Subarachnoid Hemorrhage (SAH)
Original Editor - Your name will be added here if you created the original content for this page.
Lead Editors
Introduction[edit | edit source]
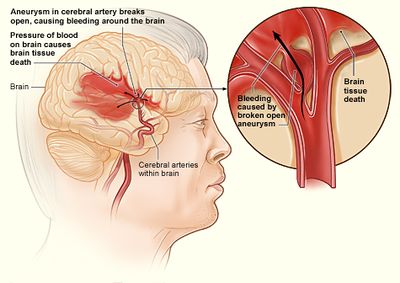
A subarachnoid hemorrhage results from an aneurysmal rupture or traumatic head injury. SAH occurs in the subarachnoid space between the arachnoid membrane and the pia mater that surrounds the brain.
15% of acute strokes are hemorrhagic strokes which are caused by bursting of a blood vessel i.e. acute hemorrhage. There are two main types of hemorrhagic strokes, intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH). SAH account for about 5% of all strokes[1].
Patients typically present complaining of a severe headache; however, only 10% of patients presenting to the emergency department complaining of a thunderclap headache end up having a SAH. Associated symptoms may include neck pain, nausea/vomiting, and photophobia[2].
Because of the very high mortality, an interprofessional team that includes a radiologist, neurosurgeon, neurologist, intensivist and ICU nurses are ideal. Unfortunately, despite optimal case, the majority of patients die within 30 days. Even those who survive are left with severe complications that are disabling[2]. Worldwide SAH incidence and its decline show large regional differences and parallel the decrease in blood pressure and smoking prevalence.[3]
Clinically Relevant Anatomy[edit | edit source]
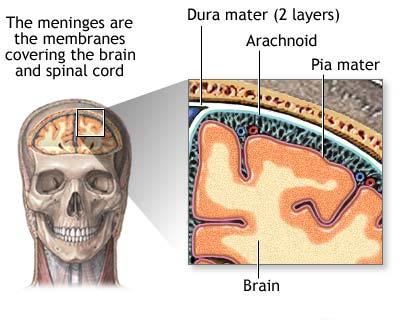
The brain and spinal cord are enveloped within three layers of membrane collectively known as the meninges, with the cranial meninges specifically referring to the section that covers the brain. From superficial to deep, the three layers are[4]
- dura mater - dense connective tissue and adheres to the inner surface of the skull and vertebrae
- arachnoid mater - thin wispy membrane that lies just deep to the dura and is superficial to the pia
- pia mater - the very thin, clear membrane that directly adheres to the surface of the brain and spinal cord.
Forming from these layers are three clinically significant spaces, or potential spaces (sometimes called cavities): the epidural, subdural and subarachnoid spaces, from superficial to deep. The chief function of the meninges is to protect the contents of the brain and spinal cord.
Subarachnoid hemorrhage is defined as blood between the arachnoid membrane and pia membrane, both layers of the dura mater.
Ninety percent of primary subarachnoid hemorrhages arise from congenitally derived arterial outpouchings (berry aneurysms) that lie at bifurcations of the major components of the circle of Willis. The most common sites are the carotid-posterior communicating and the anterior cerebral-anterior communicating artery junctions. Defects in the elastic membrane and media of the arteries are considered the basis for berry aneurysm formation[5].
Traumatic SAH: during the impact of an accident, the brain crashes back and forth inside the skull tearing blood vessels.
Epidemiology[edit | edit source]
SAH accounts for 5% of strokes[6]
• Most caused by ruptured saccular aneurysms
• SAH affects approximately 30,000 individuals annually in the US
• African American > Caucasian
• Women > Men
• Mean age at aneurysmal rupture is 55 yrs
• Mortality rates are reported between 30-40%
• Risk of permanent disability is reported as approximately 50%
Risk Factors[edit | edit source]
Risk factors for Formation of Aneurysms
- HTN
- Smoking
- Chronic alcohol consumptions
- Female sex
- 20% of individuals with have multiple
- Genetic conditions
- Family history Aneurysms
- Location of aneurysm
Risk Factors for Rupture of Aneurysms
- Race
- Hispanic ethnic group
- HTN
- Current smoking
- Alcohol abuse
- Use of sympathomimetic drugs
- Aneurysms larger than 7mm
- Location of aneurysm[6]
Mechanism of Injury / Pathological Process[edit | edit source]
Most SAH are caused by berry aneurysm - intracranial aneurysms are thin-walled protrusions from an intracranial artery that often has a thin (or absent) tunica media as well as an absent internal elastic lamina. Often hemodynamic stress leads to excessive wear on the wall of these arteries leading to turbulent blood flow within the vessel which causes structural fatigue and aneurysm development. Hypertension, cigarette smoking and a variety of connective tissue diseases can also contribute to arterial wall breakdown.[2]
Traumatic SAH usually occurs near the site of a skull fracture and intracerebral contusion. Radiologic clues of a traumatic origin include localized bleeding in a superficial sulcus, an adjacent skull fracture, and a cerebral contusion with external evidence of traumatic injury.[2]
The inciting event releases blood into the subarachnoid space. Blood close to the brain surface is an irritant, and many complications of subarachnoid hemorrhage are due to irritant effect of blood on the brain. Responses include
- seizures
- vasospasm
- confusion.
