Heterotopic Ossification: Difference between revisions
No edit summary |
No edit summary |
||
| Line 69: | Line 69: | ||
*all have low specificity in early stage of disease process | *all have low specificity in early stage of disease process | ||
*better for detection in later stages when ossification is more pronounced | *better for detection in later stages when ossification is more pronounced | ||
*MRI and CT are valuable pre-operative tools to determine relationship to blood vessels and peripheral nerve structures [[Image: | *MRI and CT are valuable pre-operative tools to determine relationship to blood vessels and peripheral nerve structures | ||
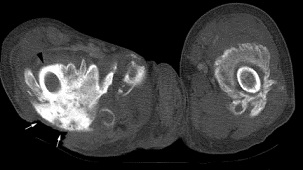
*HO demonstrable on radiographs 4-6 weeks post-injury [[Image:HO_MRI.jpg|frame|right|MRI showing heterotopic ossification (arrows) ]] | |||
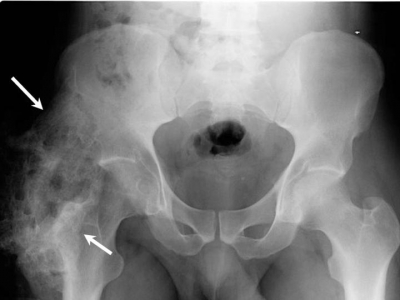
Prostaglandin E2: (PGE2) | [[Image:HO_CT_scan.jpg|left|CT scan showing heterotopic ossification of proximal femur]] | ||
Prostaglandin E2: (PGE2) | |||
*monitior PGE2 excretion in 24-hour urinalysis | *monitior PGE2 excretion in 24-hour urinalysis | ||
*PGE2 felt to be reliable bone marker for early detection and determining treatment efficacy | *PGE2 felt to be reliable bone marker for early detection and determining treatment efficacy | ||
*A sudden increase is an indication for bone scintigraphy | *A sudden increase is an indication for bone scintigraphy | ||
Revision as of 23:36, 22 March 2011
Original Editors - Bruce Tan from Bellarmine University's Pathophysiology of Complex Patient Problems project.
Lead Editors - Your name will be added here if you are a lead editor on this page. Read more.
Definition/Description[edit | edit source]
Heterotopic Ossification (HO) refers to the formation of lamellar bone inside soft tissue structures where bone does not normally exist. This process can occur in structures such as the skin, subcutaneous tissue, skeletal muscle, and fibrous tissue adjacent to bone. In more rare forms, HO has also been described in the walls of blood vessels and intra-abdominal sites such as the mesentery.[1]
Research suggests four factors which contribute to formation of heterotopic bone: 1) inciting event (usually trauma), 2) a signal from the site of injury, 3) a supply of mesenchymal cells whose genetic machinery is not fully committed, 4) an environment which is conducive to the continued formation of new bone.[1] These factors are discussed more indepth in the Etiology/Causes section.
HO was fisrt described by by Patin in 1692 while working with children diagnosed with myositis ossificans progressiva.[2] It was not until 1918 when Dejerine & Ceillier detailed the anatomical, clinical, and histological features of ectopic bone formation in soldiers who sustained spinal injuries during World War I.[3]
Prevalence[edit | edit source]
add text here
Characteristics/Clinical Presentation[edit | edit source]
Clinical signs and symptoms of HO may appear as soon as 3 weeks or up to 12 weeks after initial musculoskeletal trauma, spinal cord injury, or other precipitating event.[4]
Findings which may suggest the presence of HO are as follows:[3]
- joint swelling and warmth (initial phase)
- joint pain
- palpable mass, firm (late stage)
- decreased ROM of effected joint
- the initial inflammatory phase of HO may mimic other pathologies such as cellulitis, thrombophlebitis, osteomyelitis, or a tumorous process.[2]
Associated Co-morbidities[edit | edit source]
add text here
Medications[2][edit | edit source]
Medications are prescribed to patients who are at risk for developing heterotopic ossification for preventative measures and also to aid in the treatment after formation of heterotopic lesions. The two types of medications show to have both prophylactic and treatment benefits are as follows:
- Non-steroidal anti-inflammatory drugs: NSAIDS
Indomethacin more specifically-two fold action
- inhibition of the differentiation of mesenchymal cells into osteogenic cells (direct)
- inhibition of post-traumatic bone remodelling by supression of prostoglandin-mediated response (indirect)
Anti-inflammatory properties
- Biphosphonates:
Three-fold action
- inhibition of calcium phosphate precipitation
- slowing of hydroxyapatite crystal aggregation
- inhibition of the transformation of calcium phosphate to hydroxyapatite
Diagnostic Tests/Lab Tests/Lab Values[2][edit | edit source]
Three-phase bone scinitigraphy:
- both diagnostic and therapeutic follow-up purposes
- most sensitive imaging modality for early detection
- Phases 1 and 2 are indicative of hyperaemia and blood pooling (precursors to process of HO)
- Usually positive after 2-4 weeks
- Serial bone scans used to monitor metabolic activity of HO to determine optimal timing for surgical resection and to predict postoperative occurrence.
