Anterior Cruciate Ligament (ACL) Injury
Original Editors - Stephanie Geeurickx, Kevin Campion, Aarti Sareen as part of the Vrije Universiteit Brussel Evidence-Based Practice Project
Top Contributors - Puja Gaikwad, Aarti Sareen, Laura Ritchie, Els Van Haver, Admin, Kim Jackson, Evan Thomas, Kevin Campion, Naomi O'Reilly, Stephanie Geeurickx, Khloud Shreif, Chelsea Mclene, Rachael Lowe, Bisoffi, Mariam Hashem, Scott Cornish, Tony Lowe, Dorien Scheirs, Simisola Ajeyalemi, Laurien Henau, Bruno Serra, Sehriban Ozmen, 127.0.0.1, WikiSysop, Fasuba Ayobami, Amber Van de Maele, Claire Knott, Wanda van Niekerk, Scott Buxton, Robin Tacchetti, Rujuta Naik and Amanda Hicks
Introduction[edit | edit source]
Injuries to the ACL are relatively common knee injuries among athletes.[1] They occur most frequently in those who play sports involving pivoting (e.g. football, basketball, netball, soccer, European team handball, gymnastics, downhill skiing). They can range from mild (such as small tears/sprain) to severe (when the ligament is completely torn). Both contact and non-contact injuries can occur, although non-contact tears and ruptures are most common when the limb is in non contact and combined with valgus and internal rotation trauma It appears that females tend to have a higher incidence rate of ACL injury than males, that being between 2.4 and 9.7 times higher in female athletes competing in similar activities[2][3][4][5], and an acute rupture of ACL is a common trauma, it is incidence up to 84/ 100000 persons in USA, 78/100000 persons for sweden with 32 years is the mean age of injury[6].
Clinically Relevant Anatomy[edit | edit source]
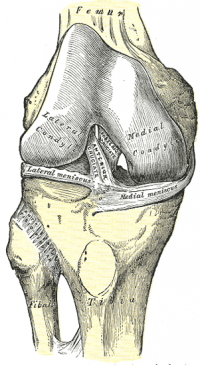
The ACL is a band of dense connective tissue which courses from the femur to the tibia. It is considered as a key structure in the knee joint, as it resists anterior tibial translation and rotational loads.[7]
The ACL arises from the posteromedial corner of the medial aspect of the lateral femoral condyle in the intercondylar notch[8] and inserted anterior to the intercondyloid eminence of the tibia, blending with the anterior horn of the medial meniscus. The ACL courses anteriorly, medially, and distally across the joint as it passes from the femur to the tibia. As it does, it turns on itself in a slight outward (lateral) spiral.
There are two components of the ACL, the smaller anteromedial bundle (AMB) and the larger posterolateral bundle (PLB), named according to where the bundles insert into the tibial plateau. When the knee is extended the PLB is tight and the AMB is moderately lax. However, as the knee is flexed, the femoral attachment of the ACL assumes a more horizontal orientation, causing the AMB to tighten and the PLB to loosen and thus leave the AMB as the restraint to anterior tibial load[6]. That means PLB has a stabilizing effect when the knee is near to extension < 30° on rotational and antero-posterior forces and AMB act as a stabilizer and becomes more tense with higher degrees of knee flexion[6].
Refer to this page for more information on the ACL Biomechanics:
Anterior Cruciate Ligament (ACL) - Structure and Biomechanical Properties
Functions of ACL[edit | edit source]
- Primary restraints to anterior tibial displacement[9]: counting for 85% of the resistance to anterior drawer test, when the knee is kept at 90 degree of flexion.
- Secondary restraints to tibial rotation & varus : valgus angulation at full knee extension.
- Proprioceptive function: presence of mechanoreceptors in the ligaments.[10]
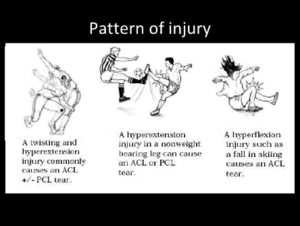
Mechanisms of Injury[edit | edit source]
Three major types of ACL injuries are described:
- Direct Contact: 30% of the cases[11].
- Indirect Contact.
- Non-Contact: 70% of the cases: by doing a wrong movement.
Anterior cruciate ligament (ACL) injuries are common in young individuals who participate in sports activities associated with pivoting, decelerating and jumping.
Most common are the non-contact injuries are more likely to occur with lower BMI[11], it caused by forces generated within the athlete’s body. While, most other sport injuries involve a transfer of energy from an external source. Approximately 75% of ruptures are sustained with minimal or no contact at the time of injury.[12] A cut-and-plant movement is the typical mechanism that causes the ACL to tear, being a sudden change in direction or speed with the foot firmly planted. Rapid deceleration moments, including those that also involve planting the affected leg to cut and change direction, have also been linked to ACL injuries, as well as landing from a jump, pivoting, twisting, and direct impact to the front of the tibia[12]. The timing during the game and during the seasone is related to the injury mechanism of ACL but the relation still unclear[11].
Women are three times[13] more prone to have the ACL injured than men and is thought to be due to the following reasons:
- Smaller size and different shape of the intercondylar notch: A narrow intercondylar notch and a plateau environment are risk factors of predisposing female non-athletes with knee OA to ACL injury aged 41-65 years. [14]
- Wider pelvis and greater Q angle: A wider pelvis requires the femur to have a greater angle towards the knee, lesser muscle strength provides less knee support, and hormonal variations may alter the laxity of ligaments.[15][16]
- Greater ligament laxity: Young athletes with non-modifiable risk factors like ligament laxity are at a particularly increased risk of recurrent injury following ACL reconstruction (ACLR). [17]
- Shoe surface interface: The pooled data from the three studies suggest that the chances of injury are approximately 2.5 times higher when higher levels of rotational traction are present at the shoe-surface interface. [18]
- Neuromuscular factors.
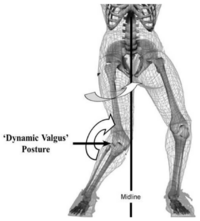
- The mechanism of ACL injury may differ in females especially with respect to the dynamic positioning of the knee, as females demonstrate greater valgus collapse of the LE primarily in the coronal plane.[19]
Risk Factors[edit | edit source]
Risk factors for ACL injuries include environmental factors (e.g. high level of friction between shoes and the playing surface) and anatomical factors (e.g. narrow femoral intercondylar notch). The injury is characterized by joint instability, which is associated with both acute dysfunction and long-term degenerative changes such as osteoarthritis and meniscal damage.[20] Knee instability leads to decreased activity, which can lead to poor knee-related quality of life. The risk factors for ACL injury have been considered as either internal or external to an individual. External risk factors include type of competition, footwear and surface, and environmental conditions. Internal risk factors include anatomical, hormonal and neuromuscular risk factors. [21][22]
External Risk Factors[edit | edit source]
Competition in games versus practice [edit | edit source]
Very little is known about the effect of type of competition on the risk of an athlete suffering ACL injury. Myklebust et al[21] reported that athletes are at a higher risk of suffering an ACL injury during a game than during practice. This finding introduces the hypothesis that the level of competition, the way in which an athlete competes, or some combination of the two, increases an athlete’s risk of suffering an ACL injury.
Footwear and playing surface [edit | edit source]
Although increasing the coefficient of friction between the sports shoe and playing surface may improve traction and sports performance, it also has the potential to increase the risk of injury to the ACL. Lambson et al[22] found that the risk of suffering an ACL injury is greater in football athletes who have boots with a higher number of cleats and an associated higher torsional resistance at the foot-turf interface. Olsen et al[21] reported that the risk of suffering an ACL injury is greater in female team handball athletes who compete on artificial floors that have a higher torsional resistance at the foot-floor interface than in those who compete on wood floors. This relationship did not exist for male athletes.
Protective equipment[edit | edit source]
There is some controversy about the use of functional bracing to protect the ACL-deficient knee. Kocher et al [23] studied professional skiers with ACL-deficient knees and found a greater risk of knee injury in those who did not wear a functional brace than in those wore a brace. McDevitt et al [24] performed a randomized controlled study of the use of functional braces in cadets attending the US military academies who underwent ACL reconstruction. At the 1-year follow-up, the use of functional bracing did not affect the rate of ACL graft re-injury. There were only three injuries among those in the unbraced group and two injuries in the braced group however.
Meteorological conditions [edit | edit source]
For sports that are played on natural or artificial turf, the mechanical interface between the foot and playing surface is highly dependent on the meteorological conditions. However, very little is known about the effect of these variables on an athlete’s risk of suffering an ACL injury. Orchard et al [25] reported that non-contact ACL injuries sustained during Australian football were more common during periods of low rainfall and high evaporation. This work introduces the hypothesis that meteorological conditions have a direct effect on the mechanical interface (or traction) between the shoe and playing surface, and this, in turn, has a direct effect on the likelihood of an athlete suffering an ACL injury.
Internal Risk Factors[edit | edit source]
Anatomical risk factors [edit | edit source]
Abnormal posture and lower extremity alignment (eg, hip, knee and ankle) may predispose an individual to ACL injury by contributing to increased ACL strain values. Alignment of the entire lower extremity should therefore be considered when assessing risk factors for ACL injury. Unfortunately, very few studies have studied alignment of the entire lower extremity and determined how it is related to the risk of ACL injury. Most of what is known has come from investigations of specific anatomical measures.
Bony morphology[edit | edit source]
The steeper the tibial plateau considered a risk factor for ACL injury, ther are recent studies found that tibial plateau slope≥ 12° was associated with higher risk to develop contralateral ACL injury after ACL reconstruction and risk for lateral meniscus tear.