Subarachnoid hemorrhage has effects outside the brain
- neurogenic pulmonary edema
- neurogenic stunned myocardium.
- Blood products eventually circulate with cerebral spinal fluid and in many cases impede normal fluid clearance resulting hydrocephalus. If hydrocephalus is not treated in timely fashion fluid collection in the ventricular system will eventually create enough pressure to cause brain herniation syndromes and possible death. Neurosurgical intervention should be the priority when hydrocephalus is identified.[7]
Clinical Presentation[edit | edit source]
The SAH is typically a cataclysmic event, heralded by severe headache, meningeal signs and neurologic dysfunction. About one-half of patients with aneurysmal rupture experience "sentinel headaches" days to weeks before a major hemorrhage. Diagnosis at this stage may permit treatment before the occurrence of a devastating neurologic event. Clinical suspicion of subarachnoid hemorrhage should be confirmed by computed tomographic evaluation[8].
- A typical presenting symptom is a thunderclap headache eg “worst headache of my life.” A headache often is associated with nausea, vomiting, and diplopia.
- Often signs of meningismus are present due to irritant blood spreading into the fourth ventricle and further down the spinal cord irritating nerves and causing neck and back pain.
- Cranial nerve deficits can occur.
- The presence of focal deficit increases the grade of subarachnoid hemorrhage and changes the perspective of post-event recovery.
- Patients with a high-grade subarachnoid hemorrhage quite often present in a state of coma that calls for a quick evaluation and urgent treatment, as the coma can be reversible.
- Patients could have intraocular hemorrhage. [7]
Diagnostic Procedures[edit | edit source]
- A CT scan is the first tool in diagnosis.
- In the presence of a typical history and a normal CT scan, lumbar puncture should be performed with spectrophotometry analysis of the supernatant.
- After subarachnoid hemorrhage has been confirmed, four-vessel angiography should be performed as soon as possible.
- The surgical treatment of subarachnoid hemorrhage caused by ruptured aneurysm is the method of choice[9].
Physiotherapy[edit | edit source]
Physiotherapy begins in the ICU. Recent studies point to the importance of early ICU mobilisation and rehabilitation. Factors to consider when doing this include • Status of aneurysm • Intracranial pressure/ Cerebral perfusion pressure • EVD • Vasospasm —> DCI • Hemodynamic Instability • Ventilatory Support.[6]
A few key points to remember with therapy [6]
• Evaluation and intervention are intertwined
• Attention capacity and sustained attention play a big role in movement dysfunction
• Setting appropriate challenge level is key
• Movement errors are valuable for motor learning
A 2017 study[6] found that early rehab produced : higher levels of mobility earlier, higher levels of mobility on discharge
• Average level of mobility achieved on day 7 by the control group was bedrest with head of bed 60 degrees; early rehab: edge of bed
• No increased risk of adverse events for the early rehab group
The core finding of the a 2017 study "Effect of early mobilization and rehabilitation on complications in aneurysmal subarachnoid hemorrhage" was that early mobilization and rehabilitation after aSAH is feasible and safe. Less clinical vasospasm occurred in the early rehab group, and a risk reduction of greater than 30% for severe vasospasm for each step of mobilization achieved during the first 4 days after aneurysm repair. Despite early mobilization, no reduction in complications including respiratory tract/pulmonary infections, thromboembolic events, and death. For details on study see ref.[10]
Other studies are also currently underway looking to this earlier rehabilitation eg a study to investigate the the safety and feasibility of early intensive physical therapy for patients diagnosed with subarachnoid hemorrhage with intervention beginning in the neurological Intensive Care Unit (ICU) and continuing for 30 days or hospital discharge[11]. This 50 minute presentation gives some great rehabilitation tips and advice for therapists and clients post discharge.
Intensive rehabilitation, with physical, occupational and speech therapy will help recovery – which may be slow and uneven.
Management / Interventions[edit | edit source]
Initial treatment of patients with spontaneous subarachnoid hemorrhage includes resuscitation and/or stabilization, management of acute effects of the hemorrhage, and prompt referral for neurosurgical treatment[8].
Prognosis[edit | edit source]
Unfortunately, SAH is often associated with a poor outcome. Nearly half of patients presenting with a SAH caused by an underlying aneurysm die within 30 days and a third of those who survive have complications. Roughly half of the patients who have had a SAH suffer from some neurocognitive impairment that impacts their quality of life. Over 60% report ongoing and recurring headaches[2].
Conclusion[edit | edit source]
Aneurysm rupture is very dangerous, being fatal in 40% or more, with stroke-like deficits in half the remainder – in these the damage to brain tissue caused by the original haemorrhage or delayed blood vessel spasm may cause speech disturbance, weakness down one side of the body or double vision.
Intensive rehabilitation, with physical, occupational and speech therapy will help recovery – which may be slow and uneven.
