Anatomy and Relevant Structures in Plantar Heel Pain
Original Editor - Merinda Rodseth based on the course by
Bernice Saban
Top Contributors - Merinda Rodseth, Wanda van Niekerk, Kim Jackson, Jess Bell, Olajumoke Ogunleye and Tarina van der Stockt
Introduction[edit | edit source]
Plantar Heel Pain (PHP) is a poorly understood, complex condition. During previous courses in this series, Plantar Heel Pain Syndrome (PHPS) has been introduced, the available literature on its risk factors, assessment and treatment explored and a new protocol for its management discussed. In order to optimally manage PHPS, it is necessary to examine the anatomical structures underlying the calf and foot area in order to establish the relationship between PHP and the tissues involved. In this document, the muscular, nervous and fascial structures underlying the foot and ankle will be explored in relation to:
- Previous theories on PHPS (including Plantar Fasciitis and Calcaneal Spurs)
- The “new protocol” for the management of PHPS
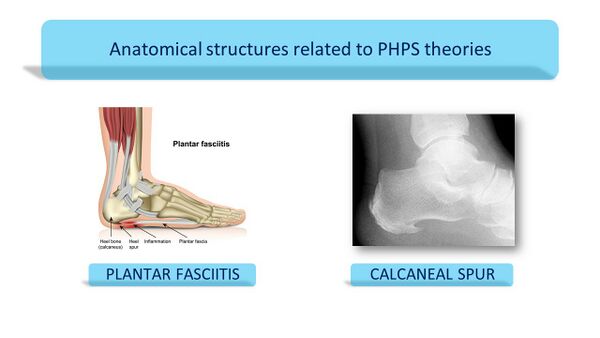
Anatomical Structures Related to PHPS Theories[edit | edit source]
In this section, the anatomical structures will be discussed in relation to the two most prominent theories about PHPS: Plantar Fasciitis and Calcaneal spurs.
Plantar Fascia[edit | edit source]
Fascia consists of sheets of connective tissue that form a continuous network through the whole body.[1][2] It is a collagen-based tissue that attaches, stabilises, imparts strength, encloses different organs, supports internal structures and envelopes whole muscles, as well as each individual muscle fibre.[1][3] Fascia can be classified as superficial, deep, visceral or parietal and is often further classified based on anatomical location.[1] Fascial thickness varies from very thin, almost transparent to strong and thickened.
The fascia of the foot consists of fibrous connective tissue that functions to separate, support and attach muscles.[4] It can also be divided into the superficial and the deep fascia. On the plantar side of the foot, the superficial fascia is involved in the formation of the plantar fat pad whereas the deep layer, known as the plantar fascia, plays an essential role in maintaining the medial longitudinal arch of the foot.[5][4][6][7][8]
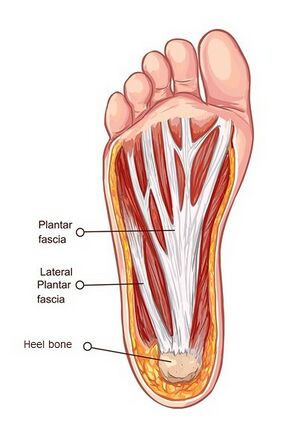
The plantar fascia (PF), also known as the plantar aponeurosis, originates proximally at the medial tubercle of the distal calcaneus and broadens as it extends distally (Figure 1).[3][4][7] Distally, at the metatarsophalangeal joints, it divides into five digital slips to each of the toes. These fuse with the fibrous flexion sheaths and the deep transverse metatarsal ligaments in each of the toes before inserting at the base of the proximal phalanges.[3][9][10] Besides its support of the longitudinal arch of the foot, the PF is also involved in the mechanisms of propulsion, transmitting the forces and stresses in the foot during loading (shock-absorbing).[4][6][8]
It is worth noting again that the PF is distal to the painful area in the heel commonly reported in PHPS, which raises the question as to whether it can be a causative factor in PHPS.
Plantar Fasciitis[edit | edit source]
Microtrauma caused by excessive tension to the PF is commonly thought to be the source of pain in PHPS. The Windlass Test has been proposed as an assessment technique in the diagnosis of plantar fasciitis based on the idea that passive lengthening of the plantar fascia, induced by extension of the big toe, can produce pain in the heel (Figure 2).