Depth of the distal femoral condyle isa nother risk factor for ACL injury, it may be associtaed with rotatory knee laxity and chhnage in the pressure points between tibia and femur[26].
Biomechanics of Injury[edit | edit source]
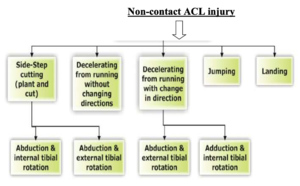
As 60-80% of ACL injuries occur in non-contact situations, it seems likely that appropriate prevention efforts are warranted. Cutting or sidestep manoeuvres are associated with dramatic increases in the varus-valgus and internal rotation moments. The risk of ACL injury increase if there are more than one of these movements happened; knee joint twisted, bent backward, or side to side stress.
When there is an anterior tibiofemoral shear force ACL is loaded and the force is first controlled by ACL, the knee valgus moment with the anterior drawer force proximal to tibia increases ACL loading significantly accprding to Varus- Valgus laxity.
Varus- valgus stability and anterior tibial translation
When there is a varus torque the interaction of the contact points between medial femoral condyle and the medial tibial plateau generates anterior tibial load because of the posterior tibial slope resulting an anterior tibial translation in antero-proximal direction. In addition under varus torque in the ACL-deficient knee there are studies demonstrated an increase in the anterior tibial translation as well as the varus angle[27].
The anterior tibial translation (ATT) in intact, healthy knee increases up to 3mm in flexion and increase to be up to 5.5 mm under the anterior tibial load. When there is an ACL rupture or tear the ATT increase up to 10-15 mm when the knee is 30 flexion and under anterior tibial load, and the tibial will located more anterior even under non-weight bearing.
Biomechanics of trunk motion, hip, and ankle musculature
A biomechanical video analysis of ACL injury in football observed that:
- There is an average trunk tilt ipsilaterally (about 5° at initial contact and injury fram) for all ACL injuries specially for pressing type injuries, and more ipsilateral trunk leaning increase the risk of ACL injury, this the lateral leaning of the trunk causes a lateral shift in centre mass, causing an abduction moment at the knee joint and hence increase ACL loading. Limited trunk rotation to the new direction with hip adduction are associated with an increase in the valgus moment at the knee joint. In addition the synergistic increase in trunk flexion and hip internal rotation moment was associated with higher internal tibial torque[28].
- Hip abduction was common in ACL injuries in football and associated with increase in hip internal rotation this increase is because of high knee abduction moment/ valgus knee with laterally orientated and planted foot position outside the base of support[28].
- For musculatures around the ankle joint considered as an agonist and antagonist for ACL that may affect ACL during landing and be a risk factor for injury. Quadriceps and hamstrings are conventionally viewed as the primary antagonist- agonist pair involved in ACL injury[28], and it may be hypothesized that vigorous eccentric quadriceps muscle action may play a role in disruption of the ACL. Although this normally would be insufficient to tear the ACL, it may be that the addition of valgus knee position and/or rotation could trigger an ACL rupture[29]. For better understanding of ACL injuries in football and illustrating figures, here.
The typical ACL injury occurs with the knee externally rotated and in 10-30° of flexion (shallow knee flexion) when the knee is placed in a valgus position as the athlete takes off from the planted foot and internally rotates with the aim of suddenly changing direction[30]. The ground reaction force falls medial to the knee joint during a cutting maneuver and this added force may tax an already tensioned ACL and lead to failure. Similarly, in landing injuries, the knee is close to full extension[31]. High-speed activities such as cutting or landing manoeuvres require eccentric muscle action of the quadriceps to resist further flexion.
A hypothesis of how non-contact ACL injuries occurs in female atheletes handball and basketball is; when valgus loading is applied, the medial collateral ligament becomes taut and lateral compression occurs. This compressive load, as well as the anterior force vector caused by quadriceps contraction, causes a displacement of the femur relative to the tibia where the lateral femoral condyle shifts posteriorly and the tibia translates anteriorly and rotates internally, resulting in ACL rupture. After the ACL is torn, the primary restraint to anterior translation of the tibia is gone. This causes the medial femoral condyle to also be displaced posteriorly, resulting in external rotation of the tibia. Valgus loading is a key factor in the ACL injury mechanism and at the same time, the knee rotates internally. A quadriceps drawer mechanism may also contribute to ACL injury as well as external rotation. [33]
Potential neuromuscular imbalances may be related to components of the injury mechanism. Women have more quadriceps dominant neuromuscular patterns than men. Hamstring recruitment has been shown to be significantly higher in men than in women. The hamstring to quadriceps peak torque ratio tends to be greater in men than in women. Due of the likely injury mechanism, it is recommended that athletes avoid knee valgus and land with more knee flexion. [34]
Grades of Injury[edit | edit source]
An ACL injury is classified as a grade I, II, or III sprain.[35]
Grade I Sprain[edit | edit source]
- The fibers of the ligament are stretched, but there is no tear.
- There is a little tenderness and swelling.
- The knee does not feel unstable or give out during activity.
- No increased laxity and there is a firm end feel.
Grade II Sprain[edit | edit source]
- The fibers of the ligament are partially torn or incomplete tear with hemorrhage.
- There is a little tenderness and moderate swelling with some loss of function.
- The joint may feel unstable or give out during activity.
- Increased anterior translation yet there is still a firm end point.
- Painful and pain increase with Lachman's and anterior drawer stress tests.
Grade III Sprain[edit | edit source]
- The fibers of the ligament are completely torn (ruptured); the ligament itself is torn completely into two parts.
- There is tenderness, but limited pain, especially when compared to the seriousness of the injury.
- There may be a little swelling or a lot of swelling.
- The ligament cannot control knee movements. The knee feels unstable or gives out at certain times.
- There is also rotational instability as indicated by a positive pivot shift test.
- No end point is evident.
- Haemarthrosis occurs within 1-2 hours.
An ACL avulsion occurs when the ACL is torn away from either the femur or the tibia. This type of injury is more common in children than adults. The term anterior cruciate deficient knee refers to a grade III sprain in which there is a complete tear of the ACL. It is generally accepted that a torn ACL will not heal.[36]
Clinical Presentation[edit | edit source]
- Occurs after either a cutting manoeuvre or single leg standing, landing or jumping.
- There may be an audible pop or crack at the time of injury.
- A feeling of initial instability which may be masked later by extensive swelling.
- Episodes of giving way especially on pivoting or twisting motions. Patient has a trick knee and predictable instability.
- A torn ACL is extremely painful, particularly immediately after sustaining the injury.
- Swelling of the knee, usually immediate and extensive, but can be minimal or delayed.
- Restricted movement, especially an inability to fully extend the knee.
- Possible widespread mild tenderness.
- Tenderness at the medial side of the joint which may indicate cartilage injury.
Associated Injuries[edit | edit source]
Injuries to the ACL rarely occur in isolation. The presence and extent of other injuries may affect the way in which the ACL injury is managed.
Meniscal Lesions[edit | edit source]
Over 50% of all ACL Ruptures have associated Meniscal injuries. If seen in combination with a medial meniscus tear and an MCL Injury, it is termed O’Donohue’s Triad which has 3 components:[1]
- Anterior Cruciate Ligament (ACL) Tear
- Medial Collateral Ligament (MCL) Tear
- Meniscal Tear
Lateral meniscus lesion are presented but with lower rate than medial meniscus (17%-51%)[6].
Medial Collateral ligament injury[edit | edit source]
Associated injury to the MCL (Grade I-III) poses a particular problem due to tendency to develop stiffness after this injury. Most orthopaedic surgeons will first treat an MCL injury in a limited motion knee brace for a period of six weeks, during which time the athlete would undertake a comprehensive rehabilitation program. Only then would ACL reconstruction be performed or be treated.[37]It was estimated to be found in every fifth case with the ACL was rupture.
Bone Contusions and Microfractures[edit | edit source]
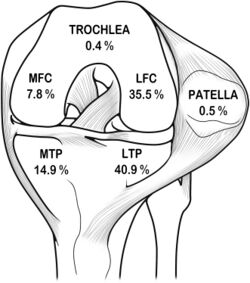
Subcortical trabecular bone injury (bone bruise) may occur due to the pressures exerted on the knee in traumatic injury and are especially associated with ACL rupture. Associated injuries of the menisci and the MCL tend to increase the progression of bone contusion.[38] The focal signal abnormalities in subchondral bone marrow seen on MRI (undetectable on radiographs) are thought to represent micro trabecular fractures, haemorrhage and edema without disruption of adjacent cortices or articular cartilage.[39] Bone contusions may occur in isolation to ligamentous or meniscal injury.[40]
Occult bony lesions have been reported in 84-98% of the patients with ACL rupture.[38][41][42] The majority of these have lesions of the lateral compartment,[43] involving either the lateral femoral condyle, the lateral tibial plateau, or both. The bony bruising itself is unlikely to cause pain or reduced function.[44] Although the majority of bony lesions resolve, permanent alterations may remain. There is confusion in the literature as to how long these bony lesions remain, but it has been reported that they can persist on MRI for years.[45] Rehabilitation and the long-term prognosis may be affected in those patients with extensive bony and associated articular cartilage injuries. In the case of severe bone bruising it has been recommended to delay return to full weight-bearing status to prevent further collapse of subchondral bone and further aggravation of articular cartilage injury.[45]
Chondral Injury[edit | edit source]
Hollis et al [46] suggested that all patients following traumatic ACL disruption sustained a chondral injury at the time of initial impact with subsequent longitudinal chondral degradation in compartments unaffected by the initial bone contusion, a process that is accelerated at 5 to 7 years’ follow-up.[46]
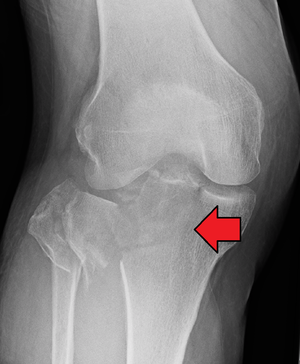
Tibial Plateau Fractures[edit | edit source]
A Tibial Plateau Fracture is a bone fracture or break in the continuity of the bone occurring in the proximal tibia affecting the knee joint, stability, and motion. The tibial plateau is a critical weight-bearing area located on the upper tibia and is composed of two slightly concave condyles (medial and lateral condyles) separated by an intercondylar eminence and the sloping areas in front and behind it.
