Stroke Medical Management
Original Editor - Naomi O'Reilly
Top Contributors - Naomi O'Reilly, Kim Jackson, Lucinda hampton, Admin, Simisola Ajeyalemi, 127.0.0.1, Karen Wilson, Claire Knott, Rucha Gadgil and Wanda van Niekerk
Introduction[edit | edit source]
The team approach to stroke management starts with Emergency Medical Services and continues at a hospital within a Stroke System of Care capable of delivering Acute Stroke Care in the Emergency Department and in a dedicated Stroke Unit. The goal for the acute medical management of patients with stroke is to stabilize the patient and to complete initial evaluation and assessment, including imaging and laboratory studies, within a short time frame. Critical decisions focus on the need for intubation, blood pressure control, and determination of risk/benefit for thrombolytic intervention.[1] Patients presenting with Glasgow Coma Scale scores of 8 or less or rapidly decreasing Glasgow Coma Scale scores, require emergent airway control via intubation[2].
Goals of Emergency Management [3]
- Support Vital Functions
- Restore Cerebral Circulation
- Reduce Neurological Deficits
- Prevent Progression and Cell Death
- Restore Patient to Optimal Level of Pre-Stroke Function
| American Stroke Association Stroke Chain of Survival | |
| Detection |
Patient/Bystander or EMS recognition of Stroke Signs & Symptoms |
| Dispatch |
EMS Activation; Priority Dispatch & Response |
| Delivery |
Prompt Triage - In the interest of saving time, some EMS actions can be accomplished enroute. Limit assessments / Care on scene to those that are urgently needed or clearly indicated by the patients presentation. Transport to most appropriate Stroke Hospital Prehospital Notification |
| Door |
Immediate Emergency Department triage to high-acuity area |
| Data |
Prompt Emergency Department Evaluation. Stroke Team Activation Lab Studies and Brain Imaging Time barriers at the hospital which should be minimised:
|
| Decision |
Diagnosis and determination of most appropriate therapy; Discussion with Patient and Family |
| Drug |
Administration of appropriate Drugs / Treatment / Interventions <span style="white-space: nowrap;" />Tissue Plasminogen Activator (tPa) for the treatment of acute ischemic stroke must be given within 4.5 hours from the onset of symptoms. Ideally, tPA should be administered (“door to drug”) in less than 60 minutes after Emergency Department arrival. Evidence confirms that the sooner tPA is administered, the more optimal the results. |
| Disposition |
Timely Admission to Stroke Unit, ICU, or Transfer Outcomes are best when patients are admitted directly to and receive care in a dedicated stroke unit, where recovery and rehab can begin immediately. Hospitals without full stroke care capabilities should have official transfer agreements with hospitals offering a full complement of stroke care, and patients should be transferred as soon as reasonably possible. |
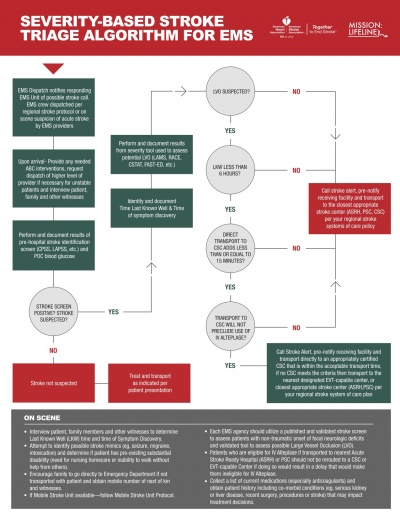
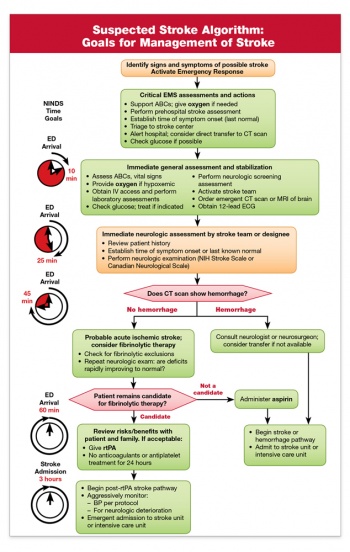
Suspected Stroke Algorithm[edit | edit source]
There are a number of Stroke Management Algorithms used both by the EMS Services and Acute Hospitals in order to effectively triage and manage the flow of patients suspected of Stroke to improve access to best possible treatments available. The new Severity-based Stroke Triage Algorithm developed by the American Heart Association/American Stroke Association provides ambulance crews with information and tools to better identify a stroke, assess a patient’s condition and determine the best hospital for specific treatment needs[3].
|
American Heart Association New Severity Based Stroke Triage Algorithm |
American Stoke Association Suspected Stroke Algorithm Goals for Management |
Imaging[edit | edit source]
According to Prasad (2011) brain imaging should be performed immediately for patients with persistent neurological symptoms if any of the following apply[4]:
- Indication for thrombolysis or early anticoagulation.