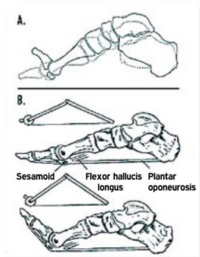
This assumption was further studied by Fessel et al.[11] who assessed the function of the PF in various phases of gait and concluded that the “windlass effect” for support of the arch is questionable due to the substantial muscle contribution to support the longitudinal arch (Figure 3).
Figure 3. Muscular support of the longitudinal arch[3][11]
In an anatomical dissection study of the PF, Stecco et al.[6] identified three parts to the PF - medial, central and lateral - with the central part being the thickest. All dissections revealed that the[6]:
- PF continued over the calcaneal bone with a thin band corresponding to the periosteum of the calcaneal bone
- This layer of the PF surrounded the calcaneus and was continuous with the paratenon of the Achilles tendon
The continuity of collagen fibres between the PF and the Achilles tendon remains highly controversial and is still widely debated.[6][12]
Further microscopic study revealed the presence of Pacini and Ruffini corpuscles inside the PF, which suggests that PF innervations have a role in proprioception and in the stability and control of foot movements.[6]
Stecco et al.[6] further raised the question of whether the PF is indeed a fascia or if it is an aponeurosis. These terms are generally used interchangeably in various studies.
Fascia - tissue with a multidirectional arrangement of the collagen fibres
Aponeurosis - tissue with a unidirectional arrangement of the collagen fibres
In their anatomical study, Stecco et al.[6] found that even though the collagen fibres of the PF were mainly arranged in a proximal-to-distal longitudinal direction, various fibres also lay in vertical, transverse and oblique directions. They concluded that this multilayer configuration of the collagen fibres is more typical of fasciae and that the term “plantar fascia” would therefore be a more appropriate term for this tissue.[6]
Besides the structure of the PF, a more pressing question is whether the PF could actually be a source of the pain reported in PHPS. Currently, no evidence could be found of the existence of nociceptive nerve endings in the PF itself. However, it has been suggested that while performing a long stretch, for example during neurodynamic testing, both the fascial and neural tissue will be stretched as both of these systems are continuous through the body.[3] There is no doubt that neurodynamic tests not only load the nervous system, but also challenge non-neural structures.[3]
Following this concept, Coppieters et al.[13] studied the impact of neurodynamic testing (slump and straight leg raise [SLR]) on the perception of experimentally induced pain to assess whether stress on the fascia could be an alternative explanation for changes in pain perception during neurodynamic tests. They injected hypertonic saline into the tibialis anterior muscle of 15 asymptomatic volunteers in order to add strain to the fascia and performed the SLR and slump neurodynamic tests while prohibiting and monitoring all other movements of the ankle (Figure 4).[13]
Figure 4. The impact of neurodynamic testing on the perception of experimentally induced muscle pain [3][13]
Coppieters et al.[13] found no change in the perception of experimentally induced pain with the SLR and slump neurodynamic tests and concluded that these neurodynamic tests have no impact on pain perception when the pain is not of neural origin (Figure 5).