It can be divided into three regions:
- The Medial Tibial Plateau (the part of the tibial plateau nearest the centre of the body and contains the medial condyle),
- The Lateral Tibial Plateau (the part of the tibial plateau that is farthest away from the centre of the body and contains the lateral condyle).
- The Central Tibial Plateau (located between the medial and lateral plateaus and contains intercondylar eminence).[47]
These fractures are also caused by varus or valgus forces combined with axial loading on knee and mostly occur with ACL injuries, rarely alone. The fracture of lateral tibial plateau is also called a Segond fracture which most commonly occurs with an ACL injury. [47]
Posterolateral Corner Injury[edit | edit source]
The stability of the posterolateral corner of the knee is provided by capsular and non-capsular structures that function as static and dynamic stabilizers[48] including the lateral collateral ligament (LCL), the popliteus muscle and tendon including its fibular insertion (popliteofibular ligament), and the lateral and posterolateral capsule. Injuries to this region, that result in posterolateral rotatory instability, are usually associated with concurrent ligamentous injuries elsewhere in the knee.[49][50][51] High-grade posterolateral corner injuries are usually associated with rupture of one or both cruciate ligaments. Importantly, failure to address instability of the posterolateral corner structures increases the forces at the ACL and PCL graft sites, and may ultimately predispose to failure of the cruciate reconstruction.[52][53][54] (See also: Knee Rotary Instability)
Popliteal Cyst[edit | edit source]
Popliteal cysts, originally called Baker’s cyst, form when a bursa swells with synovial fluid, with or without a clear inciting aetiology. Presentation ranges from asymptomatic to painful, limited knee motion. Sansone et al. found that 44 of 47 popliteal cysts studied were associated with intra-articular lesions. The lesions include medial meniscal (83%) and anterior cruciate ligament tears (32%), synovitis, chondral lesions (43%)[55], and total knee replacement. Intra-articular trauma, arthritis and infection result in knee effusions that lead to popliteal cyst formation. [56]
Most occur within the posteromedial popliteal fossa between the gastrocnemius and deep fascia, as in the present study. In the normal knee, intra-articular volume and pressure are minimized by the osmotic suction exerted by the synovial matrix. The synovial fluid is then drawn back into the veins and lymphatics of the synovium, from where it is pumped out by the articular motion of the knee. The pathological knee, associated with trauma, arthritis or infection, involves an increase in synovial fluid volume and pressure. An effusion occurs when the clearance of synovial fluid lags behind microvascular leakage. [57]
Usually, in an adult patient, an underlying intra-articular disorder is present. In children, the cyst can be isolated and the knee joint normal. A Baker's cyst is less prevalent in a paediatric orthopaedic population than in an adult population. In children, it seems that a Baker's cyst is seldom associated with joint fluid, meniscal tear, or anterior cruciate ligament tear. [58]
Diagnostic Procedures[edit | edit source]
An exact diagnosis can be made by the following procedures:
Physical assessment which includes the following tests:
1. Radiographs[edit | edit source]
Radiographs of the knee should be performed when an ACL tear is suspected, including AP (anterior to posterior) view, lateral view, and patellofemoral projection. The standing AP weight-bearing view provides a way of evaluating the joint space between the femur and tibia. It also allows for measurement of the notch width index which provides important predictive values for ACL tears.[59] The patellar tendon and height are measured on lateral radiograph. A tunnel view may also be helpful. The Merchant's radiograph view not only shows the joint space between the femur and patella but also helps to determine whether the patient has patellofemoral malalignment. The presence of the following factors should be noted from x-ray:
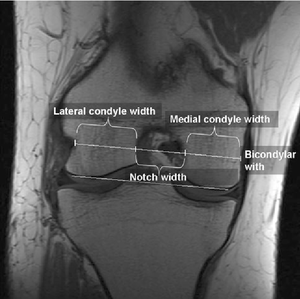
- Notch width index.
- Osteochondral fracture.
- Segond fracture.
- Bone bruise.
The Notch width index is the ratio of the width of the intercondylar notch to the width of the distal femur at the level of the popliteal groove measured on a tunnel view roentgenogram of the knee. The normal intercondylar notch ratio is 0.231 ± 0.044. The intercondylar notch width index for men is larger than that for women. It was found that athletes with non-contact ACL injuries had a notch width index that was at least 1 standard deviation below the average, meaning that a person with an ACL injury is more likely to have a small notch width index compared to normal. It is measured with the help of a ruler placed parallel to joint line. The narrowest portion of the notch at the level of ruler is measured.[60] In more chronic ACL injuries, there may be intercondylar eminence spurring or hypertrophy, or patellar facet osteophyte formation.
This is also one of the reasons why women are more prone to ACL injuries compared to men. It has also been seen that the value of inner angle of the lateral condyle of femur was significantly higher in women athletes with ACL tear compared to those without. Value of width of intercondylar notch was statistically smaller in athletes with ACL tear, compared to those without. Also it was seen that the inner angle of lateral femoral condyle is a better predictive factor for ACL tears in young female handball players compared to intercondylar notch width.
In more chronic ACL injuries, there may be interchondral eminence spurring or hypertrophy, patellar facet osteophyte formation, or joint space narrowing with marginal osteophytes. It is particularly important in skeletally immature patients to have plain radiographic assessment. This is because there is frequently a ligamentous avulsion in this age group.
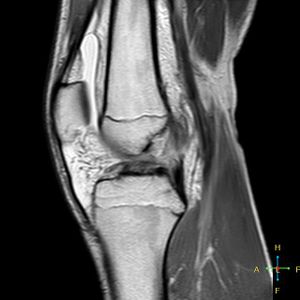
2. MRI[edit | edit source]
MRI has the advantage of providing a clearly defined image of all the anatomic structures of the knee. A normal ACL is seen as a well-defined band of low signal intensity on sagittal image through the intercondylar notch. With an acute injury to the ACL, the continuity of the ligament fibers appears disrupted and the ligament substance is ill defined, with a mixed signal intensity representing local edema and haemorrhage.[61]
MRI can diagnose ACL injuries with an accuracy of 95% or better. MRI will also reveal any associated meniscal tears, chondral injuries, or bone bruises.
A Bone bruise is usually present in conjunction with an ACL injury in more than 80% of cases. The most common site is over the lateral femoral condyle. The bone bruise is most likely caused by impaction between the posterior aspect of the lateral tibial plateau and the lateral femoral condyle during displacement of the joint at the time of the injury. The presence of bone bruise indicates impaction trauma to the articular cartilage.[62] Patients with bone bruises are more prone to develop osteoarthritis later. Bone bruise can be seen most prominently in MRIs.
3. Instrumented laxity testing/arthrometric evaluation of the knee[edit | edit source]
An adjunct to the clinical special tests in assessing anterior translation is the use of instrumented laxity testing. The most commonly cited arthrometer is the KT1000 (Medmetric, San Diego, California). The arthrometer provides an objective measurement of the anterior translation of the tibia that supplements the Lachman test in ACL injury. It can be particularly useful in the examination of acutely injured patients in whom pain and guarding may preclude evaluation. In such patients the Lachman and other tests can be difficult to perform accurately. The arthrometric results can be used as a diagnostic tool to assess ACL integrity or as part of the follow up examination after ACL reconstruction.[63] The results of the KT1000 and its sibling, the KT2000 have been noted to be both reliable and accurate.[64]
4. Dynamic Ultrasonography[edit | edit source]
Ultrasound can aid the examiner in determining the presence of an ACL injury. Direct US visualization of the ACL is challenging, but US is increasingly being used as an extension of the physical examination on the sidelines, in training rooms, and in clinics. Ultrasound can be used to objectively measure the degree of laxity when combined with functional testing (Lachman and anterior drawer tests)[65]
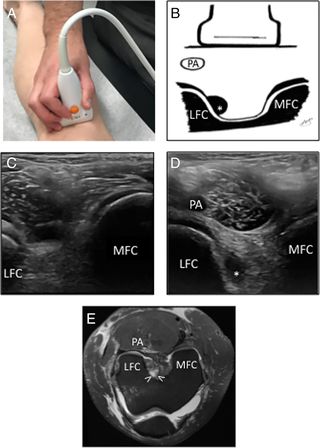
Dynamic US examinations for measuring laxity three static indirect signs of ACL rupture have been described:
- The femoral notch sign : The femoral notch sign is characterized by the presence of a hypoechoic collection adjacent to the lateral femoral condyle, where the ACL should insert.
The other indirect signs are:
- The posterior cruciate ligament (PCL) wave sign.
- capsular protrusion sign.
The validity of the US femoral notch sign shows sensitivity and specificity ranging from 88% to 96.2% and 65% to 100%, respectively. The validity improves when the symptomatic knee is compared to the asymptomatic side. But the validity of the PCL wave sign and capsular protrusion sign have not been studied with high‐resolution US.
