Chronic Traumatic Encephalopathy
Original Editor - Gareth Geoffreys
Top Contributors - Gareth Geoffreys, Lucinda hampton, Kim Jackson, Rucha Gadgil, Aminat Abolade and Carina Therese Magtibay
Introduction[edit | edit source]
Chronic Traumatic Encephalopathy (CTE) is a progressive, neurodegenerative tauopathy disorder associated with a history of repetitive brain trauma. It is characterised by the accumulation of tau protein within the brain, which leads to cognitive, behavioural, and physical impairments over time. While the condition was initially recognised in individuals involved in high-contact professions such as professional boxing, it's now understood that CTE can affect a wide range of individuals who've experienced repeated head injuries, including those from other contact sports like football, rugby, and hockey, as well as military personnel and others subject to repeated concussive and sub-concussive blows[1][2].
- CTE is a neurodegenerative disorder that can only be confirmed in a post-mortem autopsy[3].
- Unfortunately, there is currently no treatment for CTE. Prevention, therefore, is of great importance.[4]
Epidemiology[edit | edit source]
The exact incidence and prevalence of CTE is unknown. Most commonly it is seen in amateur and professional sports players where head contact is common (e.g. boxing, American football, rugby, ice hockey), as well as in military personnel exposed to explosive blasts.[2]
Neuropathology[edit | edit source]
CTE is a tauopathy that arises from the accumulation of repetitive mild traumatic brain injuries (MTBI)[5]. Post-mortem analyses have revealed that the symptoms of CTE are associated with neuropathological changes in the brain, such as atrophy of certain brain structures and degeneration of myelinated neurones.[6] Like other neurodegenerative disorders, CTE is characterised by the accumulation of abnormal tau proteins.
Historically, CTE was first recognised in boxers under the term "Dementia Pugilistica," also known as "punch-drunk" syndrome. Today, we understand that Dementia Pugilistica can be considered a subtype or precursor to our broader understanding of CTE. It's important to note that CTE encompasses a wider range of potential causes and manifestations, not restricted to professional boxing.[2]
Other conditions associated with repetitive MTBI and similar neuropathological changes include Alzheimers, Parkinsons, and, in some cases, Amyotrophic Lateral Sclerosis (ALS). Each of these conditions, while distinct in their clinical presentation and progression, share a common thread of neurodegeneration, which is likely influenced by a combination of genetic, environmental, and lifestyle factors.[7]
Symptoms[edit | edit source]
Symptoms have an insidious onset, most often years after the initial injuries, with loss of normal attention, concentration, impaired judgement, aggression, depression and memory loss. This can progress, in some cases in 2-3 years, to include motor symptoms such as impaired gait, impaired, executive function, lack of insight and poor judgment[2]
- Motor dysfunction only occurs at later stages of Neurodegeneration and is strongly correlated with age[6].
- With Age, CTE is often found to develop into more serious forms of neurological disorders, such as Motor Neurone Disease, Alzheimer's Disease and Lewy Body Disease.
Diagnosis[edit | edit source]
Single incidences of head trauma such as concussion will very rarely result in the development of CTE. However, repeated brain trauma sustained at the sub-concussive and concussive level has been found to be a strong predictor of CTE Development[6].
The core diagnostic features present in more than 70% of confirmed CTE cases without comorbidities falls into the three domains:
- Cognitive symptoms include impairments in memory and executive functioning.
- Behavioral symptoms include verbal and physical violent behavior, explosivity, and impulsivity.
- Mood symptoms often include depression.
Differentiation of these various manifestations of CTE from other forms of TBI like post-concussive syndrome, remains difficult.[8]
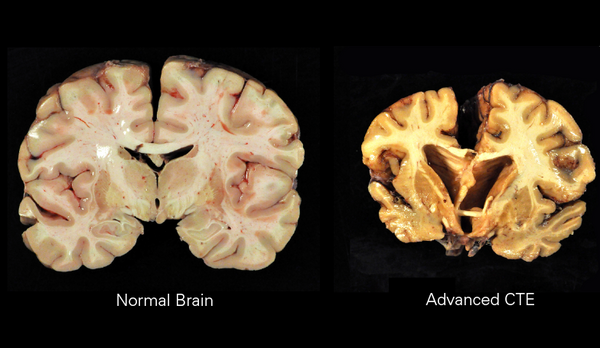
There are several brain findings of CTE on autopsy. Notably, there is accumulation in various areas of the brain of certain proteins eg as tau (distinct from Alzheimer’s disease, which shows beta-amyloid plaques). In addition there is: reduced brain weight; thinning of the corpus callosum; frequent atrophy of the frontal lobes. Other affected areas of the brain include the mammillary bodies, hippocampus, and medial temporal lobe, which are involved with memory, as well as the substantia nigra, which is involved with movement.[4]
Management[edit | edit source]
The development of CTE is specific to each patient, therefore, a case by case therapeutic intervention should be applied.