Figure 5. The outcome of neurodynamic testing on pain perception [3][13]
These findings were supported by Coppieters et al.[14] in another study that investigated whether any mechanical movements occurred in the fascia and nerves when performing a modified SLR manoeuvre. They inserted gauges into the sciatic, tibial and plantar nerves as well as the PF of 8 embalmed cadavers to measure the strain during a modified SLR test. They found that even though there was significant movement in the tibial nerve in the tarsal tunnel during the modified SLR tibial manoeuvre, no movement occurred in the PF (Figure 6).[14]
Figure 6. Effect of a modified SLR test on the nerves and fascia [3][14]
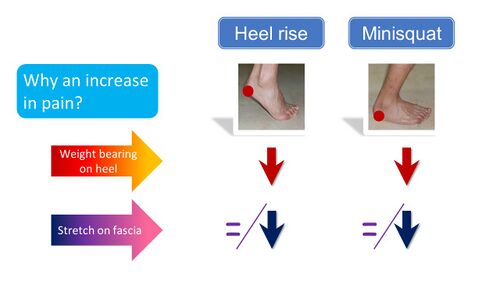
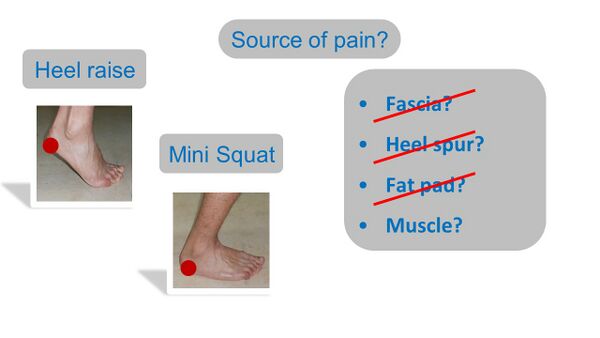
The results of this study are surprising considering the long stretch on the fascia. This brings us back to the previously asked question considering the cause of the reproduced heel pain with two standard tests - the heel raise and the mini squat (Figure 7). Both weight-bearing on the heel and stretch on the fascia can hereby be mostly excluded as reasons for the PHP elicited.[3]
Figure 7. Reasoning for reproduced heel pain with standard tests [3]
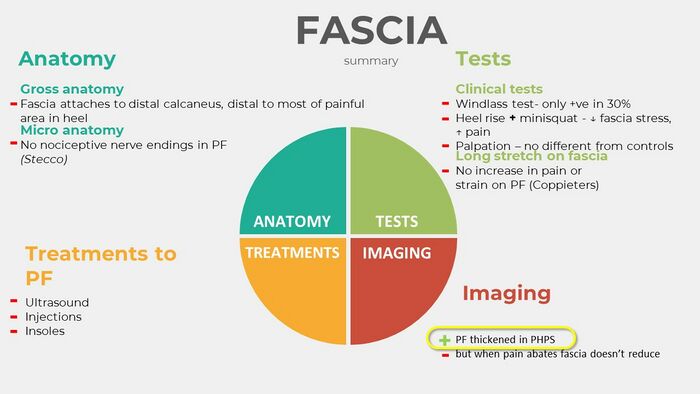
In summary of all the evidence gathered for the involvement of the PF in PHPS over the course of the various lectures on heel pain, support could only be found for the thickening of the PF in PHPS (Figure 8).[3]
Figure 8. Summary of the evidence for the involvement of the Plantar Fascia in PHPS (- refers to no support in the literature while + indicates support in the literature) [3]
Calcaneal Spurs[edit | edit source]
Plantar calcaneal spur has been widely implicated as a cause of plantar fasciitis but it remains controversial whether the calcaneal spur actually contributes to the symptoms of PHP.[7][15][16] Considering the previous review on the risk factors for PHPS, calcaneal spurs are generally considered as incidental findings as they are not located in the weight-bearing area of the heel and have also been found in asymptomatic individuals.[3][7] No clear connection, therefore, exists between the presence of calcaneal spurs and PHPS.
Anatomical Structures Related to the “New Protocol”[edit | edit source]
Many structures have, therefore, been eliminated as possible sources of the heel pain reproduced with the heel raise and mini squat tests. Thus, it is necessary to consider if the source of pain could be muscular instead (Figure 9)?