Ultrasound does not and cannot replace MRI but can help clinicians decide on further diagnostic tests and treatment in patients with acute knee injuries. These US signs are easy to determine non-invasively, especially in cases in which the clinical examination is difficult or equivocal. Ultrasound may help decrease the number of undetected ACL injuries and can spare patients unnecessary treatment for a presumed diagnosis of a knee contusion, sprain, or strain. In addition, point‐of‐care US is cost‐effective compared to MRI and can potentially give patients a diagnosis the same day while avoiding unnecessary anxiety and worry. It is also worth noting that ultrasound may be a good choice for patients with metallic implants, as MRI artifacts can interfere with accurate assessment of the ACL.[66]
Differential Diagnosis[edit | edit source]
The same characteristics for an ACL injury can be found with;
- Knee dislocations.
- Meniscal injuries.
- Collateral ligaments injury.
- Posterolateral corner injuries to the knee.
Other problems that have to be considered are:
- Patellar dislocation or fracture.
- Femoral, tibial or fibular fracture.
The differential diagnosis of an acute hemarthrosis of the knee due to ACL in addition to a major ligamentous tear would include meniscal tear or patellar dislocation or osteochondral fracture.
Differentiation can mostly be made based on a thorough examination with particular attention for the mechanism at the time of injury. An additional MRI scan can visualize the injury.
Examination[edit | edit source]
The examination of ACL injury can be done in two ways:
- Physical/Clinical examination.
- Examination under anesthesia and arthroscopy.
Physical/Clinical Examination:[edit | edit source]
An organized, systematic physical examination is imperative when examining any joint. Immediately after the acute injury, the physical examination may be very limited due to apprehension and guarding by the patient. While inspecting, the examiner should look for the following:[68]
- Overall alignment of the knee.
- Severe distortion of the normal alignment may represent a fracture of the distal femur or proximal tibia or indicate knee dislocation.
- Any gross effusion, which is most commonly present within a few hours after an ACL injury. Absence of an effusion does not mean that an ACL injury has not occurred. In fact, with more severe injuries that include the surrounding capsule and soft tissues, the hemarthrosis may be able to escape from the knee, and the degree of swelling may paradoxically be diminished. In addition, the presence of swelling and effusion does not guarantee that an ACL injury has occurred. According to Noyes et al, in the absence of bony trauma, an immediate effusion is believed to have a 72% correlation with an ACL injury of some degree.
- Bony abnormality may suggest an associated fracture of the tibial plateau.
- Palpation follows inspection and should begin with the uninvolved extremity. Palpation confirms the presence and degree of effusion and bony injury. Subtle effusions missed during inspection should be picked up by the careful manual examination. Palpation of joint lines and collateral ligaments can rule out a possible associated meniscus tear or sprained ligaments.
- Periarticular tenderness should also be examined.
- Assessing the patient’s range of motion (ROM) should be carried out to look for lack of complete extension, secondary to a possible bucket-handle meniscus tear or associated loose fragment.
- Laxity testing should be done either with the special test or with the help of arthrometer.
Grading and examining the anterior tibial subluxation post ACL injury:[edit | edit source]
| Severity | Amount of Abnormal Tibial Rotation | Positive test | 'Comment |
| Mild (Grade 1) | 1+ (< 5 mm) | Lachman and FRD | May be present with generalised joint laxity.(physiological) |
| Moderate (Grade II) | 2+ (5-10 mm) | Lachman, FRD, Losee, ALRI, Pivot 'slide' but not 'jerk' | No obvious jump with jerk and PS. |
| Severe (Grade III) | 3+ (11-15 mm) | Lachman, FRD, Losee, ALRI, jerk and PS | Obvious jump with jerk and PS and gross subluxation-reduction with test. |
| Gross (Grade IV) | 4+ (> 15mm) | Lachman, FRD, Losee, ALRI, jerk and PS | Impingement of lateral tibial plateau in subluxation position, which requires examiner to back off during pivot shift test to effect reduction. |
(FRD- flexion rotation drawer, ALRI- anterolateral rotatory instability, PS- pivot shift)
Examination under anaesthesia and arthroscopy:[edit | edit source]
Arthroscopy combined with examination under anaesthesia is an accurate way to diagnose a torn ACL. It may be indicated in the case whereby the diagnosis is suspected from the patient's history, but is not evident on clinical examination. The main value of using arthroscopy on the basis of examination is to diagnose associate joint pathologic conditions such as meniscal tears or chondral fractures.[70][71]
See this page for additional information on assessment of the knee: Knee Examination
Management[edit | edit source]
Please see Anterior Cruciate Ligament (ACL) Reconstruction
Please see Anterior Cruciate Ligament (ACL) Rehabilitation
Surgical or non-surgical management after an ACL tear is analyzed through systematic reviews and meta-analyses, where the absolute best standard of empirical research of the outcome of interventions is assessed. [72] Recent evidence based reviews have found similar results in both conservative and surgical approach groups with reference to pain levels, symptoms, function, return to sport participation, quality of life, following meniscal tear and surgery rates, and radiographic osteoarthritis of knee (OA) prevalence [73][74].
Injury Prevention[edit | edit source]
ACL injury rates appear to be on the increase and it is of concern that recent reports show the rates of ACL injury to have grown most rapidly at the younger end of the age spectrum. Therefore, it is timely to revisit the efficacy of ACL injury prevention training programs and critically evaluate the state of the current evidence for their effectiveness.[19]
Rates of non-contact ACL injury are higher among females than males. Several factors have been identified to explain this sex disparity. Gender differences have been found in motion patterns, positions, and muscular forces generated with various lower extremity coordinated activities. Anatomic and hormonal factors, such as a decrease in ACL circumference, a small and narrow intercondylar notch width, a decrease joint laxity and a pre-ovulatory phase of menstrual cycle in females, have been discussed as increased risk factors for non-contact ACL injuries. Level of evidence: [75][76]
However, modifying these particular risk factors is difficult if not impossible. In contrast, evidence indicates that neuromuscular risk factors are modifiable. Neuromuscular risk factors such as knee valgus position, muscular control (quadriceps and hamstrings muscular activation) and hip and trunk controls have been increasingly implicated in this injury aetiology. .[75][77]
Implementing an ACL injury prevention program can be extremely beneficial for all patients. Keep in mind that this program will not prevent ACL tears from occurring but can help decrease the risk. There are five key steps that should be included in the planning of this program:
- Identification.
- Exercises.
- Training load and volume.
- Training Frequency.
- Exercise timing.
Most ACL injuries occur when an anterior force is applied to the tibia. It is important to identify the risk factors that can contribute to this anterior force to reduce the chance of injury. Identification of risk factors and mechanisms of injury that are modifiable through neuromuscular based injury prevention programs would allow many athletes to continue sports participation and reduce risk for ACL injury. These modifiable risk factors are sorted into four different categories, including movement and alignment, strength, ground reaction forces (GRFs) and fatigue.
- Movement and Alignment – There are certain movement and alignment factors that can predispose a patient to an ACL tear, such as landing from a jump with a small knee flexion angle and larger knee valgus angle, decreased active and passive controls of the knee, and dynamic knee valgus positioning.
- Strength – Muscle weakness is another modifiable risk factor, specifically weak gluteus medius, gluteus minimus, quadriceps, hamstrings and hip abductor muscles.
- Weakened quadriceps may decrease knee flexion control.
- Weak hamstrings and hip abductors may lead to an increased valgus load on the knee.
- Weak core musculature will lead to decreased trunk stability and/or lateral pelvic movement.
- GRFs – If a patient has weak hamstrings or quadriceps, it may be hard for them to control GRF, which leads to a greater load on the ACL.
- Fatigue – Fatigue leads to loss of motor control, especially with the landing phase of a jump.
In 2018, Arundale, Bizzini, Giordano et al.[78] published Clinical Practice Guidelines (CPG) reviewing the latest injury prevention programs for ACL and knee ligament injuries. The results were extremely positive and state that “there is robust evidence for the advantages of exercise-based knee injury prevention programs, including reduction in risk for all knee injuries and for ACL injuries specifically, with little risk of adverse events and minimal cost”
Exercise-based prevention was defined as an intervention requiring the participant(s) to be active and move. This includes physical activity, strengthening, stretching, neuromuscular, proprioceptive, agility, or plyometric exercises and other training modalities. But it excludes passive interventions like bracing or programs that only involve education.
Recommendations[edit | edit source]
- It is recommended to implement this exercise-based knee injury prevention programs in athletes for prevention of knee and ACL injuries.
- This program should be implemented prior to training sessions or games i.e. as a part of warm-up.
- This CPG identifies three high risk populations and outlines different program most suited for each:
- Female athletes <18 years of age: PEP, Sportsmetric ,[79] Harmoknee,[80] Olsen et al,[81] Petersen et al.
- Soccer players, especially women: Caraffa et al,[82] Sportsmetric. [79]
- Male and female handball players, particularly 15-17 years of age: Olsen et al ,[81] Achenbach et al.[83]
- Dosage and Delivery: For all programs, the advice is that they ought to involve multiple components, have a session duration >20 minutes, have a weekly training volume >30 minutes, start at pre-season and continue throughout the season with high compliance.
- The most supported programs involved multiple components such as:
- Flexibility - Quadriceps, hamstrings, hip adductors, hip flexors, & calf muscles.
- Strengthening - Double-leg squats, single-leg squats, lunges, Nordic hamstring exercise.
