Pain Facilitation and Inhibition
Introduction[edit | edit source]
The pain experience among people is highly variable, even under circumstances where the soft tissue injuries are similar. The individual difference in pain sensitivity is related to sensitivity of peripheral pain receptors and variability in CNS pain processing. Reasons for this include:
- Peripheral impulses coming from tissue receptors undergo modification in dorsal horn neurones that can either result in inhibition or facilitation of pain.
- Emotional state, level of anxiety, attention and distraction, memories, stress, fatigue and many other factors can either increase or reduce the pain experience.
Increasing evidence suggests that ‘bottom–up’ and ‘top–down’ modulatory circuits within the spinal cord and brain play an important role in pain processing, which can profoundly affect the experience of pain.[1] The influence of pain facilitation and inhibitory pathways explains individual variability in pain experience. There is a dysregulated pattern of endogenous pain regulation in aged people. Aside from structural affectation in chronic pain such as osteoarthritis, low back pain, and others that are common in older adults, an age-related affectation of pain inhibition and facilitation makes older adults prone to a higher risk of having chronic pain than younger adults.[2]
Ascending pain pathway[edit | edit source]
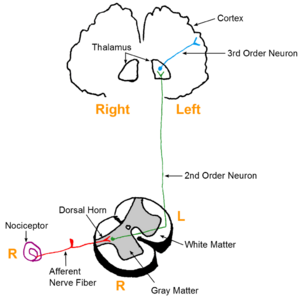
Ascending pathway is how we feel pain.
- The process begins when pain sensory neurons, called nociceptors, detect a potentially damaging stimulus. These neurons send this signal as an action potential to other neurons using the excitatory neurotransmitters glutamate, which depolarizes the cell, and substance p, which promotes inflammation and pain.
- Once the signal reaches the base of dorsal horn, a column in the spinal cord that relays sensory information, it is then sent up though a network of neurons called the spinothalamic tract that delivers sensory signals from the spinal cord to the somatosensory cortex which perceives pain[3].
Descending pain pathway[edit | edit source]
Once the signal from the ascending pathway reaches the somatosensory cortex, it triggers the descending pain modulation pathway. The goal of this pathway is to allow the organism to function enough to respond to the pain source by reducing the pain signal through neuronal inhibition. Descending pain control pathways can be both facilitatory as well as inhibitory.
- Facilitatory pathways are the one which enhances pain perception
- Inhibitory pathways suppresses pain perception.
The balance between inhibition and facilitation is dynamic, and can be altered in different behavioral, emotional, psychological and pathological states.[4] Descending pain control pathways plays a critical role in determining the experience of both acute and chronic pain.[5]
The goal of this pathway is to allow the organism to function enough to respond to the pain source by reducing the pain signal through neuronal inhibition.
- It begins in the periaqueductal gray (PAG), a region of the midbrain that process nociceptive information and relays it to the rostral ventral medulla (RVM).
- These neurons in the RVM then send a signal down the spinal cord to release endogenous opioids at neuronal synapses at multiple points in the peripheral nervous system to prevent these pain signaling neurons from sending action potentials.
- Additionally, these endogenous opioids are released in parts of the dorsal horn of the spinal cord to further block ascending pain transmission signals.
Pain is helpful since it alerts the brain to tissue damage and provides useful information about its type and location. However, when this signal starts to impede our own ability to survive and function it must be reduced, hence the need for the descending pathway and endogenous opioids[3].
The Role of Neurotransmitters[edit | edit source]
1.Opioid pathway: The endogenous opioid system is one of the most studied innate pain-relieving systems. This system consists of widely scattered neurons that produce three opioids: β-endorphin, the met- and leu-enkephalins, and the dynorphins. These opioids act as neurotransmitters and neuromodulators at three major classes of receptors, termed μ, δ, and κ, and produce analgesia[7].
2.Serotonergic pathway: Serotonin (5-HT) and norepinephrine, are involved in endogenous pain modulation, both can be released via descending pain pathways to modulate nociceptive signalling in the spinal cord.
- Norepinephrine inhibits pain through α2 adrenoceptors
- Serotonin seems to have pain facilitatory and inhibitory functions.