Figure 9. Possible sources of the heel pain reproduced with clinical tests [3]
Muscular System[edit | edit source]
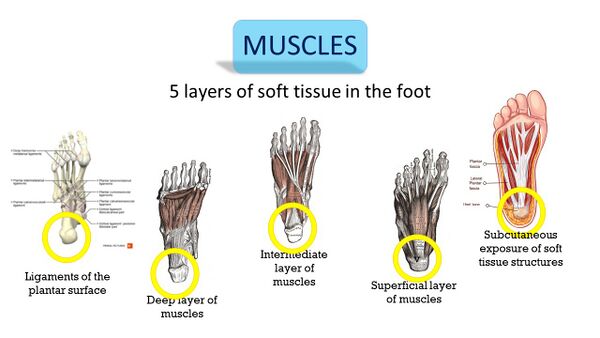
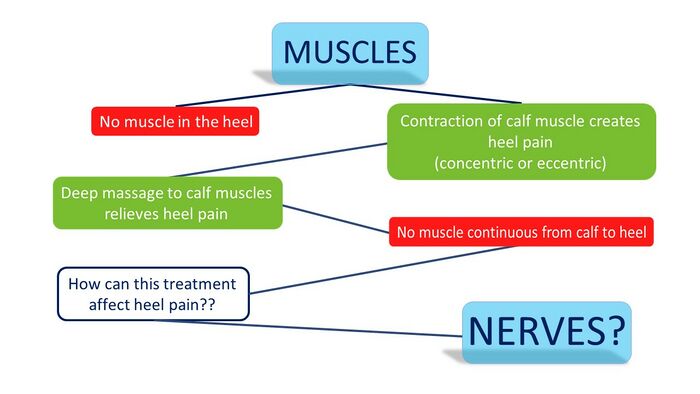
Of the five layers of soft tissue in the foot, four do not cover the heel (Figure 10). Only the superficial layer covers the heel with the fat pad, which has very little nociceptive innervation and has not been proven as a source of pain in the heel.[3] Considering that all the muscles running from the calf to the foot bypass the heel to enter the foot more distally, and that there are no muscles in the heel itself, the muscular system can be eliminated as a source of the PHP. With no evidence for the muscles as a source, only the neural tissues remain to be considered (Figure 11).[3]
Figure 10. Five layers of soft tissue in the foot [3]
Figure 11. Reasoning around the muscles as a source for the PHP with the clinical tests [3]
Neural Tissues[edit | edit source]
Before exploring the neural tissue of the foot, it is worth revisiting the results of the pressure pain threshold tests performed by Saban and Masharawi[17] in PHPS. Their results indicate that even though the presentation and sensitivity of the heel pain were not significantly different between individuals with PHPS and those without, the distal medial heel was the most sensitive area in the heel.[17] When considering the neural anatomy of the foot, it is of interest to note that the entrance of the medial and lateral plantar nerves into the foot coincides with this reported area of increased sensitivity in the heel (Figure 12).
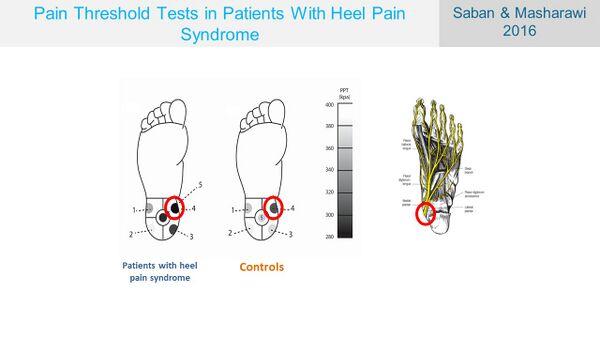 Figure 12. Similarities of the PPT test sensitivity and the neural anatomy [17]
Figure 12. Similarities of the PPT test sensitivity and the neural anatomy [17]
The tibial nerve passes through the fibrous tarsal tunnel before splitting into the medial and lateral plantar nerves.[18] In this part of its course, the tibial nerve is very superficial as it is wedged between the bone and the skin with no other protection. Palpation of the nerve will easily evoke paraesthesia similar to palpation of the ulnar nerve at the elbow.[18] From here, the medial and lateral plantar nerves, the medial calcaneal nerve branch of the tibial nerve and the arteries and veins enter the foot close to the medial calcaneal tubercle.[17][19]
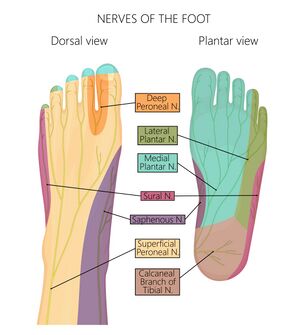
Cutaneous innervation of the foot is supplied by 7 different nerves altogether (Figure 13):
- Saphenous nerve (L3,4)
- Deep peroneal nerve (L4,5)
- Superficial peroneal nerve (L4, S1)
- Medial plantar nerve (L4,5)
- Lateral plantar nerve (S1,2)
- Medial calcaneal branch of the tibial nerve (S1,2)
- Sural nerve (S1,2)
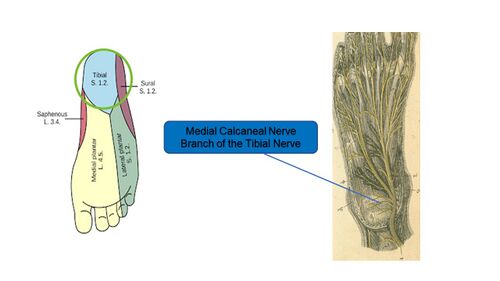
In relation to PHPS, the nerve that is of greatest interest is the medial calcaneal nerve. The medial calcaneal nerve is one of the main branches of the tibial nerve and generally separates from the tibial nerve below the medial malleolus around the area of the tarsal tunnel.[19][20] From here it enters the heel from the medial side before terminating in the skin of the heel, providing sensory innervation to the skin of the heel area (Figure 14).[19]
Figure 14. Medial calcaneal nerve branch of the tibial nerve
Other researchers have investigated the involvement of neural tissues in PHPS:
- Two studies using nerve conduction tests of the plantar nerves [21][22]
- One review explored heel pain of neural origin [23]
David Butler[18] also notes a peripheral neurogenic contribution to some heel spurs, either from the medial calcaneal nerve or the lateral plantar nerve.