- Plyometrics - Single leg hopping anterior & posterior, ice skaters, jump to header or catching a ball overhead.
- Balance & agility.
- Running - Forward & backwards, zigzag running, bounding forward & backwards.
- This CPG actually provides strong evidence to suggest that exercise-based prevention programs reduce the risk of all knee injuries, not just ACL injuries. “The pooled incident rate ratio indicated that exercise-based prevention programs are effective in reducing the incidence of knee injuries (0.73, 95% confidence interval)” (Arundale, Bizzini, Giordano et al., 2018).[78] For ACL specifically, the programs are also effective in reducing injury but the pooled ratio rate is lower ranging between 0.38-0.49.
- This information within this CPG includes all knee injuries, not just ACL injuries. The evidence and recommendations from this CPG should be used to educate and support coaches, parents, athletes and clinicians to incorporate exercise-based injury prevention programs into their training methods. It seems really important to ensure that this message reaches our young female athletes as they have been identified within each high risk population. Even though three high risk populations were identified, these recommendations should be implemented for all young athletes, particularly 12-25 years of age in high risk sports such as rugby, AFL, netball, soccer, basketball and skiing.
Phase I- dynamic warm up[edit | edit source]
Warm ups and cool downs are a critical part of a training program. The purpose of the dynamic warm-up phase is to allow the athlete to prepare for activity and it greatly reduces the risk of injury.
Part II: Foundational strengthening[edit | edit source]
This segment of the program focuses on increasing leg strength and provide more stable knee joint. Technique is everything; close attention must be paid to the performance of these exercises so as to avoid injury.
Part III: Movement coordination, deceleration, cutting, and plyometric training[edit | edit source]
These exercises are explosive and help to construct power, strength and speed. The most important component when considering performance technique is the landing. It must be soft! When landing from a jump transfer weight on the balls of feet slowly rolling back to the heel with a bent knee and a bent hip. These exercises are basic. However, it is important to perform them correctly. Start these exercise using a flat cone (2 inches) or with a visual line on the field.
The above video of on-field sport training program have been curated and published by JOSPT and provide a holistic program consistent with the recommendations of this clinical Practice guidelines for Exercise-Based Knee and Anterior Cruciate Ligament Injury Prevention. Recommended warm-up exercise sequence for athletes preparing to compete in field sports, such as soccer, football, lacrosse, field hockey, and softball etc.
Another Programs for reducing ACL injuries include HarmoKnee, FIFA 11+, Prevent Injury and Enhance Performance (PEP), and Sportsmetrics; and those used by Caraffa et al, and Olsen et al.
Fifa 11+, Harmoknee, PEP and Sportsmetric have their own Injury prevention program but what you would probably see in the table below is that no single program includes it all and from the CPG, that no single program was recommended as the number one program to follow.
| Flexibility | Running | Strength | plyometrics | core | Balance | |
| Harmoknee | Ö | Ö | Ö | Ö | Ö | |
| PEP | Ö | Ö | Ö | Ö | ||
| Sportsmetric | Ö | Ö | Ö | Ö | Ö | |
| FIFA 11+ | Ö | Ö | Ö | Ö | ||
| Olsen et al | Ö | Ö | Ö | Ö | ||
| Achenbach et al | Ö | Ö | Ö | Ö | ||
| Caraffa et al | Ö | Ö |
Below is a brief outline of the key programs presented in this CPG along with outline of dosage of each exercise.
FIFA 11+[edit | edit source]
The F-MARC 11+ program may be more effective at improving some risk factors for ACL injury among preadolescent female athletes than adolescent athletes, notably by reducing knee valgus angle and moment during a double-legged jump landing.[85]
PEP Program: Prevent injury and Enhance Performance [edit | edit source]
The PEP (Prevent injury, Enhance Performance) Program is a highly specific 15 minute training session which mainly focuses on educating an athlete on strategies to prevent injury and includes specific exercises targeting issues as identified in previous research studies.
1. Avoid vulnerable positions.
2. Increase flexibility.
3. Increase strength.
4. Include plyometric exercises in to the training program.
5.Increase proprioception though agilities. [86]
This prevention program comprises dynamic warm-up, flexibility, foundational strengthening, plyometrics, and sport specific agilities to deal with potential deficits in the strength and coordination of knee stabilizers. The coaches and trainers need to focus on correct posture, straight up and down jumps without excessive side-to-side movement, and reinforce soft landings. Optimally the program should be performed a minimum of 2-3 times per week during the season.
SPORTSMETRIC[edit | edit source]
- Flexibility: Gastrocnemius and soleus, quadriceps, hamstrings, hip adductor, hip flexors, latissimus dorsi, posterior deltoid and pectoralis major.
- Running: skipping, side shuffle and running.
- Strength: back hyperextension, leg press, calf raises, pull over, bench press, Latissimus dorsi pull down, forearm curl.
- Core strength: abdominal curl.
- Plyometrics: wall jumps, tuck jumps, broad jumps with stick landing, squat jumps, double leg cone jumps side to side, back to front and 180 degrees, bounding in place, vertical jumps bounding for distance, scissor jumps, hop, hop and stick landing, step jump up vertical, mattress jumps, single leg jumps for distance, jump into bounding.[79]
Harmoknee[edit | edit source]
- Flexibility: Standing calf stretch, standing quadricep stretch, half-kneeling hamstring stretch, half-kneeling hip flexor stretch, butterfly groin stretch and modified figure-four stretch.
- Jogging: jogging, backwards jogging on toe, high knee skipping, defensive pressure (zig zig backwards), alternate forward zig zag and backward zig zag running.
- Strength: lunges, Nordic hamstring strengthening and single leg squat with toe raise.
- Core stability: sit ups, plank on elbows and bridging.
- Plyometrics: forward and backward double leg jumps, lateral single leg jumps, forward and backward single leg jumps, double leg jump with or without the ball.[80]
In summary, there is no single program to recommend as the best exercise-based injury prevention program and there are many valuable resources available online to implement such programs to help in training. Overall, there is a robust evidence to suggest these programs are highly effective in injury prevention for an ACL injury. In conclusion, the finding of the analysis demonstrated that ACL injury reduction programs decrease the risk of all ACL injuries by half and non‐contact ACL injuries in all athletes by two‐thirds in female athletes. [88]
To successfully complete these prevention programs, time and commitment are most important. This CPG reinforces how important it is to teach our young athletes that these warms ups are the foundation for safe training and game play and to reduce the risk of injury, it is not an area we should compromise on. In fact, it might be the foremost valuable part of attending training and in the long run and keep people in the sport they love for longer.
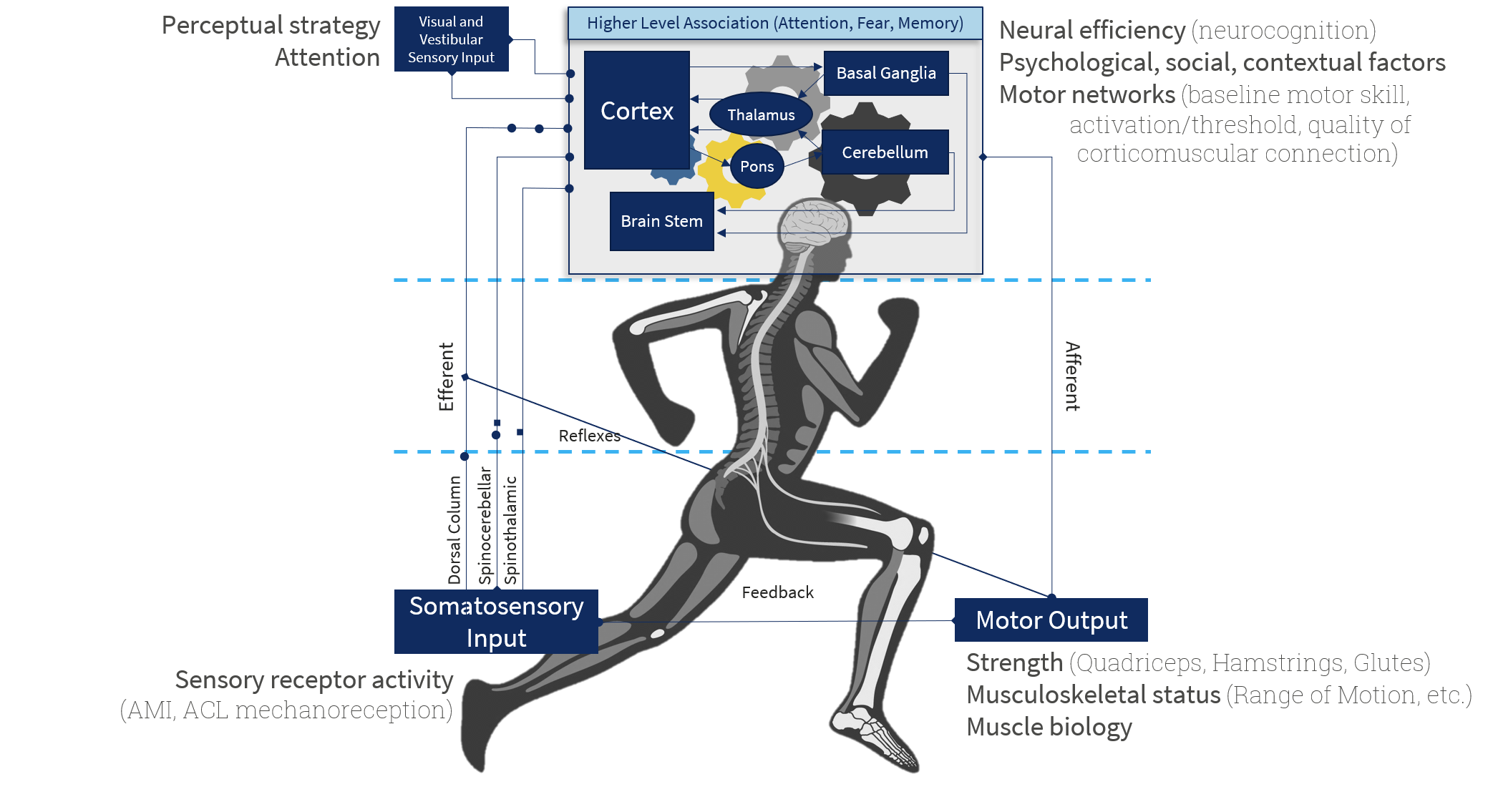
Neurocognitive Contributions to ACL injury[edit | edit source]
Neurocognition relates to a higher order of thinking or function in the brain. It includes but is not limited to the following -
- Executive functions - refers to the ability to coordinate motor, cognitive and emotional processess and play an important role in carrying out a task demanding attention as well as overriding the internal and external stimuli.
