology to quantify sensory, motor, and cognitive impairments associated with ALS. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration. 2019; 20(1-2): 43–52. https://doi.org/10.1080/21678421.2018.1550515
- ↑ 52.0 52.1 52.2 52.3 52.4 52.5 52.6 Miller RG, Jackson CE, Kasarskis EJ, England JD, Forshew D, Johnston W, et al. Practice parameter update: the care of the patient with amyotrophic lateral sclerosis: drug, nutritional, and respiratory therapies (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2009;73(15):1218–26.
- ↑ 53.0 53.1 Bourke SC, Bullock RE, Williams TL, Shaw PJ, Gibson GJ. Noninvasive ventilation in ALS: Indications and effect on quality of life. Neurology. 2003;61(2):171–7.
- ↑ 54.0 54.1 Lechtzin N, Scott Y, Busse AM, Clawson LL, Kimball R, Wiener CM. Early use of non-invasive ventilation prolongs survival in subjects with ALS. Amyotrophic lateral sclerosis. 2007;8(3):185–8.
- ↑ 55.0 55.1 Kareus SA, Kagebein S, Rudnicki SA. The importance of a respiratory therapist in the ALS clinic. Amyotrophic lateral sclerosis. 2008;9(3):173–6.
- ↑ 56.0 56.1 Andersen PM, Abrahams S, Borasio GD, de Carvalho M, Chio A, Van Damme P, et al. EFNS guidelines on the clinical management of amyotrophic lateral sclerosis (MALS) - revised report of an EFNS task force. European journal of neurology. Received 16 November 2010 Accepted 12 July 2011. 2012;19(3):360–75.
- ↑ 57.0 57.1 57.2 57.3 Yamakawa I, Yamada A, Sonoda Y, Wakita K, Nishioka T, Harada Y, et al. Occupational therapy using a robotic-assisted glove ameliorates finger dexterity and modulates functional connectivity in amyotrophic lateral sclerosis. Journal of clinical neuroscience. 2023;107:144–9.
- ↑ 58.0 58.1 Soofi AY, Dal Bello-Haas V, Kho ME, Letts L. The impact of rehabilitative interventions on quality of life: a qualitative evidence synthesis of personal experiences of individuals with amyotrophic lateral sclerosis. Quality of life research. 2018;27(4):845–56.
- ↑ 59.0 59.1 Arbesman M, Sheard K. Systematic review of the effectiveness of occupational therapy-related interventions for people with amyotrophic lateral sclerosis. The American journal of occupational therapy. 2014;68(1):20–6.
- ↑ 60.0 60.1 60.2 60.3 Guillemin L, Hofstede J, Anderson T, Walkeden H, Schellenberg K, Kang E, et al. Perceptions and experiences of nutrition interventions in individuals with amyotrophic lateral sclerosis (ALS) and their caregivers. Canadian journal of dietetic practice and research. 2022;83(4):193–7.
- ↑ 61.0 61.1 61.2 61.3 Korner S, Hendricks M, Kollewe K, Zapf A, Dengler R, Silani V, et al. Weight loss, dysphagia and supplement intake in patients with amyotrophic lateral sclerosis (ALS): impact on quality of life and therapeutic options. BMC neurology. 2013;13(1):84–84.
- ↑ Kloos AD, Bello-Haas VD, Thome R, Cassidy J, Lewis L, Cusma T, et al. Interrater and intrarater reliability of the Tinetti balance test for individuals with amyotrophic lateral sclerosis. Journal of Neurologic Physical Therapy. 2004;28(1):12–9. doi:10.1097/01.npt.0000284773.87060.c8