“Heel spurs are also one pain state where the exact pain may not be replicated on physical evaluation, but on neurodynamic testing there are clues that something is not quite right. This could be minimal restrictions in range of motion, or symptoms evoked on the problem side but not the “good” side” - David Butler [18]
He further suggested treating PHPS with neurodynamic manoeuvres, specifically combining the SLR test with dorsiflexion/eversion of the ankle.[18]
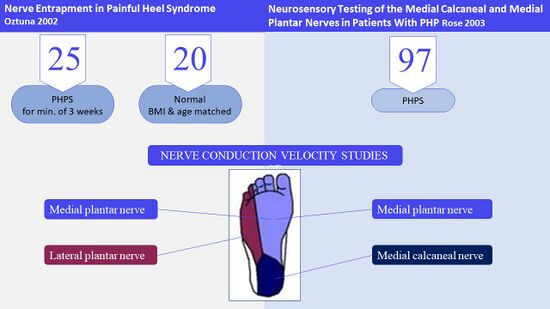
Both nerve conduction studies by Öztuna et al.[21] and Rose et al.[22] assessed the medial plantar nerve (MPN). Öztuna et al.[21] also investigated the lateral plantar nerve (LPN) while Rose et al.[22] examined the medial calcaneal nerve (MCN) in order to observe any differences in nerve conduction between these nerves (Figure 15).
Figure 15. Nerve conduction studies of plantar nerves [3][21][22]
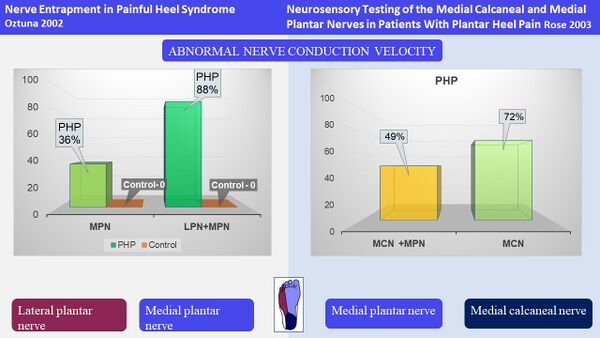
Both studies reported disturbances in nerve conduction of these nerves. Öztuna et al.[21] found a difference in nerve conduction between individuals with PHPS and those without and reported that 88% of participants had disturbances in nerve conduction of the MPN and LPN combined. Rose et al.[22] found disturbances in nerve conduction of the MCN in 72% of participants (Figure 16).