- Inhibitory control - refers to the ability to filter stimuli and focus on the task at hand. It relates to the working memory and plays an important role in the deployment of attention. It is required in tasks such as the rapidly changing environments on a football field.
- Cognitive flexibility - refers to the ability to adapt to changing conditions.
- Information Processing Speed - refers to the time required to process new information and retrieve data from memory. This is required to carry out complicated cognitive functions such as working memory.
- Reaction time - refers to the rapidness to respond to a stimuli
- Perception - refers to the ability to organize, identify, interpret, and understand the given information
- Dual or multi-tasking - refers to the ability to perform more than one action at a time
Expert athletes are those that combine motor and perceptual-cognitive skills. This addresses their ability to identify, locate and process information in a specific condition or environment. Team ball athletes like footballers and basketballers require rapid and efficient perceptive skills to interpret the opportunity to execute optimal performance like interpreting and identifying an opponent's movement before actually actioned. Errors in inhibitory control can lead to a faulty motor movement to fulfil the demand of sudden changes in action that can result in an ACL injury. Lacking the ability to switch focus can contribute to a loss of spatial awareness and disrupt motor control[89]. Studies have revealed changes or deficits in cognitive skills like reaction time, processing speed, and visual and verbal memory in ACL-injured athletes[90]. There have been advancements in noting changes in the central nervous system post ACL injury which should be noted and screening for neurocognitive factors should be implemented athletic population alongside the biomechanical factors. This has the potential to reduce the injury rate which can in turn decrease the years lost to disability.
Clinical Assessment Tools to Identify At‐Risk Athletes[edit | edit source]
Development of clinical assessment tools to identify athletes at risk for ACL injury would aid clinicians to target the populations that will benefit most from intervention. Although predictors of ACL injury that are potentially modifiable like measures of high knee abduction moment during landing tasks, these measurements utilized expensive measurement tools (e.g., motion analysis systems, force plates) and labor‐intensive data collection and reduction techniques to identify important biomechanical risk factors.
Identification of athletes with high knee abduction moments is possible with less expensive equipment and time. These clinical prediction tools show moderate to high inter‐rater reliability (intra‐class correlation co‐efficiencies 0.60–0.97) and have continued to simplify and optimize the screening tools to include a calibrated physician's scale, a standard measuring tape, standard camcorder, Image software, and an isokinetic dynamometer. These optimized measures predict high knee abduction moments status with 84% sensitivity and 67% specificity. A clinician‐friendly nomogram tool demonstrates over 75% prediction accuracy for identification of high knee abduction moments in individual athletes. Creation of clinician‐friendly, inexpensive techniques to identify and subsequently enroll athletes into appropriate injury prevention programs may help reduce ACL injuries in athletes.[19]
Assess the Effectiveness of Intervention[edit | edit source]
Common assessment tools such as the star excursion balance test, functional hop tests, strength measures, balance and stability measures and dynamometry, in addition to development of new techniques to help identify lower extremity asymmetry and high risk landing and cutting. These assessment tools as well as standard performance tests (e.g., power cleans, bench press, leg press) have been used to identify biomechanical and neuromuscular risk factors for ACL injury and provide measures of athletic performance. Assessments of the reliability of the assessment tools and performance measures have helped evaluate and optimize intervention strategies. In order to provide immediate, objective feedback that can be systematically tracked and used to evaluate intervention effectiveness. Clinical assessment tools such as the tuck jump assessment and nomogram that predicts high knee abduction measures may also help rehabilitation specialists working with athletes to monitor functional deficits and determine level of readiness to meet the functional demands of sports with minimal risk of re‐injury.[19]
Clinical Bottom Line[edit | edit source]
In order to provide the injured athlete with the best care, physiotherapists should have in-depth knowledge of the anatomy and functioning of the ACL. The keystone to proper care of an ACL injury is obtaining the correct diagnosis within the first hour of injury before the development of significant hemarthrosis. This should also include the detection of and diagnosis of associated injuries.[91] Injury treatment and the return to activities for an individual is entirely dependent upon the ACL injury grade and any associated injuries.
Resources[edit | edit source]
- ACL injury prevention - practical advice
- Clinical Practice Guidelines: Knee Ligament Sprain Revision 2017
- ACL tear (sports injury)
References[edit | edit source]
- ↑ 1.0 1.1 Nagano Y, Ida H, Akai M, Fukubayashi T. Biomechanical characteristics of the knee joint in female athletes during tasks associated with anterior cruciate ligament injury. The Knee. 2009 Mar 1;16(2):153-8.
- ↑ Arendt EA, Agel J,Dick R.Anterior cruciate ligament injury patterns among collegiate men and women. J Athl Train 1999;34:86-92.
- ↑ Garrick JG, Requa RK. Anterior cruciate ligament injuries in men and women: how common are they? In: Griffin LY, ed. Prevention of noncontact ACL injuries. Rosemont,IL:American Academy Orthopaedic Surgeons,2001:1-10.
- ↑ Agel J, Arendt E, Bershadsky B.Anterior cruciate ligament injury in national collegiate athletic association basketball and soccer: a 13 year review.Am J Sports Med 2005;33(4):524-30.
- ↑ Beynnon BD, Johnson RJ, Abate JA, Fleming BC, Nichols CE. Treatment of anterior cruciate ligament injuries, part I. The American journal of sports medicine. 2005 Oct;33(10):1579-602.
- ↑ 6.0 6.1 6.2 6.3 Domnick C, Raschke MJ, Herbort M. Biomechanics of the anterior cruciate ligament: Physiology, rupture and reconstruction techniques. World journal of orthopedics. 2016 Feb 18;7(2):82.
- ↑ Matsumoto, H., Suda, Y., Otani, T., Niki, Y., Seedhom, B. B., Fujikawa, K. (2001). Roles of the anterior cruciate ligament and the medial collateral ligament in preventing valgus instability. J Orthop Sci, 6(1), 28-32.
- ↑ Mark L. Purnell, Andrew I. Larson, and William Clancy. Anterior Cruciate Ligament Insertions on the Tibia and Femur and Their Relationships to Critical Bony Landmarks Using High-Resolution Volume-Rendering Computed Tomography. Am J Sports Med November 2008 vol. 36 no. 11 2083-2090
- ↑ Gerami MH, Haghi F, Pelarak F, Mousavibaygei SR. Anterior cruciate ligament (ACL) injuries: A review on the newest reconstruction techniques. Journal of Family Medicine and Primary Care. 2022 Mar;11(3):852.
- ↑ Singh JK, Verma A. PREVENTION OF ANTERIOR CRUCIATE LIGAMENT (ACL) INJURY AND ENHANCE PERFORMANCE PROGRAM. IJRAR-International Journal of Research and Analytical Reviews (IJRAR). 2020 Feb;7(1):715-27.
- ↑ 11.0 11.1 11.2 Brophy RH, Wojtys EM, Mack CD, Hawaldar K, Herzog MM, Owens BD. Factors associated with the mechanism of ACL tears in the National Football League: A video-based analysis. Orthopaedic journal of sports medicine. 2021 Nov 8;9(11):23259671211053301.
- ↑ 12.0 12.1 Wetters N, Weber AE, Wuerz TH, Schub DL, Mandelbaum BR. Mechanism of Injury and Risk Factors for Anterior Cruciate Ligament Injury. Operative Techniques in Sports Medicine. 2015 Oct 17.
- ↑ Parsons JL, Coen SE, Bekker S. Anterior cruciate ligament injury: towards a gendered environmental approach. British Journal of Sports Medicine. 2021 Sep 1;55(17):984-90.
- ↑ Geng B, Wang J, Ma JL, Zhang B, Jiang J, Tan XY, Xia YY. Narrow intercondylar notch and anterior cruciate ligament injury in female nonathletes with knee osteoarthritis aged 41–65 years in plateau region. Chinese medical journal. 2016 Nov 5;129(21):2540.
- ↑ McLean SG, Huang X, Van Den Bogert AJ. Association between lower extremity posture at contact and peak knee valgus moment during sidestepping: implications for ACL injury. Clinical biomechanics. 2005 Oct 1;20(8):863-70
- ↑ Mountcastle SB, Posner M, Kragh JF, Taylor Jr DC. Gender differences in anterior cruciate ligament injury vary with activity: epidemiology of anterior cruciate ligament injuries in a young, athletic population. The American journal of sports medicine. 2007 Oct;35(10):1635-42.