- On Anticoagulant Treatment
- Known Bleeding Tendency
- Depressed Level of Consciousness (GCS below 13)
- Severe Headache at onset of Stroke Symptoms
- Papilloedema, Neck Stiffness, Subhyaloid Hemorrhage or Fever
Patients with acute stroke without the above indications for immediate brain imaging, scanning should be performed within 24 hours after onset of symptoms.
Level I Clinical Guideline Recommendations
- Emergency imaging of the brain is recommended before initiating any specific treatment for acute stroke. In most instances, nonenhanced CT will provide the necessary information to make decisions about emergency management. (Unchanged from the 2013 Guideline)[2]
- If Endovascular Therapy is contemplated, a noninvasive intracranial vascular study is strongly recommended during the initial imaging evaluation of the acute stroke patient but should not delay intravenous r-tPA if indicated. For patients who qualify for intravenous r-tPA according to guidelines from professional medical societies, initiating intravenous r- tPA before noninvasive vascular imaging is recommended for patients who have not had noninvasive vascular imaging as part of their initial imaging assessment for stroke. Noninvasive intracranial vascular imaging should then be obtained as quickly as possible (Class I; Level of Evidence A). (New recommendation)[2]
Ischemic Stroke[edit | edit source]
In the case of an Ischemic Stroke, the more rapidly blood flow is restored, the more likely for improved recovery and fewer brain cell deaths. Primary treatment is aimed at breaking down the clot through means of medication (Thrombolysis) or mechanical removal of clot (Thrombectomy). Other treatments aim to minimise the clot becoming larger or prevention of new clot formation by means of anticoagulant type medications such as Aspirin, Clopidogrel or Dipyridamole. In conjunction with this, overall patient medical condition should be managed including monitoring of blood sugars, oxygen levels and hydration and providing adequate treatment for same[5].
- All patients with disabling acute ischemic stroke who can be treated within 3 - 4.5 hours after symptom onset should be evaluated without delay to determine their eligibility for treatment with intravenous tissue plasminogen activator [2].
Thrombolysis[edit | edit source]
Tissue Plasminogen Activator (tPA) is a medication that dissolves blood clots. It is called a thrombolytic agent or more commonly referred to as the “clot buster.” It is an intravenous or IV medication usually given through a catheter inserted into a vein in the arm. Rapid administration of intravenous tPA to appropriate patients remains the mainstay of early treatment of acute ischemic stroke. For patients who meet national and international eligibility guidelines, intravenous tPA administration improves functional outcomes at 3 to 6 months when given within 4.5 hours of Ischemic Stroke Onset and should be administered. Every effort should be made to shorten any delays in initiation of treatment as earlier treatments are associated with increased benefits [2].
The goal of therapy for ischemic stroke is to safely maximize patient functional recovery to pre-stroke baseline by rapidly reperfusing the ischemic penumbra. tPA has optimal use within 3 hours from onset of symptoms but in carefully selected patients there is a longer time window for treatment up to 4.5 hours. Studies continue to support that time from onset to treatment with tPA is directly related to outcomes in Ischemic Stroke. The sooner tPA is given, the greater the benefit (Stroke, 2013).
Beyond the approved and recommended time windows for tPA use, risk of complications rises. tPA is no different from other medications in that it has potential adverse effects, which makes it essential that a careful consideration is given to exclusion criteria, risks and benefits for use in each individual patient. The major complications is symptomatic intracranial hemorrhage which occurs at a rate of roughly 5.2%. Other complications include orolingual angioedema (allergic reaction), acute hypotension, and systemic bleeding [2] [6].
Level I Clinical Guideline Recommendations
- Patients eligible for intravenous r-tPA should receive intravenous r-tPA even if endovascular treatments are being considered
(Class I; Level of Evidence A). (Unchanged from the 2013 Guideline)[2]
You can read more about tPA here: Tissue Plasminogen Activator (tPA) - What You Should Know
Endovascular Therapy[edit | edit source]
Physical removal of a blood clot can be completed by Endovascular Therapy either through a Catheter Based Drug or Mechanical Thrombectomy to dissolve or remove the clot and is another strongly recommended treatment option in Ischemic Stroke. Recent research has shown positive results for carefully selected patients with severe stroke due to proximal arterial occlusion and in whom recanalisation can be achieved within six hours of symptom onset, and compared to patients treated with standard medical treatment alone those who also had mechanical thrombectomy were more likely to be functionally independent at three to six months[7] [8].