- There is no treatment available for CTE once it has developed. As is usually the case, prevention is the best medicine.
- The need for a safe culture in sports and the rest of life is becoming increasingly emphasized.
Physiotherapy[edit | edit source]
Although the precise mechanisms are not fully understood, exercise (specifically aerobic exercise) can increase neurogenesis and neuroplasticity[9]. As little as 30 minutes of light to moderate intensity exercise five days a week can greatly slow down the onset of neurological disorder[10].
More information regarding the role exercise has in the regulation of neurological conditions can be found here.
Cognitive Rehabilitation[edit | edit source]
Implementing cognitive rehabilitation at the early stages of cognitive impairment has a greater chance of positively effecting the quality of life of the individual as they age[3]. Cognitive rehabilitation can be divided into components: restorative and compensatory[11]. The restorative approach aims at restoring impaired skills by carrying out repeated exercise of standardized cognitive tests. These tests should aim to increase in difficulty and should target specific cognitive domains appropriate to the patients needs. Compensatory approach teaches ways of bypassing or compensating for the impaired function[11]. Assistive technology, such as prospective memory aids (PMAs) and retrospective memory aids (RMAs) are used in compensatory interventions. these are context-aware aids that make use of artificial intelligence to determine if a particular guidance is necessary or not at the moment, thus help to remember future intentions[12].
Mood/Behaviour therapy[edit | edit source]
For patients with prominent mood (depression, hopelessness, and anxiety) and behavioral (explosivity, impulsivity, short fuse) symptoms, psychological therapy/counseling by a clinical psychologist, neuropsychologist, or psychiatrist is recommended[3].
References[edit | edit source]
- ↑ Omalu B, DeKosky S, Minster R, Kamboh M, Hamilton R, Wecht C. Chronic Traumatic Encephalopathy in a National Football League Player. Neurosurgery. 2006;:E1003.
- ↑ 2.0 2.1 2.2 2.3 Radiopedia CTE Available: https://radiopaedia.org/articles/chronic-traumatic-encephalopathy?lang=us(accessed 18.3.2022)
- ↑ 3.0 3.1 3.2 Cantu R, Budson A. Management of chronic traumatic encephalopathy. Expert Review of Neurotherapeutics. 2019 Oct 3;19(10):1015-23.
- ↑ 4.0 4.1 Very well health CTE Available: https://www.verywellhealth.com/chronic-traumatic-encephalopathy-2488875(accessed 18.3.2022)
- ↑ Gardner RC, Yaffe K. Epidemiology of mild traumatic brain injury and neurodegenerative disease. Molecular and Cellular Neuroscience. 2015 May 1;66:75-80.
- ↑ 6.0 6.1 6.2 McKee AC, Stein TD, Kiernan PT, Alvarez VE. The neuropathology of chronic traumatic encephalopathy. Brain pathology. 2015 May;25(3):350-64.
- ↑ Gardner RC, Yaffe K. Epidemiology of mild traumatic brain injury and neurodegenerative disease. Molecular and Cellular Neuroscience. 2015 May 1;66:75-80.
- ↑ Pierre K, Dyson K, Dagra A, Williams E, Porche K, Lucke-Wold B. Chronic Traumatic Encephalopathy: Update on Current Clinical Diagnosis and Management. Biomedicines. 2021 Apr;9(4):415.Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8069746/(accessed 18.3.2022)
- ↑ Kimhy D, Vakhrusheva J, Bartels MN, Armstrong HF, Ballon JS, Khan S, Chang RW, Hansen MC, Ayanruoh L, Lister A, Castrén E. The impact of aerobic exercise on brain-derived neurotrophic factor and neurocognition in individuals with schizophrenia: a single-blind, randomized clinical trial. Schizophrenia bulletin. 2015 Jul 1;41(4):859-68.
- ↑ Müllers P, Taubert M, Müller NG. Physical exercise as personalized medicine for dementia prevention?. Frontiers in Physiology. 2019 May 29;10:672.
- ↑ 11.0 11.1 Shoulson I, Wilhelm EE, Koehler R, editors. Cognitive rehabilitation therapy for traumatic brain injury: evaluating the evidence. National Academies Press; 2012 Jan 28.
- ↑ Gupta S, Mishra C, Katyayan P, Joshi N. Assistive Technology for Neurological Disorders. InProceedings of 3rd International Conference on Internet of Things and Connected Technologies (ICIoTCT) 2018 Apr 20 (pp. 26-27).