- ↑ Price MJ, Tuca M, Cordasco FA, Green DW. Nonmodifiable risk factors for anterior cruciate ligament injury. Current opinion in pediatrics. 2017 Feb 1;29(1):55-64.
- ↑ Thomson A, Whiteley R, Bleakley C. Higher shoe-surface interaction is associated with doubling of lower extremity injury risk in football codes: a systematic review and meta-analysis. British journal of sports medicine. 2015 Oct 1;49(19):1245-52.
- ↑ 19.0 19.1 19.2 19.3 Hewett TE, Myer GD, Ford KR, Paterno MV, Quatman CE. Mechanisms, prediction, and prevention of ACL injuries: cut risk with three sharpened and validated tools. Journal of Orthopaedic Research. 2016 Nov;34(11):1843-55.
- ↑ Haim A, Pritsch T, Yosepov L, Arbel R. Anterior cruciate ligament injuries. Harefuah. 2006 Mar;145(3):208-14.
- ↑ 21.0 21.1 21.2 Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. The American journal of sports medicine. 2004 Jun;32(4):1002-12.
- ↑ 22.0 22.1 Lambson RB, Barnhill BS, Higgins RW. Football cleat design and its effect on anterior cruciate ligament injuries: a three-year prospective study. The American journal of sports medicine. 1996 Mar;24(2):155-9.
- ↑ Kocher MS, Sterett WI, Briggs KK, Zurakowski D, Steadman JR. Effect of functional bracing on subsequent knee injury in ACL-deficient professional skiers. J Knee Surg. 2003 Apr;16(2):87-92. PMID: 12741421
- ↑ McDevitt ER, Taylor DC, Miller MD, Gerber JP, Ziemke G, Hinkin D, Uhorchak JM, Arciero RA, Pierre PS. Functional bracing after anterior cruciate ligament reconstruction: a prospective, randomized, multicenter study. Am J Sports Med. 2004 Dec;32(8):1887-92. doi: 10.1177/0363546504265998. PMID: 15572317.
- ↑ Orchard J, Seward H, McGivern J, Hood S. Intrinsic and extrinsic risk factors for anterior cruciate ligament injury in Australian footballers. The American journal of sports medicine. 2001 Mar;29(2):196-200.
- ↑ Musahl V, Nazzal EM, Lucidi GA, Serrano R, Hughes JD, Margheritini F, Zaffagnini S, Fu FH, Karlsson J. Current trends in the anterior cruciate ligament part 1: biology and biomechanics. Knee Surgery, Sports Traumatology, Arthroscopy. 2021 Dec 20:1-4.
- ↑ Ohori T, Mae T, Shino K, Tachibana Y, Fujie H, Yoshikawa H, Nakata K. Varus-valgus instability in the anterior cruciate ligament-deficient knee: effect of posterior tibial load. Journal of experimental orthopaedics. 2017 Dec;4(1):1-7.
- ↑ 28.0 28.1 28.2 Della Villa F, Buckthorpe M, Grassi A, Nabiuzzi A, Tosarelli F, Zaffagnini S, Della Villa S. Systematic video analysis of ACL injuries in professional male football (soccer): injury mechanisms, situational patterns and biomechanics study on 134 consecutive cases. British journal of sports medicine. 2020 Dec 1;54(23):1423-32.
- ↑ Shimokochi Y, Shultz SJ. Mechanisms of noncontact anterior cruciate ligament injury. Journal of athletic training. 2008 Jul;43(4):396-408.
- ↑ Waldén M, Krosshaug T, Bjørneboe J, Andersen TE, Faul O, Hägglund M. Three distinct mechanisms predominate in non-contact anterior cruciate ligament injuries in male professional football players: a systematic video analysis of 39 cases. British journal of sports medicine. 2015 Nov 1;49(22):1452-60.
- ↑ Lin CF, Liu H, Gros MT, Weinhold P, Garrett WE, Yu B. Biomechanical risk factors of non-contact ACL injuries: A stochastic biomechanical modeling study. Journal of Sport and Health Science. 2012 May 1;1(1):36-42.
- ↑ City Clinic on YouTube. ACL Tear (Sports Injury). Available from: http://www.youtube.com/watch?v=lpIOMuqXWrE [last accessed 04/10/14]
- ↑ Koga H, Nakamae A, Shima Y, Iwasa J, Myklebust G, Engebretsen L, Bahr R, Krosshaug T. Mechanisms for noncontact anterior cruciate ligament injuries: knee joint kinematics in 10 injury situations from female team handball and basketball. The American journal of sports medicine. 2010 Nov;38(11):2218-25.
- ↑ Renstrom P, Ljungqvist A, Arendt E, Beynnon B, Fukubayashi T, Garrett W, Georgoulis T, Hewett TE, Johnson R, Krosshaug T, Mandelbaum B. Non-contact ACL injuries in female athletes: an International Olympic Committee current concepts statement. British journal of sports medicine. 2008 Jun 1;42(6):394-412.
- ↑ William E.Prentice, Rehabilitation techniques for sports medicine and athletic training; fourth ed. McGraw Hill publications.
- ↑ Souryal TO, Freeman TR. Intercondylar notch size and anterior cruciate ligament injuries in athletes: a prospective study. The American journal of sports medicine. 1993 Jul;21(4):535-9.
- ↑ Shekhar¹ A, Singh¹ A, Laturkar¹ A, Tapasvi¹ S. Anterior Cruciate Ligament Rupture with Medial Collateral Ligament Tear with Lateral Meniscus Posterior Root Tear with Posterolateral Tibia Osteochondral Fracture: A New Injury Tetrad of the Knee. Journal of Orthopaedic Case Reports. 2020 May;10(3):36-42.
- ↑ 38.0 38.1 Yoon KH, Yoo JH, Kim KI.J. fckLRBone contusion and associated meniscal and medial collateral ligament injury in patients with anterior cruciate ligament rupture. Bone Joint Surg Am. 2011 Aug 17;93(16):1510-8.
- ↑ Niall DM, Bobic V, Surgeon CO, Lodge N. Bone bruising and bone marrow edema syndromes: incidental radiological findings or harbingers of future joint degeneration. J ISAKOS. 2004:22-5.
- ↑ Rick W. Wright, Mary Ann Phaneuf, Thomas J. Limbird and Kurt P. Spindler. Clinical Outcome of Isolated Subcortical Trabecular Fractures (Bone Bruise) Detected on Magnetic Resonance Imaging in Knees. Am J Sports Med September 2000 vol. 28 no. 5 663-667
- ↑ Mark A. Rosen, Douglas W. Jackson, Paul E. Berger. Occult osseous lesions documented by magnetic resonance imaging associated with anterior cruciate ligament ruptures. Arthroscopy: The Journal of Arthroscopic and Related SurgeryfckLRVolume 7, Issue 1 , Pages 45-51, March 1991
- ↑ R.B. Frobell, H.P. Roos, E.M. Roos, M.-P. Hellio Le Graverand, R. Buck, J. Tamez-Pena, S. Totterman, T. Boegard, L.S. Lohmande. The acutely ACL injured knee assessed by MRI: are large volume traumatic bone marrow lesions a sign of severe compression injury? Osteoarthritis and Cartilage, Volume 16, Issue 7, July 2008, Pages 829-836
- ↑ Viskontas DG, Giuffre BM, Duggal N, Graham D, Parker D, Coolican M. Bone bruises associated with ACL rupture: correlation with injury mechanism. Am J Sports Med. 2008 May;36(5):927-33. Epub 2008 Mar 19.
- ↑ Szkopek K, Warming T, Neergaard K, Jørgensen HL, Christensen HE, Krogsgaard M. Pain and knee function in relation to degree of bone bruise after acute anterior cruciate ligament rupture. Scand J Med Sci Sports. 2011 Apr 8. doi: 10.1111/j.1600-0838.2011.01297.x. [Epub ahead of print]
- ↑ 45.0 45.1 Atsuo Nakamae, Lars Engebretsen, Roald Bahr, Tron Krosshaug and Mitsuo Ochi. Natural history of bone bruises after acute knee injury: clinical outcome and histopathological findings. Knee Surgery, Sports Traumatology, Arthroscopy, Volume 14, Number 12, 1252-1258
- ↑ 46.0 46.1 Hollis G. Potter, Sapna K. Jain,Yan Ma, Brandon R. Black, Sebastian Fung and Stephen Lyman. Cartilage Injury After Acute, Isolated Anterior Cruciate Ligament Tear Immediate and Longitudinal Effect With Clinical/MRI Follow-up. Am J Sports Med February 2012 vol. 40 no. 2 276-285
- ↑ 47.0 47.1 Stallenberg B, Gevenois PA, Sintzoff Jr SA, Matos C, Andrianne Y, Struyven J. Fracture of the posterior aspect of the lateral tibial plateau: radiographic sign of anterior cruciate ligament tear. Radiology. 1993 Jun;187(3):821-5.
- ↑ Baker CL, Norwood LA, Hughston JC. Acute posterolateral rotatory instability of the knee. J Bone Joint Surg Am1983 ; 65:614 –618
- ↑ Chen FS, Rokito AS, Pitman MI. Acute and chronic posterolateral rotatory instability of the knee. J Am Acad Orthop Surg 2000; 8:97 –110
- ↑ Fanelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: part II. Arthroscopy1995 ; 11:526 –529
- ↑ Davies H, Unwin A, Aichroth P. The posterolateral corner of the knee: anatomy, biomechanics and management of injuries. Injury 2004; 35:68 –75
- ↑ Moorman CT 3rd, LaPrade RF. Anatomy and biomechanics of the posterolateral corner of the knee. J Knee Surg2005 ; 18:137 –145
- ↑ Harner CD, Vogrin TM, Hoher J, Ma CB, Woo SL. Biomechanical analysis of a posterior cruciate ligament reconstruction: deficiency of the posterolateral structures as a cause of graft failure. Am J Sports Med 2000; 28:32 –39
- ↑ LaPrade RF, Resig S, Wentorf F, Lewis JL. The effects of grade III posterolateral knee complex injuries on anterior cruciate ligament graft force: a biomechanical analysis. Am J Sports Med 1999 ; 27:469 –475
- ↑ Sansone V, De Ponti A, Paluello GM, Del Maschio A. Popliteal cysts and associated disorders of the knee. International orthopaedics. 1995 Oct 1;19(5):275-9.