The availability of endovascular treatment raises new opportunities for patients with severe stroke but also poses new challenges for stroke services. Observational and clinical trial data suggest that 7% to 13% of patients with acute stroke are potentially eligible for endovascular treatment but currently availability and access to endovascualr therapy poses new challeges ie not all hospitals have the resources and expertise necessary to provide this treatment option. Stroke Services must develop systems in order to work with Hospitals who have Interventional Radiology available and be able to triage and transport eligible patients within the required timeframes[7].
There are a range of Clot Retrieval Devices currently used for Endovascular Treatments and include [9] [2];
- The Mechanical Embolus Removal in Cerebral Ischemia (MERCI) Thrombectomy System which is a looped wire with progressively smaller loops, that is sent through to the clot via a small catheter. As the catheter is withdrawn, the coiled wire is deployed, which captures the clot for removal.
- The Penumbra Thrombectomy System uses aspiration for clot removal from large intracranial vessels. With a suction catheter at the proximal end of the clot, the device continuously debulks and aspirates clot debris until the vessel is clear. The device can remove a clot in a matter of minutes.
- The Solitaire Flow Restoration Device and the Trevo Retriever Device have the ability to bypass the clot and restore flow and retrieve the clot.
Studies have shown that when clot retrieval is performed within six hours of symptom onset, 60-68% of patients achieved post-procedure revascularization. Patients eligible for clot retrieval are those with recent ischemic stroke, who can receive treatment within at least preferably 3-6 hours of the onset of stroke signs and symptoms. Those patients with uncontrolled HTN, internal bleeding, seizures, blood clotting problems, or those on anticoagulants are not candidates for this procedure. Complications from clot retrieval procedures include hemorrhage, internal bleeding, death and recurrence of stroke[2] [7] [8].
Level I Clinical Guideline Recommendations
- Patients should receive endovascular therapy with a stent retriever if they meet all the following criteria
(Class I; Level of Evidence A).(New Recommendation):
(a) Prestroke mRS score 0 to 1,
(b) Acute ischemic stroke receiving intravenous r-tPA within 4.5 hours of onset according to guidelines from professional medical societies,
(c) Causative Occlusion of the Internal Carotid Artery or Proximal MCA (M1),
(d) Age ≥18 Years,
(e) NIHSS Score of ≥6,
(f) ASPECTS of ≥6, and
(g) Treatment can be initiated (groin puncture) within 6 hours of symptom onset[2] - Reduced time from symptom onset to reperfusion with endovascular therapies is highly associated with better clinical outcomes. To ensure benefit, reperfusion to TICI Grade 2b/3 should be achieved as early as possible and within 6 hours of Stroke Onset
(Class I; Level of Evidence B-R). (Revised from the 2013 Guideline)[2]
Haemorrhagic Stroke[edit | edit source]
In the case of Haemorrhagic Stroke the main aim is to stop the bleeding as early as possible, often requiring Neurosurgical Intervention. The treatment of a hemorrhagic stroke depends upon the cause of the bleeding (eg, high blood pressure, use of anticoagulant medications, head trauma, blood vessel malformation). Most patients are monitored closely in an intensive care unit during and after a hemorrhagic stroke. The initial care of a person with hemorrhagic stroke includes several components [10] [11] [12]:
- Determining the cause of the bleeding
- Controlling the blood pressure
- Stopping any medication that could increase bleeding (eg, warfarin, aspirin). If the patient has been taking warfarin, specific treatments such as factor VIIa or transfusions of blood clotting factors, may be given to stop ongoing bleeding
- Measuring and controlling the pressure within the brain
Basic life support, control of bleeding, seizures, blood pressure (BP), and intracranial pressure, are critical. Medications used in the treatment of acute stroke include the following[11] [10]:
- Anticonvulsants - Prevent Seizure recurrence
- Antihypertensive Agents - Reduce BP and other risk factors of Heart Disease
- Osmotic Diuretics - Decrease Intracranial Pressure in the Subarachnoid Space
Neurosurgery[edit | edit source]
Once a hemorrhagic stroke is brought under control with emergency care, further treatment measures can be taken. If the rupture is small and produces only a small amount of bleeding and pressure, supportive care may be the only other form of care you need. But in some case surgical procedure may be recommended to repair the ruptured vessel, stop bleeding, or prevent further bleeding, for more serious strokes. Surgery may also be required to relieve the pressure caused by the bleeding and brain swelling [11] [10].