Radiography, MRI, CT Scan:
- all have low specificity in early stage of disease process
- better for detection in later stages when ossification is more pronounced
- MRI and CT are valuable pre-operative tools to determine relationship to blood vessels and peripheral nerve structures
- HO demonstrable on radiographs 4-6 weeks post-injury File:HO MRI.jpgMRI showing heterotopic ossification (arrows)
Prostaglandin E2: (PGE2)
- monitior PGE2 excretion in 24-hour urinalysis
- PGE2 felt to be reliable bone marker for early detection and determining treatment efficacy
- A sudden increase is an indication for bone scintigraphy
Ultrasonography:
- earlier detection than conventional ragiograph
- local signs of inflammation in SCI patients are suggestive of HO
- high sensitivity and specificity for early detection of HO 1-week post-THR.
Etiology/Causes[3][edit | edit source]
Systemic Involvement[edit | edit source]
add text here
Medical Management (current best evidence)[edit | edit source]
add text here
Physical Therapy Management (current best evidence)[edit | edit source]
add text here
Alternative/Holistic Management (current best evidence)[edit | edit source]
add text here
Differential Diagnosis[edit | edit source]
add text here
Case Reports/ Case Studies[edit | edit source]
- Kennedy MA, Sama AE, Sigman M. Tibiofibular syndesmosis and ossification. case report: sequelae of ankle sprain in an adolescent football player. Journ of Emer Med 2000; 18: 233-240.
- Bovo G, Romano F, Perego E, Franciosi C et al. Heterotopic mesenteric ossification ("intraabdominal myositis ossificans"): a case report. International Journal of Surgical Pathology 2004; 12: 407-409.
- Brower RS, Vickroy NM. A case of psoas ossification from the use of BMP-2
for posterolateral fusion at L4–L5.Spine 2008; 18: 653-655.
Resources
[edit | edit source]
add appropriate resources here
Recent Related Research (from Pubmed)[edit | edit source]
Failed to load RSS feed from http://eutils.ncbi.nlm.nih.gov/entrez/eutils/erss.cgi?rss_guid=1NOY_AEF8gNY3HRCwoXdtFZj9G-OSG0TNDAFjiqNOh_xvySEXY|charset=UTF-8|short|max=10: Error parsing XML for RSS
References[edit | edit source]
- ↑ 1.0 1.1 McCarthy EF, Sundaram M. Heterotopic ossification: a review. Skeletal Radiol 2005; 34: 609-619.
- ↑ 2.0 2.1 2.2 2.3 Bossche LV, Vanderstraeten G. Heterotopic ossification: a review. J Rehabil Med 2005; 37: 129-136.
- ↑ 3.0 3.1 3.2 Pape HC et al. Current concepts in the development of hetetrotopic ossification. Journ Bone and Joint Surg 2004; 86: 783-787.
- ↑ Shehab D, Elgazzar AH, Collier BD. Heterotopic ossification. Jour of Nuclear Medicine 2002; 43: 346-353.