- ↑ Stein D, Cantlon M, MacKay B, Hoelscher C. Cysts about the knee: evaluation and management. JAAOS-Journal of the American Academy of Orthopaedic Surgeons. 2013 Aug 1;21(8):469-79.
- ↑ Labropoulos N, Shifrin DA, Paxinos O. New insights into the development of popliteal cysts. British journal of surgery. 2004 Oct 1;91(10):1313-8.
- ↑ De Maeseneer M, Debaere C, Desprechins B, Osteaux M. Popliteal cysts in children: prevalence, appearance and associated findings at MR imaging. Pediatric radiology. 1999 Jul 1;29(8):605-9.
- ↑ Shelbourne KD,Davis TJ, Klootwyk TE. The relationship between intercondylar notch width of the femur and the incidence of anterior cruciate ligament tears. A prospective study.Am J Sports Med 1998;26:402-408
- ↑ Souryal TO, Moore HA, Evans JP,Intercondylar notch size and anterior cruciate ligament injuries in athletes.A prospective study: Am J Sports Med 16:449,1988.
- ↑ Turner da,Podromos CC, Petsnick JP, Clark JW: Acute injury of the knee: Magnetic resonance evaluation.Radiology 154:711-722,1985.
- ↑ Johnson DL, Urban WP, Caborn DN, Vanarthos WJ, Carlson CS. Articular cartilage changes seen with magnetic resonance imaging-detected bone bruises associated with acute anterior cruciate ligament rupture. The American journal of sports medicine. 1998 May;26(3):409-14.
- ↑ DeLee, Drez, Muller. Orthopaedic sports Medicine,Principles and Practice. Vol 2; 2nd edition.Saunder's publication, printed in USA.
- ↑ Kowalk DL,Wojtys EM,Disher J,Loubert P:Quantitative analysis of the measuring capabilities of the KT1000 knee ligament arthrometer. Am J Sports Med 21:744-747,1993.
- ↑ Sun Hwa Lee, Seong Jong Yun, Efficiency of knee ultrasound for diagnosing anterior cruciate ligament and posterior cruciate ligament injuries: a systematic review and meta-analysis, Skeletal Radiology, 10.1007/s00256-019-03225-w, (2019)
- ↑ Schwenke M, Singh M, Chow B. Anterior Cruciate Ligament and Meniscal Tears: A Multi-modality Review. Appl Radiol. 2020;49(1):42-49
- ↑ Tony Lowe. MRI scan left knee. Available from: http://www.youtube.com/watch?v=cOWszWYN_a8[last accessed 04/10/14]
- ↑ DeLee, Drez, Muller. Orthopaedic sports Medicine,Principles and Practice. Vol 2; 2nd edition. Saunder's publication, printed in USA.
- ↑ Lower Extremity- Flexion- Rotation Drawer Test (Noyes). Available from:https://www.youtube.com/watch?v=NrwWBRGL-1w
- ↑ DeHaven KE: Diagnosis of acute knee injuries with hemarthrosis, Am J Sports Med 8:9,1980.
- ↑ Noyes FR, Bassett RW, Grood ES, Butler DL. Arthroscopy in acute traumatic hemarthrosis of the knee. Incidence of anterior cruciate tears and other injuries. The Journal of bone and joint surgery. American volume. 1980 Jul;62(5):687-95.
- ↑ Traver JL, Kocher MS. Return-to-Sport Considerations in the Pre-Adolescent Athlete. InReturn to Sport after ACL Reconstruction and Other Knee Operations 2019 (pp. 593-605). Springer, Cham.
- ↑ Smith TO, Postle K, Penny F, McNamara I, Mann CJ. Is reconstruction the best management strategy for anterior cruciate ligament rupture? A systematic review and meta-analysis comparing anterior cruciate ligament reconstruction versus non-operative treatment. The Knee. 2014 Mar 1;21(2):462-70.
- ↑ Monk AP, Davies LJ, Hopewell S, Harris K, Beard DJ, Price AJ. Surgical versus conservative interventions for treating anterior cruciate ligament injuries. Cochrane Database of Systematic Reviews. 2016(4).
- ↑ 75.0 75.1 Sugimoto D, Myer GD, Bush HM, Klugman MF, McKeon JM, Hewett TE. Compliance with neuromuscular training and anterior cruciate ligament injury risk reduction in female athletes: a meta-analysis. Journal of athletic training. 2012;47(6):714-23.
- ↑ Alentorn-Geli E, Myer GD, Silvers HJ, Samitier G, Romero D, Lázaro-Haro C, Cugat R. Prevention of non-contact anterior cruciate ligament injuries in soccer players. Part 1: Mechanisms of injury and underlying risk factors. Knee surgery, sports traumatology, arthroscopy. 2009 Jul 1;17(7):705-29.
- ↑ Thompson JA, Tran AA, Gatewood CT, Shultz R, Silder A, Delp SL, Dragoo JL. Biomechanical effects of an injury prevention program in preadolescent female soccer athletes. The American journal of sports medicine. 2017 Feb;45(2):294-301.
- ↑ 78.0 78.1 Arundale AJ, Bizzini M, Giordano A, Hewett TE, Logerstedt DS, Mandelbaum B, Scalzitti DA, Silvers-Granelli H, Snyder-Mackler L, Altman RD, Beattie P. Exercise-based knee and anterior cruciate ligament injury prevention: clinical practice guidelines linked to the international classification of functioning, disability and health from the academy of orthopaedic physical therapy and the American Academy of sports physical therapy. Journal of Orthopaedic & Sports Physical Therapy. 2018 Sep;48(9):A1-42.
- ↑ 79.0 79.1 79.2 Hewett TE, Lindenfeld TN, Riccobene JV, Noyes FR. The effect of neuromuscular training on the incidence of knee injury in female athletes. The American journal of sports medicine. 1999 Nov;27(6):699-706.
- ↑ 80.0 80.1 Kiani A, Hellquist E, Ahlqvist K, Gedeborg R, Byberg L. Prevention of soccer-related knee injuries in teenaged girls. Archives of internal medicine. 2010 Jan 1;170(1):43-9.
- ↑ 81.0 81.1 Olsen OE, Myklebust G, Engebretsen L, Holme I, Bahr R. Exercises to prevent lower limb injuries in youth sports: cluster randomised controlled trial. BMJ. 2005;330:449
- ↑ Caraffa A, Cerulli G, Projetti M, Aisa G, Rizzo A. Prevention of anterior cruciate ligament injuries in soccer. A prospective controlled study of proprioceptive training. Knee Surg Sports Traumatol Arthrosc. 1996;4(1):19-21. doi: 10.1007/BF01565992. PMID: 8963746.
- ↑ Achenbach L, Krutsch V, Weber J, Nerlich M, Luig P, Loose O, Angele P, Krutsch W. Neuromuscular exercises prevent severe knee injury in adolescent team handball players. Knee surgery, sports traumatology, arthroscopy. 2018 Jul 1;26(7):1901-8.
- ↑ Knee injury prevention CPG: warm up exercise sequence for field sports Available from: https://youtu.be/RfROpda4kvg
- ↑ Thompson-Kolesar JA, Gatewood CT, Tran AA, Silder A, Shultz R, Delp SL, Dragoo JL. Age influences biomechanical changes after participation in an anterior cruciate ligament injury prevention program. The American journal of sports medicine. 2018 Mar;46(3):598-606.
- ↑ Mandelbaum BR, Silvers HJ, Watanabe DS, Knarr JF, Thomas SD, Griffin LY, Kirkendall DT, Garrett Jr W. Effectiveness of a neuromuscular and proprioceptive training program in preventing anterior cruciate ligament injuries in female athletes: 2-year follow-up. The American journal of sports medicine. 2005 Jul;33(7):1003-10.
- ↑ ACL injury prevention Exercises (PEP program) Available from:https://youtu.be/7Lag8uNU6AQ
- ↑ Webster KE, Hewett TE. Meta-analysis of meta-analyses of anterior cruciate ligament injury reduction training programs. J Orthop Res. 2018 Oct;36(10):2696-2708. doi: 10.1002/jor.24043. Epub 2018 Jun 13. PMID: 29737024.
- ↑ Gokeler A, Benjaminse A, Della Villa F, Tosarelli F, Verhagen E, Baumeister J. Anterior cruciate ligament injury mechanisms through a neurocognition lens: implications for injury screening. BMJ open sport & exercise medicine. 2021 May 1;7(2):e001091.
- ↑ Piskin D, Benjaminse A, Dimitrakis P, Gokeler A. Neurocognitive and neurophysiological functions related to ACL injury: a framework for neurocognitive approaches in rehabilitation and return-to-sports tests. Sports Health. 2022 Jul;14(4):549-55.
- ↑ Brukner, Khan. Clinical Sports Medicine. 3rd edition.Ch 27. Tata McGraw-Hill Publishing. New Delhi.