3.Noradrenergic pathways: Norepinephrine is involved in intrinsic control of pain. Main sources of norepinephrine are sympathetic nerves peripherally and noradrenergic brainstem nuclei A1-A7 centrally. These nuclei have noradrenergic projections to the spinal cord, which can inhibit the response of dorsal horn pain transmission neurons. Dorsal horn neuron recordings have shown that activated a2-adrenergic receptors hyperpolarize presynaptic neurons and decrease the release of excitatory neurotransmitters from primary afferent terminals, resulting in pain inhibition[4].
Non-pharmacological agents affecting pain modulation[edit | edit source]
Non- pharmacological agents are the other agents than that of medication which affect the pain modulation systems. They iclude:
- Psychological interventions (including distraction, stress management, hypnosis, and other cognitive-behavioral interventions),
- Acupuncture and acupressure,
- Transcutaneous electrical nerve stimulation,
- Physical therapies (including massage, heat/cold, physiotherapy).[8]
Accumulating evidence supports the important role of supraspinal pain modulation for both analgesia and hyperalgesia. Multiple cortical and subcortical brain and brainstem regions integrate and process sensory, autonomic and emotional information, resulting in activation of the PAG and RVM, with subsequent inhibition or facilitation of pain-related dorsal horn neurons. This top–down modulation is relevant for experimental, as well as clinical pain. These pain modulatory pathways are affected by memories and mood, as well as sociocultural background as different cortical regions like amygdala, hypothalamus are involved in the descending pain modulation pathways.[9]
Nonpharmacological analgesia therefore involves the inhibition of nociceptive input by activating separate antinociceptive outputs. Electroacupuncture has been shown to enhance the expression of serotonin and reduce the release of substance P during electroacupuncture inhibition of acute nociceptive responses.TENS appears to produce both segmental and descending pain inhibition since inhibition remains after spinalization (removal of descending inhibition) and concentrations of endogenous opioids have been shown to increase in cerebrospinal fluid following TENS procedure.[8]
References[edit | edit source]
- ↑ Staud R. The important role of CNS facilitation and inhibition for chronic pain. International journal of clinical rheumatology. 2013 Dec 1;8(6):639. Available:https://www.openaccessjournals.com/articles/the-important-role-of-cns-facilitation-and-inhibition-for-chronic-pain.pdf (accessed 17.4.2022)
- ↑ Naugle KM, Ohlman T, Naugle KE, Riley ZA, Keith NR. Physical activity behavior predicts endogenous pain modulation in older adults. Pain. 2017 Mar 1;158(3):383-90.
- ↑ 3.0 3.1 Tufts Pain Transmission Pathway Available:https://sites.tufts.edu/opioidpeptides/pathways-and-receptors/classical-opioid-signaling/ (accessed 17.4.2022)
- ↑ 4.0 4.1 Staud R. The important role of CNS facilitation and inhibition for chronic pain. International journal of clinical rheumatology. 2013 Dec 1;8(6):639.
- ↑ Heinricher MM, Tavares I, Leith JL, Lumb BM. Descending control of nociception: specificity, recruitment and plasticity. Brain research reviews. 2009 Apr 1;60(1):214-25.
- ↑ Armando Hasudungan. PAIN! Physiology - The Ascending Pathway, Descending Pain Pathway and the Substantia Gelatinosa. Available From: https://www.youtube.com/watch?v=5c8maFAhqIc [last accessed 22/2/2020]
- ↑ Holden JE, Jeong Y, Forrest JM. The endogenous opioid system and clinical pain management. AACN Advanced Critical Care. 2005 Jul;16(3):291-301.Available: https://aacnjournals.org/aacnacconline/article-abstract/16/3/291/14207/The-Endogenous-Opioid-System-and-Clinical-Pain?redirectedFrom=fulltext(accessed 17.4.2022)
- ↑ 8.0 8.1 Pak SC, Micalos PS, Maria SJ, Lord B. Nonpharmacological interventions for pain management in paramedicine and the emergency setting: a review of the literature. Evidence-Based Complementary and Alternative Medicine. 2015;2015.
- ↑ Zhuo M. Descending facilitation: From basic science to the treatment of chronic pain. Molecular pain. 2017 Mar;13:1744806917699212.