Patients in the immediate aftermath of a subarachnoid haemorrhage should not
- smoke
- drive until given permission to do so by their doctor
- climb ladders, swim unaccompanied or do other activities which may be dangerous if they have a fit.
Patients should gradually be able to resume their normal lives as they feel able to do so. Any doubts (such as when to return to work, or the advisability of resuming sporting activities) should be discussed with the doctor. It is essential clients maintain a regimen of regular exercise, and continue medication as advised by the doctor, especially if treatment for high blood pressure is needed.
About 10% of strokes are due to subarachnoid haemorrhage. Recurrence is unlikely once the aneurysm has been treated. Patients can now be checked from time to time with special CT or MRI scans to make sure they do not develop further aneurysms. This service is also available to relatives where there is a family history of aneurysms[13]
References[edit | edit source]
- ↑ Tadi P, Lui F. Acute Stroke (Cerebrovascular Accident). Available from: https://www.ncbi.nlm.nih.gov/books/NBK535369/ (last accessed 2.1.2020)
- ↑ 2.0 2.1 2.2 2.3 2.4 Kairys N, Garg M. Acute Subarachnoid Hemorrhage (SAH). InStatPearls [Internet] 2019 May 4. StatPearls Publishing. Available from: https://www.ncbi.nlm.nih.gov/books/NBK518975/ (last accessed 2.1.2020)
- ↑ Etminan N, Chang HS, Hackenberg K, de Rooij NK, Vergouwen MD, Rinkel GJ, Algra A. Worldwide incidence of aneurysmal subarachnoid hemorrhage according to region, time period, blood pressure, and smoking prevalence in the population: A systematic review and meta-analysis. JAMA neurology. 2019 May 1;76(5):588-97. Available from:https://jamanetwork.com/journals/jamaneurology/fullarticle/2722652 (last accessed 2.1.2020)
- ↑ Ghannam JY, Al Kharazi KA. Neuroanatomy, Cranial Meninges. Available from:https://www.ncbi.nlm.nih.gov/books/NBK539882/ (last accessed 2.1.2020)
- ↑ DISORDERS OF THE NERVOUS SYSTEM - REEVES & SWENSON Chapter 27. Cerebrovascular disorders Available from:https://www.dartmouth.edu/~dons/part_3/chapter_27.html#chpt_27_ICH (last accessed 2.1.2020)
- ↑ 6.0 6.1 6.2 6.3 6.4 Mary Beth Holmes PT, DPT, NCS: Boston University Stacey Maguire PT, DPT, NCS: Beth Israel Deaconess Medical Center Jon Robinson PT, DPT, NCS: University of Vermont Medical Center Feb 2018 Aneurysmal subarachnoid hemorrhage: medical management and role of PT in the acute setting Available from:https://cdn.ymaws.com/www.acutept.org/resource/resmgr/csm/CSM_2018/2018_Handouts/Holmes_Mary_Beth_-_Aneurysma.pdf (last accessed 3.1.2020)
- ↑ 7.0 7.1 Ziu E, Mesfin FB. Subarachnoid Hemorrhage. InStatPearls [Internet] 2019 Feb 19. StatPearls Publishing. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441958/ (last accessed 3.1.2020)
- ↑ 8.0 8.1 Sawin PD, Loftus CM. Diagnosis of spontaneous subarachnoid hemorrhage. American family physician. 1997 Jan;55(1):145-56. Available from:https://www.ncbi.nlm.nih.gov/pubmed/9012274 (last accessed 3.1.2020)
- ↑ Krajickova D. Clinical picture and diagnosis of subarachnoid hemorrhage caused by a ruptured aneurysm. Sbornik vedeckych praci Lekarske fakulty Karlovy univerzity v Hradci Kralove. Supplementum. 1995;38(1):75-9. Available from:https://www.ncbi.nlm.nih.gov/pubmed/9441375 (last accessed 3.1.2020)
- ↑ Karic T, Røe C, Nordenmark TH, Becker F, Sorteberg W, Sorteberg A. Effect of early mobilization and rehabilitation on complications in aneurysmal subarachnoid hemorrhage. Journal of neurosurgery. 2017 Feb 1;126(2):518-26. Available from: https://thejns.org/view/journals/j-neurosurg/126/2/article-p518.xml (last accessed 3.1.2020)
- ↑ NIH Clinincal Trials Early intensive physiotherapy in SAH Available from: https://clinicaltrials.gov/ct2/show/NCT02675985 (last accessed 3.1.2020)
- ↑ A Cohen Gadol Rehabilitation After Subarachnoid Hemorrhage: How Can We Accelerate Recovery Available from:https://www.youtube.com/watch?v=d9Rxv0h6wZw&app=desktop (last accessed 3.1.2020)
- ↑ Brain Foundation SAH Available from: https://brainfoundation.org.au/disorders/subarachnoid-haemorrhage/ Available from:https://brainfoundation.org.au/disorders/subarachnoid-haemorrhage/ (last accessed 3.1.2020)