Figure 16. Results of nerve conduction studies [3][21][22]
Rose et al.[22] further suggest that the reduction in nerve conduction velocity was caused by pressure on the nerve somewhere along its course down the leg before reaching the foot/heel. This supports the findings of Coppieters et al.[14] of the neural tissue involvement in PHPS as seen during the modified SLR neurodynamic test with tibial nerve bias. It is also interesting that a nerve block to the posterior tibial nerve is suggested as a treatment option for individuals suffering from severe PHP, which further supports the involvement of the nerves in PHP.[3]
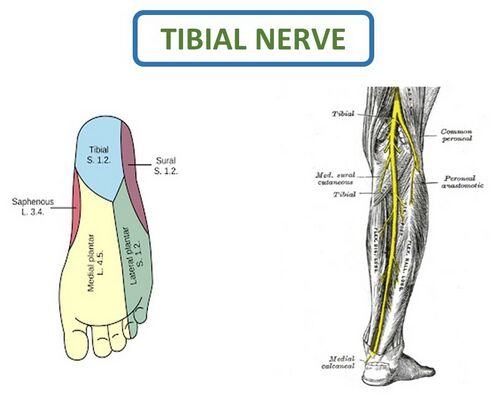
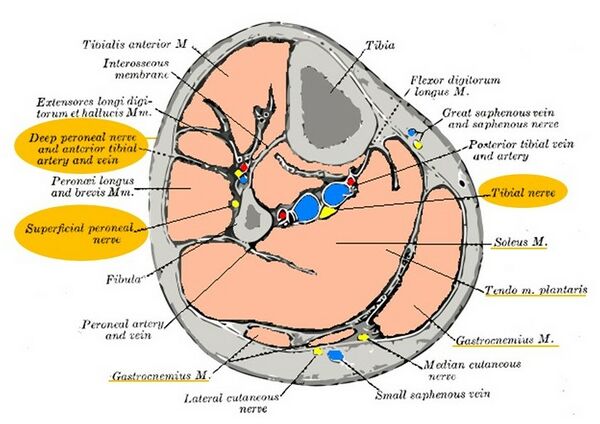
The theory of pressure on the tibial nerve necessitates further study of its course down the leg.[22] After exiting the popliteal fossa, the tibial nerve travels down the posterior leg between the superficial and deep muscles of the calf where the muscles could potentially cause pressure on the nerve. These are the same muscles that are found to be stiff, incompliant and painful on manual palpation in the study by Saban and Deutscher.[24] This suggests that these muscles might in fact be disrupting the conduction of the nerve along its course (Figure 17).[3] When looking at a cross-sectional image of the calf, it is clear how intertwined the nerves and muscles are, which highlights the possibility that muscles are putting pressure on the nerve (Figure 18).
Figure 17. Course of the Tibial nerve
Figure 18. Cross-sectional image of the lower leg
Conclusion[edit | edit source]
It remains unclear when the first dysfunction would appear in this interaction between the nerves and the muscles. Did the:
pain in the foot ⇒ patient walk differently ⇒ dysfunction of the muscle ⇒ disturbance of the nerve
Or did it happen the other way around? Even though the sequence of events might still be unknown, it is clear that there is a treatment available and the treatment is supported by sound theory. The next course in this series will look specifically at the treatment options proposed in the new protocol.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 Gatt A, Agarwal S, Zito PM. Anatomy, Fascia Layers. In: StatPearls. StatPearls Publishing, Treasure Island (FL); 2020. PMID: 30252294.
- ↑ Schleip R, Gabbiani G, Wilke J, Naylor I, Hinz B, Zorn A, Jäger H, Breul R, Schreiner S, Klingler W. Fascia is able to actively contract and may thereby influence musculoskeletal dynamics: a histochemical and mechanographic investigation. Frontiers in physiology. 2019 Apr 2;10:336.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 3.19 3.20 3.21 3.22 3.23 Bernice Saban. Anatomy and Relevant Structures in Plantar Heel Pain. Plus Course. 2021
- ↑ 4.0 4.1 4.2 4.3 Bourne M, Varacallo M. Anatomy, bony pelvis and lower limb, foot fascia. Europe PMC. StatPearls [Internet]. StatPearls Publishing, Treasure island (FL): Sept 26, 2018
- ↑ Chen Hua-you, Ma Ji-yuan, Pan Li-ya, Tian Wen, Hong Yang, Qin Xiang-zheng. Anatomy of the plantar fascia. Acta Anatomica Sinica. 2017 Oct 6;48(5):561-564.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 Stecco C, Corradin M, Macchi V, Morra A, Porzionato A, Biz C, De Caro R. Plantar fascia anatomy and its relationship with Achilles tendon and paratendon. Journal of anatomy. 2013 Dec;223(6):665-76.
- ↑ 7.0 7.1 7.2 7.3 Draghi F, Gitto S, Bortolotto C, Draghi AG, Belometti GO. Imaging of plantar fascia disorders: findings on plain radiography, ultrasound and magnetic resonance imaging. Insights into imaging. 2017 Feb;8(1):69-78.
- ↑ 8.0 8.1 Guo J, Liu X, Ding X, Wang L, Fan Y. Biomechanical and mechanical behavior of the plantar fascia in macro and micro structures. Journal of biomechanics. 2018 Jul 25;76:160-6.