Aneurysm Treatment[edit | edit source]
An aneurysm is a blood vessel that has a weak area that balloons out. If the area ruptures and bleeds, a hemorrhagic stroke can occur. A clamp can be placed at the base of the aneurysm to prevent bleeding before a stroke or to prevent re-bleeding. This surgery requires removing a piece of the skull and locating the aneurysm within the brain tissue.
Coil embolization is an interventional procedure that is less invasive than clipping. It involves inserting a catheter into an artery in the groin. The catheter is guided along blood vessels in the body into the vessel in the brain where the aneurysm is located. A tiny coil is advanced into the aneurysm, filling the area with the coil. A blood clot forms within the coil, blocking blood flow into the aneurysm and preventing it from rupturing again. Other materials may also be injected to obliterate an aneurysm or AVM[11] [10] [12].
Arteriovenous Malformation Treatment[edit | edit source]
If the stroke is caused by an Arteriovenous Malformations (AVMs) surgery may be used to remove it. This is not always possible, however, and depends on the location of the AVM. Some AVMs have a significant risk of further bleeding. The decision to treat an AVM depends on several factors; includin patients age, location and size of AVM, and abnormalities of the veins that drain the AVM and whether or not the AVM has previously bled. Treatment could include surgery, radiosurgery (use of radiation to shrink blood vessels), or embolization techniques[11] [12].
Decompressive Craniotomy[edit | edit source]
When a patient’s life appears to be threatened because of the pressure effects of a blood clot in the brain, the physician may consider a procedure to open the skull and/or remove the blood. Considerations include the location and size of the hemorrhage, the patient’s age and medical condition, and the likelihood of making a recovery from the stroke[11] [10] [12].
Emergency Stroke Evaluation Simulation[edit | edit source]
- This is a video simulation of what its like to be a stroke patient at Massachusetts General Hospital (MGH).
- The full video is approximately 15 minutes.
- These videos are created the Massachusetts General Hospital (MGH) Stroke Service in an effort to increase stroke education among patients and caregivers.
|
Goal Times |
Arrival & Triage |
Entry & Registration |
Clinical Assessment |
|
CT Scan |
tPA Consent & Order |
tPA Delivery |
References[edit | edit source]
- ↑ Adams H, Adams R, Del Zoppo G, Goldstein LB. Guidelines for the early management of patients with ischemic stroke: 2005 guidelines update a scientific statement from the Stroke Council of the American Heart Association/American Stroke Association. Stroke. Apr 2005;36(4):916-23.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Powers WJ, Derdeyn CP, Biller J, Coffey CS, Hoh BL, Jauch EC, Johnston KC, Johnston SC, Khalessi AA, Kidwell CS, Meschia JF. 2015 AHA/ASA focused update of the 2013 guidelines for the early management of patients with acute ischemic stroke regarding endovascular treatment. Stroke. 2015 Jan 1:STR-0000000000000074.
- ↑ 3.0 3.1 AHA. Severity Based Stroke Triage Algorithm for EMS. http://www.heart.org/HEARTORG/Professional/MissionLifelineHomePage/Mission-Lifeline-Stroke_UCM_491623_SubHomePage.jsp. [Accessed 01 May 2017]
- ↑ Prasad K, Kaul S, Padma MV, Gorthi SP, Khurana D, Bakshi A. Stroke management. Annals of Indian Academy of Neurology. 2011 Jul 1;14(5):82.
- ↑ Verheyen G and Ashbury A. IN Physical Management for Neurological Conditions 3rd Edition. Southampton: Elsevier 2011 pp.12-13
- ↑ Jauch EC, Cucchiara B, Adeoye O, Meurer W, Brice J, Gentile N, Hazinski MF. Part 11: Adult Stroke. Circulation. 2010 Nov 2;122(18 suppl 3):S818-28.
- ↑ 7.0 7.1 7.2 Merino José G. Endovascular treatment for stroke BMJ 2016; 353 :i2098
- ↑ 8.0 8.1 Rodrigues FB, Neves JB, Caldeira D, Ferro JM, Ferreira JJ, Costa J. Endovascular treatment versus medical care alone for ischaemic stroke: systematic review and meta-analysis. BMJ 2016;353:i1754.
- ↑ Rod
- ↑ 10.0 10.1 10.2 10.3 10.4 Medscape. Hemorrhagic Stroke Treatment & Management. http://emedicine.medscape.com/article/1916662-treatment. [Accessed 02 May 2017]
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 BMJ Best Practice. Haemorrhagic Stroke. http://bestpractice.bmj.com/best-practice/monograph/1079/treatment/guidelines.html (accessed 2 May 2017).
- ↑ 12.0 12.1 12.2 12.3 Up to Date. Hemorrhagic stroke treatment (Beyond the Basics. [Accessed 02 May 2017]