- ↑ Young JR, Sternbach S, Willinger M, Hutchinson ID, Rosenbaum AJ. The etiology, evaluation, and management of plantar fibromatosis. Orthopedic research and reviews. 2019;11:1.
- ↑ Shiotani H, Yamashita R, Mizokuchi T, Naito M, Kawakami Y. Site-and sex-differences in morphological and mechanical properties of the plantar fascia: A supersonic shear imaging study. Journal of biomechanics. 2019 Mar 6;85:198-203.
- ↑ 11.0 11.1 Fessel G, Jacob HA, Wyss CH, Mittlmeier T, Müller-Gerbl M, Büttner A. Changes in length of the plantar aponeurosis during the stance phase of gait–an in vivo dynamic fluoroscopic study. Annals of Anatomy-Anatomischer Anzeiger. 2014 Dec 1;196(6):471-8.
- ↑ Zwirner J, Zhang M, Ondruschka B, Akita K, Hammer N. An ossifying bridge–on the structural continuity between the Achilles tendon and the plantar fascia. Scientific reports. 2020 Sep 3;10(1):1-0.
- ↑ 13.0 13.1 13.2 13.3 13.4 Coppieters MW, Kurz K, Mortensen TE, Richards NL, Skaret IÅ, McLaughlin LM, Hodges PW. The impact of neurodynamic testing on the perception of experimentally induced muscle pain. Manual therapy. 2005 Feb 1;10(1):52-60.
- ↑ 14.0 14.1 14.2 14.3 Coppieters MW, Alshami AM, Babri AS, Souvlis T, Kippers V, Hodges PW. Strain and excursion of the sciatic, tibial, and plantar nerves during a modified straight leg raising test. Journal of Orthopaedic Research. 2006 Sep;24(9):1883-9.
- ↑ Alatassi R, Alajlan A, Almalki T. Bizarre calcaneal spur: A case report. International journal of surgery case reports. 2018 Jan 1;49:37-9.
- ↑ Ahmad J, Karim A, Daniel JN. Relationship and classification of plantar heel spurs in patients with plantar fasciitis. Foot & ankle international. 2016 Sep;37(9):994-1000.
- ↑ 17.0 17.1 17.2 17.3 Saban B, Masharawi Y. Pain threshold tests in patients with heel pain syndrome. Foot & ankle international. 2016 Jul;37(7):730-6.
- ↑ 18.0 18.1 18.2 18.3 18.4 Butler DS. The sensitive nervous system. South Australia: Noigroup publications. 2000.
- ↑ 19.0 19.1 19.2 Zhang Y, He X, Li J, Ye J, Han W, Zhou S, Zhu J, Wang G, Chen X. An MRI study of the tibial nerve in the ankle canal and its branches: a method of multiplanar reformation with 3D-FIESTA-C sequences. BMC Medical Imaging. 2021 Dec;21(1):1-1.
- ↑ Warchol Ł, Walocha JA, Mizia E, Bonczar M, Liszka H, Koziej M. Ultrasound guided topographic anatomy of the medial calcaneal branches of the tibial nerve. Folia Morphol. 2020 Jun 3.
- ↑ 21.0 21.1 21.2 21.3 21.4 21.5 Öztuna V, Özge A, Eskandari MM, Çolak M, Gölpinar A, Kuyurtar F. Nerve entrapment in painful heel syndrome. Foot & ankle international. 2002 Mar;23(3):208-11.
- ↑ 22.0 22.1 22.2 22.3 22.4 22.5 22.6 22.7 Rose JD, Malay DS, Sorrento DL. Neurosensory testing of the medial calcaneal and medial plantar nerves in patients with plantar heel pain. The Journal of foot and ankle surgery. 2003 Jul 1;42(4):173-7.
- ↑ Alshami AM, Souvlis T, Coppieters MW. A review of plantar heel pain of neural origin: differential diagnosis and management. Manual therapy. 2008 Apr 1;13(2):103-11.
- ↑ Saban B, Deutscher D, Ziv T. Deep massage to posterior calf muscles in combination with neural mobilization exercises as a treatment for heel pain: a pilot randomized clinical trial. Manual therapy. 2014 Apr 1;19(2):102-8.